Islet cell therapies for type 1 diabetes (T1D) replace destroyed insulin-producing beta cells with functional cells that allow the body to make insulin again. To ensure there are enough islet cells for everyone with T1D who wants them, Breakthrough T1D is prioritizing manufactured cell therapies, which can be made in large quantities in a laboratory, adhering to high levels of quality control.
These therapies are quickly advancing through the drug development pipeline. How are we making sure the clinical community is prepared for their arrival?
The answer: Centers of Reference.
Centers of Reference
Centers of Reference are expert multidisciplinary T1D care centers that are preparing healthcare professionals (HCPs) around the world to administer manufactured islet cell therapies to people with T1D. The goal is to make sure that expert clinical teams—who are already doing islet cell transplants at low scale—are ready to integrate these therapies into clinical practice at their institutions and train others, once they have regulatory approval.
The second annual Centers of Reference meeting was held at Breakthrough T1D HQ in early May by our Medical Affairs team. Read on to learn more about the attendees, what the meeting covered, and what we can expect next year.
Key Takeaways: Breakthrough T1D’s Second Annual Centers of Reference Meeting
- Seven international Centers are established, and four interprofessional working groups meet every month to keep progress moving.
- Attendees, which include the leading physicians and islet transplant support teams from the U.S., Canada, UK and Italy, continued the conversation around what kinds of education, training, resources, clinical care, and benchmarking are needed for Centers to be effective.
- A manuscript is underway to lay out a clinical implementation roadmap for Centers.
Assembling the team at Breakthrough T1D HQ
This meeting was hosted by Thomas Danne, M.D., Ph.D., Chief Medical Officer International, Brynn Marks, M.D., Senior Director of Medical Affairs, Alessandro Bisio, M.D., International Director of Medical Affairs, and Amara Ogbonnaya-Whittlesey, M.D., U.S. Director of Medical Affairs, who brought together the best and brightest minds who can make Centers of Reference a reality.
Physicians, transplant surgeons, nurses, scientists, and other HCPs from various global medical institutions were in attendance, coming from:
- University of Minnesota Medical Center
- University of Wisconsin Health Transplant Center
- The Penn Rodebaugh Diabetes Center
- University of Chicago Medicine
- IRCCS Ospedale San Raffaele (Italy)
- Institute of Transplantation, Newcastle upon Tyne (UK)
- Edmonton, University of Alberta (Canada)
Additional attendees from Breakthrough T1D included CEO Aaron Kowalski, Ph.D., Senior Vice President of Research Esther Latres, Ph.D., multiple members of our Medical Affairs and Advocacy teams, and two members of our Participant Advisory Council who are living with T1D.
What has changed since last year’s meeting?
- There are now seven global Centers of Reference: Edmonton, Canada; Newcastle, UK; Milan, Italy; Wisconsin, Chicago (IL), Minnesota, and Pennsylvania in the U.S.
- Steady progress has been made toward a clinical roadmap for the Centers of Reference model.
- Four interprofessional working groups were established, co-led by Melena Bellin, M.D. (Minnesota) and Jim Shaw, M.D., Ph.D. (Newcastle, UK), that meet virtually every month and in-person annually at Breakthrough T1D HQ.
- The four working groups are HCP Training and Education, Clinical Access to Cell Therapy, Harmonizing Clinical Care, and Benchmarking and Registries.
Convening experts with the power to make real change
The objective of this meeting was to build on the progress that the working groups have made over the past year. Each group focuses on a key aspect of the development of Centers of Reference, taking a divide-and-conquer approach to capitalize on experts’ strengths and workshop ideas with the larger team.
We have learned recently that when a first-of-its-kind therapy comes to market, it isn’t magically going to be adopted by physicians and administered to people. There is a significant amount of prep work that has to happen to ensure that every connection is made so that these therapies, and ultimately people with T1D, succeed.
This meeting is that work, led by the leaders in the field.

Working group #1: HCP Training and Education
The group identified which topics around islet cell therapies should be included in educational programs for aspiring T1D medical professionals, including eligibility for islet cell therapies, the transplant procedure itself, post-transplant care, and long-term monitoring. Importantly, the ultimate career path of the trainee should play a factor in which topics require further education versus a brief overview. For example, a psychologist working with the T1D community may not have the same educational needs as a transplant surgeon, but both should understand T1D cell therapy basics.
Working group #2: Clinical Access to Cell Therapies
This group focused on how to best educate HCPs and people with T1D about emerging islet cell therapies. The team found that most people who receive cell therapies self-refer, but misconceptions are a major barrier to pursuing them. Priority areas for increasing awareness in the T1D community are communicating through social media, creating an easy-to-use landing page or website with information and opportunities to connect with medical teams, and connecting people with peers who have received islet cell transplants. In terms of HCPs, the group is focused on creating referral pathways, using artificial intelligence to help establish clinical workflows for islet cell transplants, and utilizing professional organizations to connect with and educate the T1D medical community.
Working group #3: Harmonizing Clinical Care
This group has one of the most challenging tasks: creating a roadmap for the clinical implementation of islet cell therapies. This includes determining eligibility based on defined clinical measures, reaching consensus on the best way to approach care before, during, and immediately after islet cell therapy, and consistent long-term monitoring. To add to the challenge, this roadmap must be applied across diverse clinical settings in different countries. The experts in the room—some of the most prominent T1D physicians and transplant surgeons in the world—made significant progress in defining clinical measures and tools that should be used in islet cell transplants.
Working group #4: Benchmarking and Center Readiness
Centers of Reference will be held to the highest standards to ensure they are providing top-quality clinical care to people receiving islet cell transplants. To make this possible, this group created a framework for assessing Center readiness, collaborating with registries for data-sharing, and benchmarking to evaluate Center effectiveness. This includes development of specific criteria laying out what each Center needs, such as a complete islet cell therapy care team, established processes for quality and safety, research teams, and more.
One theme was clear throughout nearly all of the working groups: incorporating the perspectives of people with T1D is absolutely critical. The groups agreed that clinical decision-making around islet cell therapies is about more than just someone’s blood sugar—the way a person feels, their level of diabetes distress, and their individual circumstances are key factors to integrate into shared decisions between people and their care teams.
Over the next 12 months, each team will follow up on the challenges, solutions, and immediate next steps identified during the meeting to keep progress moving. Thanks to everyone’s hard work over a productive and exciting two days, we are accelerating faster than ever toward the development of expert Centers of Reference.
What the experts are saying

“Cell therapies are getting closer and closer to reality, and our partners in the clinic must be ready to implement them. This is a good problem to have! We know that new therapies will take far too long to reach the people that need them if we don’t have an established clinical infrastructure. We have the best people in the world in the room, working through the problem with us, to ensure that when these therapies do become an approved therapy option, the T1D community can benefit as soon as possible.”
-Thomas Danne, M.D., Ph.D., Chief Medical Officer International at Breakthrough T1D
“Our hope is that Centers of Reference enable preparedness and expedite the delivery of new cell therapies—as they are approved—to become immediately accessible to people with T1D most in need of a novel intervention for the treatment of their diabetes.”
-Michael Rickels, M.D., M.S., Medical Director, Pancreatic Islet Cell Transplant Program, Hospital of the University of Pennsylvania


“Breakthrough T1D is proactively working now to identify and remove current and future barriers to T1D cell therapy coverage. By working with payers and policymakers now, we will accelerate broader access to T1D cell therapies.”
-Aaron Turner-Phifer, Senior Director of Health Policy at Breakthrough T1D
“Centers of Reference can help prepare healthcare systems around the globe for future cell therapies in T1D by building the expertise and standards needed to deliver them safely. They also help share experiences and best practices so more hospitals are ready to adopt these treatments over time.”
-Carmen Hurtado del Pozo, Ph.D., Director of European Research at Breakthrough T1D

