
Remembering a Champion, a Philanthropist, a Friend
John J. McDonough was a self-made man with a successful career in business and finance. But to us, he will be remembered for his service to Breakthrough T1D and his perseverance in moving life-changing research forward for the many people living with type 1 diabetes (T1D) today. John served on our International Board of Directors […]

Game Over, T1D! Fundraiser Brings Video Game Community Together in Support of Breakthrough T1D
From August 13–22, Breakthrough T1D Game2Give held Game Over, T1D, a livestream fundraising event that raised over $60,000 for type 1 diabetes (T1D) research.

Vertex Pharmaceuticals Investigating Cell Therapy for Type 1 Diabetes
Vertex is currently enrolling for a clinical trial evaluating their stem cell derived islet cell infusion therapy, VX-880.

Back to School with Diabetes Amidst the COVID-19 Variants
Kids with diabetes need to get back to school, but how can we best keep them safe? Experts weigh in.

Breakthrough T1D & NIH Workshop at the Intersection of Immunology and Bioengineering
Breakthrough T1D and NIH bring together top scientists and engineers to strategize on how to allow implanted beta cells to thrive.

Kid-Approved Back-to-School T1D-Friendly Recipes
Tasty, easy-to-make diabetes-friendly treats for school lunches, after school snacks, and school night dinners.

Abbott FreeStyle Libre 2 iOS App Receives FDA Approval
On August 2, 2021, Abbott announced the FreeStyle Libre 2 iOS app received approval from the FDA for adults and children ages 4 and up with diabetes.

FDA Approves Semglee® as the First Interchangeable Biosimilar Insulin Product
The U.S. Food and Drug Administration (FDA) approved Semglee® (insulin glargine-yfgn) as the first ever interchangeable biosimilar insulin product. Semglee® (insulin glargine-yfgn) is indicated to improve glycemic control in adults and pediatric people with type 1 and adults with type 2 diabetes, and is biosimilar and interchangeable to Lantus®, a long-acting insulin. In other words, […]

Two Studies Shed Light on Diabetic Kidney Disease
Two recent publications shed light on which people with type 1 diabetes will develop end-stage kidney disease, a complication of diabetes.
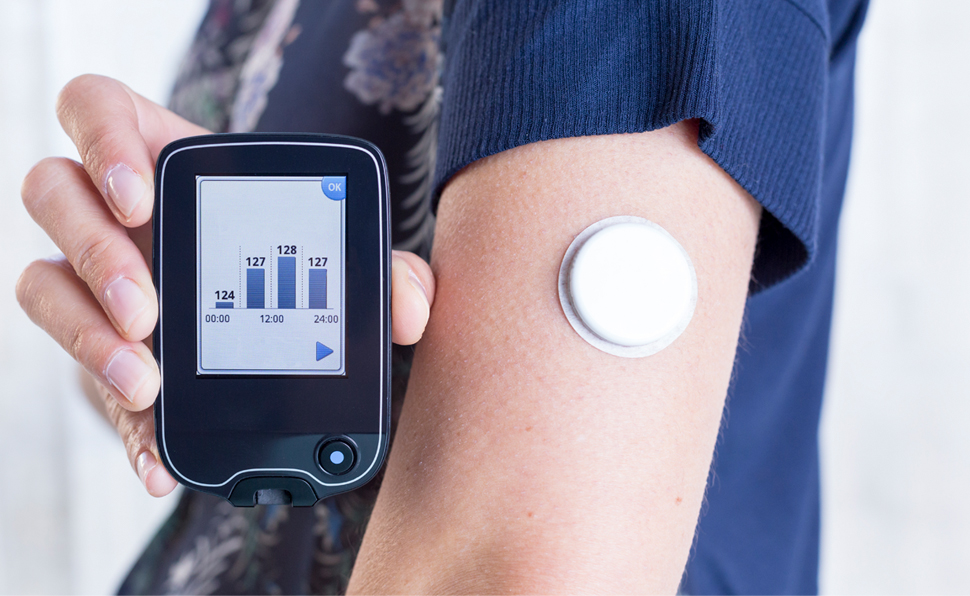
On Medicare? Accessing CGMs Just Got Easier
A recent update to continuous glucose monitor (CGM) coverage requirements will make it easier for people covered by Medicare to access CGM technology.