Steady progress towards our goal
Manufactured islet cell therapies are coming. It is not a matter of if, but when. We are at a critical moment and need to ensure that HCPs are ready. Teamwork will get these therapies into clinics so people with T1D don’t have to wait years to get them.
This is why Breakthrough T1D is acting now: when the first manufactured islet cell therapy becomes commercially available, multidisciplinary care teams around the world will be prepared. This is essential to our Project ACT initiative, which is accelerating islet cell therapies that do not require immunosuppression for the T1D community.
Our goal is to advance islet cell transplants and establish Centers of Reference around the world, so that people anywhere can access these therapies as they become available. Centers of Reference are leading the way for safe and effective integration of manufactured cell therapies into clinics. These annual meetings provide a platform for international experts to connect and refine these Centers over two days of idea-sharing and collaboration. We’re already looking forward to seeing what the team will have accomplished by this time next year!
Our Statement on Deceased Donor Islet Cell Availability & The ISLET Act
Curing type 1 diabetes (T1D) is Breakthrough T1D’s number one priority. As the leading global T1D research and advocacy organization, we are currently funding over $450 million in cures research in 23 countries. Everything we do is focused on making cures happen for the T1D community, sooner.
We are very excited to see the trials we fund having such positive results, including University of Chicago’s trial testing Eledon’s tegoprubart. This trial uses deceased donor islets, which come from the pancreases of deceased organ donors. We know we need more deceased donor islets available so more eligible adults can have access to islet cell transplants. To achieve this pressing goal, we have urged the Department of Health and Human Services (HHS) to reclassify deceased donor islet cells from biologics (drugs) to organs in the United States.
We are very grateful to the sponsors and supporters of the ISLET Act for their engagement on deceased donor islet cell transplantation and for helping to generate a discussion about increasing deceased donor islet cell availability.
Passing legislation is extremely difficult at best, and the ISLET Act does not require development of safety standards for reclassified deceased donor islet cell transplants that would ensure the islet cells are healthy and safe for transplant. Nor does it address insurance coverage, which could limit access to these costly treatments.
Reclassifying deceased donor islet cells as organs can be accomplished using existing HHS authority. New legislation is not needed. And so, with the full support of leading islet transplant surgeons, we’re putting 100% of our effort toward working with HHS, which has much better odds of success.
If implemented, our proposal to HHS would increase deceased donor islet availability, ensure rigorous safety standards, and address insurance coverage.
Continued research funded by Breakthrough T1D and others has shown deceased donor islet cell transplants to be safe and effective for adults with T1D who experience severe hypoglycemia and hypoglycemia unawareness. These procedures can significantly reduce—or even eliminate—the need to administer insulin.
With the science clear and patients waiting, Breakthrough T1D is calling on HHS to use its existing authority to safely expand availability of deceased donor islet cell transplants in the United States.
Deceased Donor Islet Cell Transplantation 101
Here’s what you need to know about deceased donor islet cell transplants—and our plan to expand availability to them safely and responsibly.
What Is Deceased Donor Islet Cell Transplantation?
Deceased donor islet cell transplantation is a procedure in which insulin-producing islet cells are isolated from the pancreases of deceased donors and transplanted into a person living with type 1 diabetes (T1D). These islets are infused into the liver, where they can begin producing insulin in response to blood glucose levels.
Deceased donor islet cell transplants are intended for, and only FDA-approved for, adults with T1D who experience:
- Severe hypoglycemia (dangerous low blood sugar)
- Hypoglycemia unawareness (the inability to sense when blood sugar is dropping)
These individuals often cannot safely manage their blood glucose levels with the standard therapies used to manage T1D, including continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems.
Do They Work?
Yes. For adults with T1D who experience severe hypoglycemia and hypoglycemia unawareness, deceased donor islet cell transplantation has been shown to significantly reduce—or even eliminate—the need to take external insulin.
Do Deceased Donor Islet Cell Transplants Require Immunosuppression?
Yes. Recipients of deceased donor islet cell transplants must take immunosuppressives to prevent the body from rejecting the transplanted cells. Due to the side effects of immunosuppressives, these transplants are only approved for adults whose risk from severe hypoglycemia outweighs the risks associated with immunosuppression.
Breakthrough T1D is working on alternatives to traditional immunosuppression so more people can benefit. This includes our funding of the University of Chicago’s clinical trial with Eledon’s drug, tegoprubart.
Why Aren’t They Widely Available?
Deceased donor islet cells come from organ donors. A single transplant often requires multiple pancreases as isolating islet cells is a complicated process. In addition to those challenges, the availability in the U.S. is more limited than it is in other countries due to regulatory classification.
The U.S. currently regulates deceased donor islet cells as biological drugs requiring a Biologics License Application (BLA). This is different from how whole organ transplants are regulated in the U.S. and how deceased donor islet cells are regulated in many other countries, including Canada, the United Kingdom and countries in the European Union.
Our Strategy to Increase Availability
How Is Breakthrough T1D Advocating for Expanded Deceased Donor Islet Cell Transplant Availability?
Breakthrough T1D has strongly urged HHS to reclassify and expand availability of deceased donor islet cells.
Our proposal outlines a framework to ensure safety and accessibility for the T1D community. You can read the letter here.
We are putting 100% of our energy behind this approach, and we are doing it with the full support of leading transplant surgeons and organizations.
Why Is Breakthrough T1D Pursuing This Route (HHS) as Opposed to Legislation (ISLET Act)?
This is our best chance of success. Passing legislation is extremely difficult at best, and HHS has existing authority over the regulatory framework governing cell therapies and organ transplantation. They can make this change.
Additionally, the ISLET Act does not require the development of safety standards for reclassified deceased donor islet cell transplants, nor does it address insurance coverage, which could limit access to these costly treatments.
What Are the Details of Breakthrough T1D’s Recommendations in the Letter to HHS?
Our proposal outlines a clear strategy to ensure the safety and quality of deceased donor islet cells under this reclassification. They include:
- Designating Centers of Excellence staffed with experts in deceased donor islet cell transplants
- Strict quality control to ensure islets are functional—and they work
- A way to get cells from the deceased donors’ pancreases and into people with T1D
- A strategic roadmap for adding additional centers
Why Not Work with HHS and Support the ISLET Act?
Our Breakthrough T1D legislative agenda includes several critical priorities essential to advancing our mission. This includes the ongoing renewal of the Special Diabetes Program (SDP) to fund T1D research and efforts to improve insulin affordability. These issues require legislative action, which is why we continue to focus our legislative advocacy on them.
Not every priority is best achieved through Congress. Reclassifying deceased donor islet cells as organs, for example, can be accomplished under current law through the Department of Health and Human Services (HHS), without new legislation. This is one of the key reasons we believe the path through HHS is the best path for this issue.
Simply put, we are pursuing each of our priorities through the channel where it has the greatest likelihood of success. And given how difficult it is to pass legislation, putting 100% of our effort toward working with HHS has the best chance of success.
Why Doesn’t Breakthrough T1D Recommend an Amendment to the ISLET Act?
Given how difficult it is to pass legislation, we believe our effort to reclassify deceased donor islet cells as organs is much more likely to succeed focusing on HHS.
What effect would this have on stem cell-derived islets?
Breakthrough T1D believes that stem cell-derived islets are appropriately regulated by FDA as biologic products. There is no need to change the regulatory status for manufactured islets. If adopted, our proposal would not affect them.
Through Project ACT, Breakthrough T1D is working with all parties to clear the way for future, manufactured islet therapies and to ensure they are accessible.
Breakthrough T1D’s Role in Islet Cell Therapies
Has Breakthrough T1D Supported This Work?
Yes—Breakthrough T1D has been a leader in islet cell transplantation for decades. Breakthrough T1D is currently funding over $450 million in cures research in 23 countries, including the University of Chicago trial testing Eledon’s drug, tegoprubart. Breakthrough T1D’s support goes beyond funding these clinical trials. The T1D Fund: A Breakthrough T1D Venture, invested in Eledon in 2023.
What Is Breakthrough T1D’s Strategy to Increase Access?
In 2024, Breakthrough T1D launched a flagship initiative, known as Project ACT (Accelerate Cell Therapies), designed to advance breakthroughs in T1D cell therapies, an anticipated pathway toward cures for T1D. Learn more about our work within research, medical affairs, and advocacy via the timeline at the bottom of our Project ACT landing page.
Is Breakthrough T1D Alone in These Efforts?
No. Breakthrough T1D is working with other T1D and diabetes organizations in the U.S. and around the globe, and top islet transplant surgeons. They support our efforts and agree that this is the best way forward.
How Can I Get Involved in This Work?
If you are in the United States, sign up to be a grassroots advocate here to get real time policy updates, action alerts, and tips for contacting your lawmakers.
If you live with type 1 diabetes, you can also be part of advancing treatments like this by participating in clinical trials. Visit our dedicated T1D clinical trial match tool here.

Breakthrough T1D envisions a world where the burden of type 1 diabetes (T1D) no longer exists.
It’s a world where people don’t have to manage their diabetes—don’t take insulin, don’t wear devices, don’t have blood sugar highs and lows, and don’t develop complications. That’s how we define cures for type 1 diabetes, and there are three ways we can get to cures faster:
Cell therapies
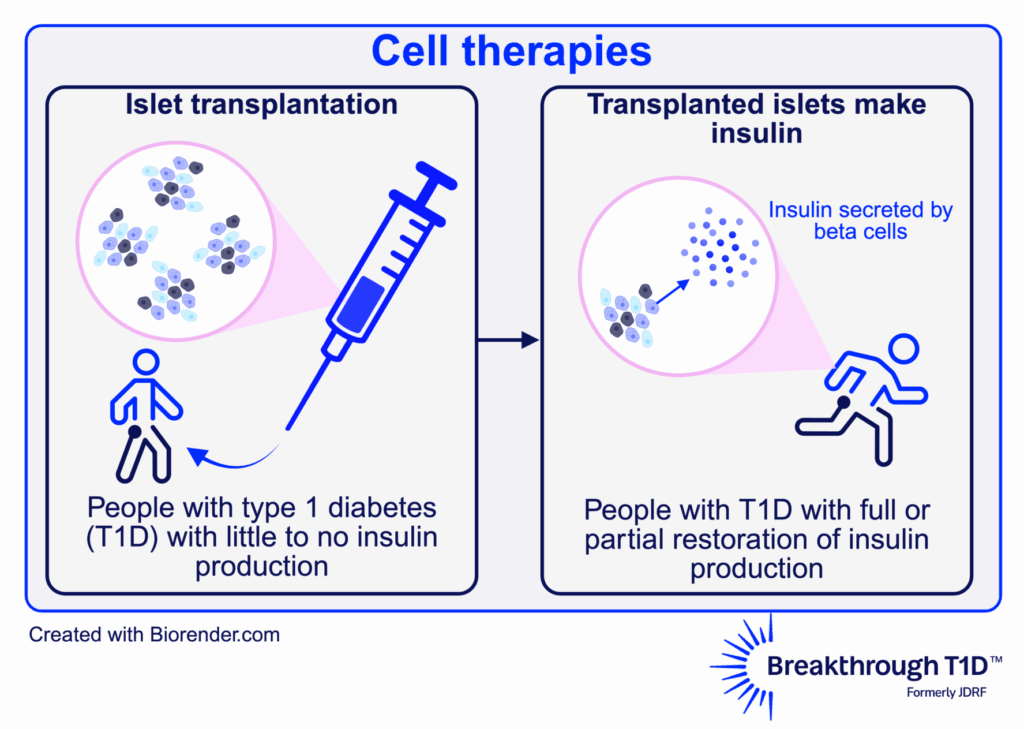
What are cell therapies?
In people with T1D, the immune system destroys the beta cells in the pancreas, which produce insulin. One known way to cure T1D is through cell therapies, which replace destroyed beta cells with external cells that make insulin and protect them so that they can function for a very long time.
What work is Breakthrough T1D doing in this area?
117
Number of preclinical cell therapies research projects we are supporting
5
Number of cell therapies clinical trials we are supporting
We are funding research to:
- Develop a renewable beta cell source so that every person with T1D who wants this treatment can get it.
- Find ways to maintain these beta cells so they stay functional in the body for years—or even decades.
- Shield these beta cells from immune attack to prevent the need for anti-rejection immunosuppressants or the recurrence of T1D.
What does the future hold for cell therapies?
Our ultimate goal is large-scale cell therapies with the fewest side effects possible, accessible by anyone with T1D who wants them. Our Project ACT initiative will get us there faster than ever. We believe that, in the future, these therapies will create a path for people with T1D to walk away from the condition for good.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To get cell replacement therapies to people with T1D faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Early detection

What is early detection?
Type 1 diabetes doesn’t show up overnight (even though for many people it feels like it does). The condition develops in stages over time:

From a therapy perspective, early detection is important for identifying people who could benefit from the FDA-approved disease-modifying therapy Tzield, which can delay T1D progression for years.
From a research perspective, early detection can open doors to research opportunities, like clinical trials of other disease-modifying therapies in development.
From the clinical perspective, early detection can prevent complications at diagnosis, like diabetic ketoacidosis (DKA), and help families and individuals prepare for T1D onset.
Screening is done through a simple blood test to detect T1D autoantibodies, which signal that the immune system may be attacking the body’s insulin-producing cells.
What work is Breakthrough T1D doing in this area?
52
Number of preclinical early detection research projects we are supporting
26
Number of early detection clinical trials we are supporting
Through our Advocacy and Medical Affairs efforts, we are:
- Supporting the SCREEN for Type 1 Diabetes Act
- Leading a campaign to expand access to early detection of T1D in the United States
- Providing accredited education and resources on T1D and early detection to healthcare professionals
What does the future hold for early detection of type 1 diabetes?
We expect to improve ways to detect T1D early and prevent it from advancing by attaining a better understanding of the natural progression of T1D, which may include additional biomarkers for the condition. We will continue to expand access to early detection and find ways to improve the standard of care for early-stage T1D.
Disease-modifying therapies
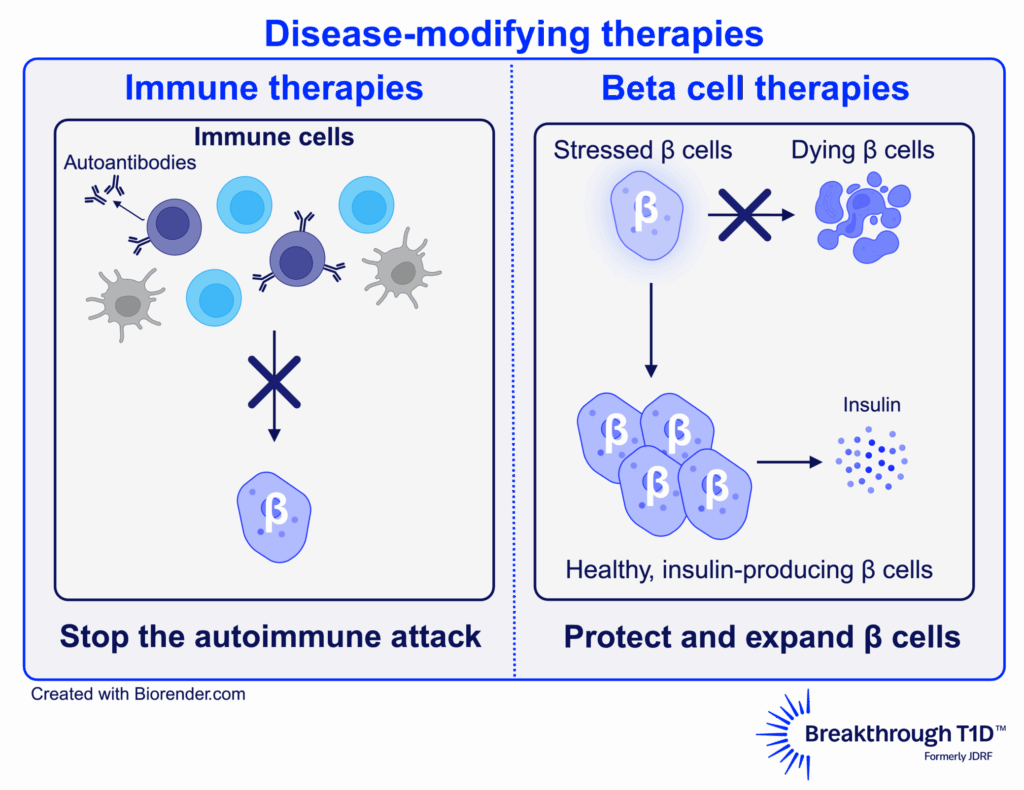
What are disease-modifying therapies?
T1D is a progression. At earlier stages, the immune system has begun attacking the insulin-producing beta cells, but people are not symptomatic yet. Some people at later stages have had T1D for decades and have lost nearly all insulin production. Disease-modifying therapies, or DMTs, are aimed at helping everyone with T1D—regardless of where they are on the disease continuum—to slow, halt, prevent, or reverse the condition.
What is Breakthrough T1D doing in this area?
138
Number of preclinical disease-modifying therapy research projects we are supporting
16
Number of disease-modifying therapies clinical trials we are supporting
We recently published research on the importance of C-peptide—a biomarker for insulin production—as a key measure of how effective a DMT is in clinical trials. Use of C-peptide will be essential to speeding up development and approval of new DMTs—getting potential cures to people with T1D faster.
What does the future hold for disease-modifying therapies?
As we see more DMT clinical trials advance to later-stage human testing, we expect to see additional DMTs submitted for regulatory approval—giving people with T1D more options.
Improving Lives
Cures for type 1 diabetes are coming down the pipeline. While we look forward to their arrival, Breakthrough T1D is improving the lives of people with T1D today.
There has never been a more exciting time for T1D research—cures for type 1 diabetes and other breakthroughs are within reach. With the support of the T1D community, we will succeed!

ADA Recap Series
This article is the second of our three-part ADA Recap Series. Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’re here to report on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding. Look out for tomorrow’s article for updates on Medical Affairs.
Cures
Breakthrough T1D’s Cures program focuses on early detection, disease-modifying therapies, and cell therapies with the goal of working toward effective cures for T1D.
Cell therapies were front-and-center at ADA 2025. We have some exciting clinical trial updates and new ideas for optimizing islet transplantation.
Cell therapies

Autologous cell transplantation
Autologous cells are those removed from an individual and implanted back into the same individual. These cells can be modified in a laboratory before implantation. Autologous cells are still susceptible to autoimmunity in T1D, so cell protection strategies (gene-editing, encapsulation, immune modulation, etc.) are expected to be required.

Allogenic cell transplantation
Allogenic cells are those that are derived from a source other than the recipient, such as deceased donors or precursor-derived manufactured cells. Allogenic cell transplants require immunosuppression because they stimulate an immune response. Breakthrough T1D’s Cell Therapies program is focused on allogenic cells—specifically manufactured cells—because they can be generated at large scale.
One-year updates on Vertex’s manufactured cell therapy, zimislecel
- Presenter: Michael Rickels, M.D. (University of Pennsylvania)
- Zimislecel (VX-880) is a manufactured islet therapy that requires immunosuppression, infused into a vein in the liver in people with T1D who have impaired hypoglycemic awareness and severe hypoglycemic events.
- The phase 1/2 clinical trial, which is part of the pivotal phase 1/2/3 FORWARD-101 trial, is complete. Twelve participants received a single infusion of a full dose of cells and were followed for at least one year.
- All 12 participants achieved the primary endpoint, which was elimination of severe hypoglycemic events and HbA1c levels less than 7%. 10/12 (83%) participants are insulin independent.
- All 12 participants demonstrated sustained insulin production as measured by C-peptide, reduced external insulin therapy use, and achieved greater than 70% time in range.
- There were no serious adverse events. Mild to moderate adverse events were consistent with the immunosuppression regimen, infusion procedure, and complications from T1D.
- These data were published in the New England Journal of Medicine and represent further evidence of the curative potential of manufactured islet transplantation for T1D.
- Breakthrough T1D’s support for Doug Melton, Ph.D.—whose proprietary lab-created beta cells are now being advanced by Vertex—goes back decades, both via research grants and an investment from the T1D Fund: A Breakthrough T1D Venture.
6-month update on Sana Biotechnology’s immune-evasive islets
- Presenter: Per-Ola Carlsson, M.D., Ph.D. (Uppsala University)
- Sana’s donor-derived islet therapy engineered with Hypoimmune (HIP) technology can evade the immune system without immunosuppression.
- These cells were implanted intramuscularly in a first-in-human study into a person with T1D with no measurable insulin production.
- Six months post-transplant, this person is consistently making their own insulin, as measured by C-peptide levels. Yet, they still require external insulin therapy because they received a smaller dose of cells than the dose that would be required to achieve insulin independence. They did not experience any serious side effects, so the cells and procedure are safe and well-tolerated.
- A Mixed Meal Tolerance Test (MMTT) confirmed that these cells are not only surviving but also responding to changes in blood glucose levels.
- This is a promising first step toward a functional cure for T1D that does not require immunosuppression. Sana Biotechnology is planning on applying this technology to manufactured islets.
- Sana has received support from the T1D Fund to advance their HIP technology in islets, and Breakthrough T1D continues to work closely with them.
A new transplantation site for autologous manufactured islets
- Presenter: Hongkui Deng, M.D. (Peking University)
- Cells derived from adipose tissue (fat) can be removed from a person and chemically induced in the laboratory to become islet cells.
- Implantation of autologous manufactured islets into the sub-anterior rectus sheath in preclinical models of T1D improves glycemic control.
- In humans, this implantation site is easily accessible by an ultrasound-guided needle.
- In a first-in-human study, autologous manufactured islets were implanted into this site in a person with T1D. This person no longer needs external insulin therapy and has greatly improved blood glucose control. This person had also received a liver transplant and was taking immunosuppressants.
A new encapsulation device for immune protection of transplanted islets
- Presenter: Nicolas Laurent, Ph.D. (Adocia)
- Adoshell® is a novel islet cell encapsulation device that can shield islets from the immune system, meaning that immunosuppressants are not needed.
- The hydrogel-based device is non-degradable, easily retrievable, and allows the exchange of glucose and insulin from the vasculature surrounding the device while excluding immune cells from encapsulated islets based on pore size.
- This device showed promise in animal models, and human clinical testing is next.

Cell therapies highlight: Breakthrough T1D-funded research
Tom Bollenbach, Ph.D. (Advanced Regenerative Manufacturing Institute; ARMI) presented on challenges and solutions for large-scale manufacturing of islet cells. The goal is to generate scalable, automated manufactured islets that can be used by labs around the world to accelerate research progress using a unique, validated, and reliable cell source. Some challenges include ensuring the manufactured cells can survive shipment from the facility to research labs and maintain their insulin-producing capacity when they are used in different labs. ARMI is working with the Beta Cell Replacement Consortium to address these challenges.
Antonio Citro, Ph.D. (San Raffaele Hospital) presented his work on ensuring that transplanted islets have enough oxygen and nutrients (“vascularization”) to survive and function. Dr. Citro described an approach tested in animal models referred to as “natural scaffolds” in which all cells are removed from a donor organ, such as a lung, leaving behind blood vessel structures and other structural components. Islet cells can be injected and grafted onto this scaffold to create a mini organ of islet cells, which can then be tested for functionality before and after implantation.
Andrew Pepper, Ph.D. (University of Alberta) also presented vascularization strategies to increase the survival and functionality of transplanted islets. The implantation of a biomaterial under the skin will trigger the immune system and initiate a foreign body reaction, which results in the formation of blood vessels and structural components around the foreign object. Removal of the object leaves a hollow pre-vascularized core suitable for islet transplantation. This process can be optimized by using a biodegradable material, so no removal is required, and by the addition of “accessory cells” that help maintain a vascularized environment for islets.
Key takeaways
Cell therapies are making significant headway in clinical trials, and people receiving manufactured cells are becoming insulin independent. Researchers are tackling the biggest challenges for optimizing islet transplantation, including large-scale manufacturing, ensuring cell survival, and preventing detection by the immune system.
Disease-modifying therapies
A major focus at ADA 2025 was addressing the underlying immune mechanisms of T1D—including alterations in immune cells that facilitate beta cell destruction and other factors that contribute to autoimmunity onset. Read on for some highlights.
The role of B cells in T1D autoimmunity
- Presenter: Mia Smith, Ph.D., DVM (University of Colorado)
- B cells are a type of immune cell that can activate destructive immune cells that facilitate autoimmunity in T1D.
- B cells can become wrongly activated against insulin-producing beta cells due to converging dysregulation of factors that regulate immunity.
- These cells represent another potential target for disease-modifying therapies in T1D.
Disease-modifying therapies highlight: Breakthrough T1D-funded research
Emrah Altindis, Ph.D. (Boston College) presented on the role of the gut microbiome in T1D. His studies found that people with T1D tend to have more inflammatory bacteria in their gut microbiome. Dr. Altindis and his team identified a particular bacterial population that can enhance T1D onset in animal models due to changes in immune cells that contribute to T1D autoimmunity. These studies provide insight into additional factors that can drive immune changes in T1D.
Laura Sanz Villanueva, MSc (St. Vincent’s Institute of Medical Research), who works in the lab of Breakthrough T1D-funded researcher Professor Thomas Kay, MBBS, Ph.D., presented on a mechanistic follow-up study to the BANDIT clinical trial. The Breakthrough T1D-funded phase 2 BANDIT study showed that baricitinib, a JAK1/2 inhibitor that prevents immune cell communication, can increase insulin production as measured by C-peptide in people with recently diagnosed T1D. The present study found that baricitinib can reduce the number of natural killer (NK) cells in the pancreas, which are involved in the autoimmune destruction of beta cells. These data provide valuable insight into the mechanism of baricitinib-mediated protection of beta cells.
Key takeaways
T1D is driven by dysregulation of the immune system, which results in an attack on insulin-producing beta cells. Researchers at ADA 2025 spoked about novel factors—including B cells, NK cells, and T1D autoimmunity triggers—that may contribute to immune cell dysfunction in T1D.
Early detection
A key focus at ADA 2025 was the growing recognition of the heterogeneity of T1D, including autoantibody-negative disease onset, genetic variation, and the frequent misdiagnosis of T1D in adults, underscoring the need for greater diversity and inclusion in research and care. The expanded role of continuous glucose monitoring (CGM) and continuous ketone monitoring (CKM) was also highlighted, not only for daily management but as essential tools for understanding disease progression.
Using genetics to predict T1D risk
- Presenters: Richard Oram, M.D., Ph.D. (University of Exeter), Leslie Lange, Ph.D. (University of Colorado), Aaron Deutsch, M.D. (Massachusetts General Hospital), Josep Mercader, Ph.D.(Massachusetts General Hospital) and Eimear Kenny, Ph.D. (Icahn School of Medicine at Mount Sinai)
- Polygenic risk scores (PRS) estimate the risk a person has for developing a disease like T1D based on variations in different genes.
- Ancestry is a major influence on PRS, particularly based on differences in genes that regulate whether the immune system can distinguish between “self” and “non-self.”
- Most PRS models have been developed using data from European populations and have a limited ability to accurately determine risk in other ethnic groups, such as individuals of African and East Asian descent.
- Potential applications of PRS include incorporation into screening to better understand T1D risk, ensure accuracy in diagnostic tests, and develop precision medicine-based therapeutic approaches.
Understanding how genetic diversity contributes to T1D
- Presenters: Suna Onengut-Gumuscu, Ph.D. (University of Virginia), Dominika A. Michalek, MS (University of Virginia), Aaron Deutsch, M.D. (Massachusetts General Hospital), and Stephen I Stone, M.D. (Washington University School of Medicine), among others.
- These talks highlighted several studies conducted in diverse populations to better understand the pathophysiology of T1D.
- Work presented from Consortia, such as RADIANT, focused on rare and atypical forms of diabetes.
Controversies in CGM and benefits for early detection
- Presenters: Peter Calhoun, Ph.D. (Jaeb Center for Health Research), Michael Kohn, M.D., MPP (University of California San Francisco), Nicole Ehrhardt, M.D. (University of Washington) and Tadej Battelino, M.D., Ph.D. (University of Ljubljana)
- CGM use holds value in identifying progression in early stages of T1D prior to symptomatic onset.
- There was a call to update the clinical guidelines so that the benefits of CGM can be maximized within the T1D community—including at early and later stages of T1D.
- Integrating newer measures of blood glucose, like the glucose management indicator (GMI) and time in tight range (TITR), will be essential.
Contributions of CKM to early detection
- Presenters: Ketan Dhatariya, MBBS, M.D., Ph.D. (Norfolk and Norwich University Hospitals), Lori Laffel, M.D., MPH (Harvard University), Jennifer Sherr, M.D., Ph.D. (Yale University), and Richard Bergenstal, M.D. (HealthPartners Institute).
- It will be critical to explore whether ketone monitoring could help reduce the incidence of diabetic ketoacidosis (DKA) at stage 3 clinical T1D onset.
- Early detection of rising ketones will be important for people with T1D to take action before DKA occurs.
Early detection highlight: Breakthrough T1D-funded research and awardees
Brigitte Frohnert, M.D., Ph.D. (Barbara Davis Center for Diabetes), co-investigator with Breakthrough T1D-funded researcher Andrea Steck, M.D., presented on the evolution of CGM patterns prior to stage 3 T1D. Their longitudinal approach, with CGM data collected at three-month intervals, successfully distinguished individuals who progressed to stage 3 T1D clinical onset from those who did not. This research suggests that CGM may be beneficial for predicting clinical progression of T1D.
Anette-Gabriele Ziegler, M.D. (Helmholtz Munich) was awarded the Harold Hamm Prize for Biomedical Research in Diabetes 2025, highlighting her important contributions on T1D screening in the pediatric population.
Barbara B Kahn, M.D. (Beth Israel Deaconess Medical Center and Harvard Medical School) received the 2025 Albert Renold Award, and her role model for women in science was highlighted at the Women’s Interprofessional Network of the ADA (WIN ADA).
Key takeaways
T1D arises in diverse ways across age groups and ancestries, complicating diagnosis and treatment. At ADA 2025, experts highlighted how genetic and clinical heterogeneity demands more inclusive strategies. Tools like CGM and CKM were highlighted for their potential to enhance clinical management in the early stages of T1D.

Breakthrough T1D’s Cures team making an impact
Sanjoy Dutta, Ph.D., Chief Scientific Officer, participated in a panel discussion titled “Encapsulation vs. Naked Cell Therapy—Immune Challenges and Beta-Cell Perspectives in Diabetes Treatment.” The panelists covered different approaches to preventing immune attack of transplanted islets, including gene-editing, in addition to pros and cons of different transplantation sites.
Esther Latres, Ph.D., Vice President of Research, and Jay Tinklepaugh, Ph.D., Senior Scientist, hosted a workshop immediately after ADA titled “Islet Cells in T1D.” Breakthrough T1D-funded researchers gathered from around the world to discuss cell therapy clinical trial updates, mechanistic insights into islet function, targeted delivery of therapeutics to beta cells, and optimization strategies for islet transplantation.
Look out for tomorrow’s article for an update on Medical Affairs presented at ADA 2025!
We’ve made major progress in the development of cell replacement therapies for type 1 diabetes (T1D) over the past few decades. We know that manufactured islets can be safely implanted into people and produce insulin. Yet, there is more work to do to advance cell therapy research and bring these therapies to the larger T1D community. Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) will make manufactured cell replacement therapies that do not require broad immunosuppression a reality, faster.
Project ACT
To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
The Breakthrough T1D x Stem Cell Network Partnership
To drive innovation in manufactured islet therapies, Breakthrough T1D, Breakthrough T1D Canada, and the Stem Cell Network (SCN) have partnered to support four new projects led by Canadian researchers. The organizations issued a joint Request for Applications and together will maximize resources to drive high-impact research into manufactured cells. This partnership is a novel and meaningful part of Breakthrough T1D’s global Project ACT effort to power high-impact cell therapies research.

“Accelerating cell therapies is a central focus of Breakthrough T1D’s research strategy to drive toward cures for type 1 diabetes,” said Breakthrough T1D Vice President of Research Esther Latres, Ph.D. “We’re excited to join Stem Cell Network and Breakthrough T1D Canada in funding these outstanding cell therapy researchers and projects that can build on the current momentum to overcome barriers and advance cell therapies that can benefit all those who live with type 1 diabetes.”
These projects will receive support from May 2025 to April 2027. They are a part of a broader SCN investment totaling more than $33 million to support 36 regenerative medicine research projects and clinical trials.
Read on to learn more about the exciting, newly funded projects.
A closer look at the projects
The project
Combining manufactured islets and vasculature for a better islet replacement product
Fueling Biotechnology Partnerships Award
The team
Dr. Tim Kieffer (UBC), Dr. James Shapiro (University of Alberta), Dr. Takanori Takebe (Cincinnati Children’s Hospital), & Lunar Therapeutics (Vancouver, BC)
Current cell therapies for T1D, while often effective, are hampered by reliance upon donor-derived cells and poor cell survival after transplant, necessitating large doses of cells and repeat procedures. This ambitious new project will address both the source of islet cells and the low cell survival rates associated with islet transplantation by accelerating Lunar Therapeutics’ preclinical development of a manufactured islet replacement product. Takebe’s lab describes it as “complex miniature organs” for T1D.
This product will consist not only of insulin-producing cells, but also endothelial cells, which line blood vessels. Endothelial cells will support islet cell survival and engraftment upon transplantation.

Organoid
An organoid is a three-dimensional tissue grown in the lab that resembles an organ.
To accomplish this objective, Lunar Therapeutics will bring together Canadian expertise in manufactured islets and clinical islet transplantation led by Drs. Timothy Kieffer and James Shapiro. The team will also include U.S.-based Dr. Takanori Takebe, who specializes in designing complex organoids composed of various cell types. Using technologies developed across each laboratory, this multidisciplinary team will work to address challenges in islet cell transplantation.
The project
Using naturally-derived gels to optimize cryopreservation (extreme cold storage) of manufactured islets
Impact Award
The team
Dr. Marya Ahmed & Dr. James Shapiro (University of Alberta)
The implantation of manufactured islets into people with T1D can restore insulin production, eliminating the for external insulin and improving quality of life. However, after islet cells are derived from donors or manufactured in the lab, they must be stored before being used to treat a person with T1D. Currently, the storage and transportation of islet cells is difficult, and the only storage method is freezing at low temperatures in the presence of chemical solutions that help with the freezing process. However, these solutions cause cell death during thawing and may also cause allergic reactions in people after transplantation.
This project will address this gap in the field by aiming to develop non-toxic, naturally derived gels to optimize islet freezing and storage. The gel-based products will be evaluated for large-scale commercial production. The success of this project will provide new intellectual property that will be of interest to researchers and companies in regenerative medicine in Canada and across the globe.
The project
Using blood vessels to create a better encapsulation device for islet replacement therapies
Impact Award
The team
Dr. Corinne Hoesli (McGill), Dr. André Bégin-Drolet (Laval), Dr. Richard Leask (McGill), Dr. Andras Nagy (Sinai Health, Toronto), Dr. Steven Paraskevas (McGill)
Manufactured islets offer a potentially unlimited source of islets for transplantation. Since manufactured islets carry unique risks compared to donor-derived islets, containment within a device could allow retrieval if off-target growth ever occurs. However, encapsulation devices that have been tested in clinical trials so far and have shown minimal success, mainly because blood supply to the cells is limited by the device barrier. In this project, the team proposes to develop a device where the manufactured islets are placed around pre-established vessels that can improve islet cell survival and speed of insulin responses via improved blood supply. In this project, they will optimize their device design and conduct advanced preclinical studies.
This project could lead to better survival and function of manufactured islets, so they can keep producing insulin. The project may also pave the way for other engineered human-scale encapsulation devices, also sometimes called bioartificial organs.
The project
Using manufactured cells to create a human T1D immune system model in a petri dish
Impact Award
The team
Dr. Megan Levings, Dr. Bruce Verchere, Dr. Francis Lynn & Dr. Peter Zandstra (UBC)
There are many new treatments on the horizon for T1D, including those that block autoimmunity or replace insulin-producing cells. However, a major barrier to these therapies is the lack of an easy-to-use model in which their effects on human cells can be tested before advancing to human trials. The standard preclinical model is to test therapies in small animal models of T1D, but this has significant limitations since it is nearly impossible to replicate the human immune system. In fact, diabetes has been “cured” hundreds of times in a mouse model, which has not translated to humans.
To overcome this barrier, Dr. Levings and her team will establish a new a model that recreates human T1D autoimmunity in the lab. The model will use manufactured cells to create the three types of cells that are involved in the disease: insulin-producing cells and two different types of immune cells. Using the model, cells can then be combined in different ways to recreate what usually happens during autoimmunity.
A model of human T1D that can be generated in the lab will help test potential treatments and prompt new questions about why T1D develops, and how to prevent it. Thus, this research has the potential to support the further development of innovative therapies that may offer new approaches to prevent or treat people with T1D.
Putting it all together
Curing T1D is the north star of Breakthrough T1D. These partnerships will help us work together toward our shared goal of a world without T1D—through innovation, forward-thinking cell therapy research, and the best and brightest scientists.
Key Takeaways
- Breakthrough T1D helped organize a meeting in European Parliament to bring the unmet needs of the type 1 diabetes (T1D) community to the forefront and discuss how we can accelerate cures, especially cell therapies, in the EU.
- This meeting was attended by several Breakthrough T1D leadership and staff.
- Building long-term partnerships with the European Union (EU) Institutions will allow us to work together toward global T1D cures.
A meeting of the minds

This past week, Breakthrough T1D helped organize an event hosted by Member of European Parliament Tomislav Sokol, Ph.D., titled “Accelerating Breakthroughs to Address Unmet Needs in Type 1 Diabetes.” This meeting, a significant coming-together of Breakthrough T1D and European policymakers, focused on the role of the EU in addressing the needs of the T1D community and accelerating T1D breakthroughs.
The purpose of this meeting was to raise awareness of T1D and the urgent need for the accelerated development and approval of breakthrough therapies in the EU. Conversations between Breakthrough T1D and European policymakers homed in on barriers and opportunities to advancing cures—including cell therapies and disease-modifying therapies—in the EU to get them into the hands of people with T1D, faster. This was an important step in establishing an open dialogue between Breakthrough T1D and the EU Institutions about working together to address T1D globally.
“This event in the EU Parliament allows us to engage with important decision-makers to ensure that the research and policy environments are oriented in a way to accelerate development of T1D breakthrough therapies in the EU as we also do in other countries,” explained Campbell Hutton, Senior Vice President of Global Advocacy at Breakthrough T1D.
Attendees
Several Breakthrough T1D leadership and staff members attended the meeting, including Thomas Danne, M.D., Ph.D., Chief Medical Officer, Global; Sanjoy Dutta, Ph.D., Chief Scientific Officer; Lynn Starr, Chief Global Advocacy Officer, Carmen Hurtado del Pozo, Director, European Research; and Campbell Hutton, Senior Vice President of Global Advocacy.
Several Members of the European Parliament (MEP) in addition to host Tomislav Sokol, Ph.D., were in attendance. Other attendees included people with a lived experience of T1D, health staff from EU Member States, researchers in the EU, and other European diabetes organizations.
Dr. Dutta delivered a talk on the role of breakthrough therapies in transforming T1D. Dr. Danne moderated a panel to provide insight about unlocking the potential of cell therapies breakthroughs in the EU with T1D cell therapy researchers: Professor Lorenzo Piemonti, M.D., Director of the Diabetes Research Institute at Vita-Salute San Raffaele University and Associate Professor Francoise Carlotti, Ph.D., Head of the Islet Research Lab at Leiden University Medical Center. Finally, Lynn Starr closed with remarks about our shared global responsibility to work toward breakthrough T1D therapies.

When and where it took place
The event took place on June 5, 2025, in Brussels, Belgium, at the European Parliament.
Driving toward T1D cures in the EU and beyond
T1D is on the rise around the world, including in Europe. Recent publications by Breakthrough T1D staff and leadership brought attention to the rise in incidence and global T1D burden. We need to act now in conjunction with governments around the world—like the EU—to address the unmet needs of everyone around the world affected by T1D.

T1D cures, including cell therapies, are advancing through the clinical pipeline. Bringing awareness of T1D to the forefront—and educating key people on the progress we’re making toward cures—will be incredibly important for driving T1D research forward in the EU. This represents a critical opportunity for the EU to accelerate cell therapies faster than ever. As a global organization, Breakthrough T1D is collaborating with the EU government to help make this possible.
As the largest global funder of T1D research, Breakthrough T1D has provided funding to researchers across the world in addition to the EU. Right now, Breakthrough T1D is supporting €56 million in European initiatives, including 31 clinical trials—representing 19% of our funded research (including the U.K.). Breakthrough T1D has expert teams in research, medical, regulatory, and advocacy in Europe, meaning we are uniquely positioned to provide guidance on how the EU can strengthen its T1D efforts and collaborate on a global scale to drive T1D breakthroughs, especially in cell therapies.

Type 1 diabetes is a critical disease in Europe, and I was pleased to host an event for the T1D community and my colleagues in the European Parliament to learn about the unmet needs in T1D and how we can work together to accelerate breakthrough therapies in Europe to address those needs.”
What Breakthrough T1D leadership is saying

“This meeting is critically important to bringing the unmet needs of the T1D community into the spotlight in the EU. Global advocacy for curative T1D research is essential to achieving Breakthrough T1D’s mission, and continued collaboration with EU policymakers will get us there faster.”
Lynn Starr
Chief Global Advocacy Officer
“International efforts to accelerate global adoption of T1D cures will become increasingly important as newer, emerging cell therapies become available to people with T1D. Conversations like these with EU policymakers is bringing this urgent need to the forefront.”
Thomas Danne, M.D., Ph.D.
Chief Medical Officer, Global


“Cell therapies are accelerating through the clinical pipeline faster than ever. We need to act now on a global scale to ensure that people with T1D around the world can access these transformative therapies. This meeting is a significant step toward that goal.”
Sanjoy Dutta, Ph.D.
Chief Scientific Officer
Accelerating global action is paramount to our mission

Global problems require global solutions. This meeting served as a critical launching point for a continued partnership with the EU to fill gaps and address unmet needs for the T1D community. Building relationships and fostering long-term partnerships is critically important for reaching our common goal of bringing cures to people with T1D as soon as possible.
These important conversations between Breakthrough T1D and the EU government align with our Project ACT (Accelerate Cell Therapies) initiative to accelerate the development of cell therapies that do not require immunosuppression—for everyone with T1D in every country. In addition, through international Centers of Reference, Breakthrough T1D’s Medical Affairs team is developing expert clinical care centers that will be trained and ready to provide cell therapies to people with T1D once they become available. We are at the forefront of global action to prepare the world for curative cell therapies.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
We are driving toward a future in which everyone with T1D—no matter where they are—has access to therapies, treatments, and care, bringing us closer to achieving our mission of a world without T1D. The more people we have working toward our mission, the faster we will get there.
Outside the Cammett family’s Michigan home sat a new toy truck. John, around six years old, admired its sturdy metal frame, fresh rubber wheels, and bright yellow decals. But it would not stay that way for long.
John and his two brothers were athletic, boisterous, and yes, at times, destructive. After the truck was thoroughly battered, John’s mother, Barbara, began her work on the piece. She was creative and artistic and knew just what to do. With paint brushes in hand, she covered the damaged truck in a collage of color and something new emerged. It was no longer a wreck, it was a work of art.
John Cammett, now 62, says he’ll always remember that day and the special lesson that came from it.
“Even with all that destruction, she could make something look beautiful,” he said.
Like mother, like son
John’s mom was diagnosed with type 1 diabetes (T1D) in her 30s and passed away in 2021 at age 89. She inspired John, who also lives with T1D, to become a champion for others with the condition. John has since become a longtime volunteer, leader, and advocate for Breakthrough T1D—providing transformational support of our mission.
In recognition of his deep commitment, the Breakthrough T1D Center of Excellence in New England was recently named for his mother. The center will now be known as the Breakthrough T1D Barbara Dewey Cammett Center of Excellence in New England. John helped establish the center and provided foundational support for Project ACT (Accelerate Cell Therapies), Breakthrough T1D’s initiative to make cell therapies as cures for T1D a reality.

My mom was the strongest person I’ve ever known—a real warrior. Even back then, with limited technology, she never let T1D set her back.”
Honoring her positive spirit
Since its inception, the Breakthrough T1D Barbara Dewey Cammett Center of Excellence in New England has made significant progress advancing islet cell-based T1D research, enhancing the understanding of the immune response following islet transplantation to prevent rejection, and creating genetically modified islet cells that could withstand the immune attack after transplantation into people living with T1D.

The breakthrough research happening at the center—one of five Breakthrough T1D Centers of Excellence around the world powering advances to deliver cures and life-improving breakthroughs for T1D—is particularly inspiring to John. He takes pride in knowing that the center he helped establish will not only advance this important work but also honor his mother’s positive spirit and enduring legacy.
“You can’t be a researcher without optimism—every breakthrough stands on the back of countless failures. My mom lived the same way. She was the most optimistic person I’ve ever known, never said a bad word about anyone, and kept going no matter how hard things got. Just like the researchers pushing forward every day, she stayed focused, kind, and hopeful through it all.”
A beacon of strength
John remembers his mom as a woman whose generous heart, zest for life, and unwavering optimism inspired everyone she met.
She managed T1D for nearly 60 years with grace, determination, and a smile. Known for her vibrant personality, Barbara embraced life fully. You could often find her cheering on her Wisconsin Badgers, painting beautiful art, volunteering in her community, and effortlessly outdriving her husband on the golf course.

Her kindness, resilience, and passion for connecting with others made her a beloved friend, devoted wife, and beacon of strength to those navigating life with T1D.
“I wish I could have done this while she was still with me,” John said. “But I know she’d look back and smile. She wanted to help everyone she could. That spirit lives on.”
By: Adam Baker
Breakthrough T1D’s newest mission pillar, Medical Affairs, is bridging the gap between access to and adoption of T1D therapies. The establishment of this program is essential to Project ACT (Accelerate Cell Therapies): Breakthrough T1D’s initiative to accelerate the development of manufactured islet cell replacement therapies that do not require immunosuppression. The goal is to make sure that people with type 1 diabetes (T1D) can get these therapies as soon as they hit the market.
The field is moving quickly: people are becoming insulin-independent in cell therapy clinical trials. We are advancing towards the submission of the first-generation manufactured islet cell therapy that requires immunosuppression, Vertex’s zimislecel (VX-880). We are at a critical moment and need to act now to ensure that healthcare providers (HCPs) are ready to bring manufactured islet cell therapies into clinical settings.
Enter Medical Affairs
This is where Medical Affairs comes in. The team, led by Thomas Danne, M.D., Chief Medical Officer International, is working with the medical community to anticipate obstacles to getting manufactured islet therapies into clinics and find ways to overcome them now.
To accomplish this, Breakthrough T1D recently hosted two cell therapy workshops, convening multidisciplinary, international experts in islet cell transplantation to discuss a clinical roadmap for manufactured islet cell therapies—and how to ensure that clinical teams are in place and prepared to provide these therapies to people with T1D who qualify. By preparing now, we can get this first-generation therapy into the hands of people with T1D as soon as possible after regulatory approval.
The cell therapy workshops were hosted by Breakthrough T1D’s Thomas Danne, M.D., Chief Medical Officer International, and Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, Vice President of Medical Affairs.
Read on to learn more about the cell therapy workshops, the attendees, and what each accomplished.
Workshop #1: Who may benefit most from manufactured cell replacement therapies
Key Takeaways
- T1D leaders are working on a five-year roadmap for the clinical adoption of manufactured islet cell therapies.
- It will include key criteria and evidence to help clinicians determine who is the best fit for manufactured islet cell therapies.
- The guidelines are prioritizing shared decisions between people with T1D and their care team to optimize outcomes and maximize long-term health.
Attendees
The first of the cell therapy workshops, held in late April, convened transplant surgeons, T1D clinicians and researchers, a member of Vertex’s leadership team, a member of Breakthrough T1D’s Participant Advisory Council to represent people with a lived experience of T1D, and Breakthrough T1D Vice President of Research Esther Latres, Ph.D.
The purpose of this workshop was to start developing a five-year roadmap to help guide the T1D care community to support the adoption of manufactured islet cell therapies in clinical care. The evidence-based recommendations will be vetted by a larger group of clinical experts, diabetes organizations, and people with lived experience with T1D to ensure there is broad agreement. The consensus document will ultimately be published to expand its reach.
This process will summarize the essential evidence that will help HCPs decide who may benefit the most from manufactured islet cell replacement therapies. These decisions will take into account the perspective of people with T1D and differences in age, hypoglycemia unawareness, or kidney health, to name a few. This can help HCPs better understand the benefits versus risks for manufactured cell therapies on a person-by-person basis—making sure that each clinical decision is made jointly to prioritize long-term health.
Workshop #2: Pilot workshop to develop international Centers of Reference for T1D cell therapy
Key Takeaways
- Centers of Reference are expert T1D care centers that are preparing to bring manufactured islet cell therapies to the T1D community.
- This workshop was the first step to understanding what kind of education, training, and resources are needed for Centers of Reference to be effective.
- Attendees discussed how to make sure people T1D who receive manufactured cell therapies at Centers of Reference will have the best clinical experience possible.
Attendees
The second cell therapy workshop, held in early May, convened clinicians from various global medical institutions, including University of Minnesota Medical Center, University of Wisconsin Health Transplant Center, the Penn Rodebaugh Diabetes Center, University of Chicago Medicine, IRCCS Ospedale San Raffaele (Italy), Institute of Transplantation, Newcastle upon Tyne (United Kingdom), and University of Alberta (Edmonton, Alberta, Canada).
Additional attendees from Breakthrough T1D included CEO Aaron Kowalski, Ph.D., Vice President of Research Esther Latres, Ph.D., the Medical Affairs team, a volunteer, and a member of the Participant Advisory Council, who is a person living with T1D.
The objective of this workshop was to take the first step toward creating Centers of Reference for T1D manufactured cell therapies. “The initial purpose is to accelerate readiness of healthcare professionals to deliver manufactured islet cell therapies once they become available,” explained Dr. Danne. “…making such a treatment a success needs teamwork. Accredited Centers of Reference will not only deliver advanced T1D treatments but also serve as a training hub for professionals aspiring to become experts.”
This workshop focused on better understanding what potential Centers of Reference need to be successful. The attendees covered a range of topics: what an ideal T1D care team might look like, the education and training required for experts in T1D manufactured islet cell therapy, and career development for early-stage T1D professionals.
The goal is to prepare expert clinicians—who are already doing islet cell transplants—to bring manufactured islet cell therapies into clinical practice at their institutions and others, once they have regulatory approval. These centers will serve as a benchmark for best practices in T1D manufactured cell therapy, establishing a network of expert teams to make sure that everyone who can benefit from manufactured cell replacement therapy is given the opportunity to consider it.
What the experts are saying

“We need to build consensus and teamwork. When manufactured cell therapies exist, it’s going to take significant coordination between endocrinologists, transplant surgeons, and people with T1D to ensure as many people as possible are benefiting from these therapies.”
Jon Odorico, M.D.
Professor of Surgery and Director of Pancreas and Islet Transplantation at University of Wisconsin Health Transplant Center
“We’re trying to solve access and awareness. There’s a definite gap between primary care endocrine diabetes specialists and transplant specialists…there’s so much more that we have to fill in for [people with T1D].”
Helen Nelson, BSN, RN, CCTC, CPTC
Program Manager, Organ Allocation/Clinical Triage and Pancreas Transplant Program at University of Wisconsin Health Transplant Center


“It’s going to be a significant problem if we have a cure but no one has access to it because no one can deliver it. We must work together—transplant surgeons, endocrinologists, researchers, everyone. It’s like building Cape Canaveral in anticipation of sending rockets into space.”
Peter Senior, MBBS, Ph.D.
Islet Transplant Endocrinologist, Professor in the Department of Medicine, and Director of the Alberta Diabetes Research Institute at the University of Alberta, Canada
“There is amazing excitement around creating cell therapies. People are excited about it. We must ensure that organizations like Breakthrough T1D bridge the gap between research and the T1D population so that there is no difference between an individual’s reality and what therapies are available.”
James Shaw, M.D., Ph.D.
Transplant Endocrinologist and Professor of Regenerative Medicine for Diabetes at the Institute of Transplantation, Newcastle upon Tyne, United Kingdom

This is just the beginning
Manufactured islet cell therapies are coming. We need teamwork to get these therapies into clinics so people with T1D don’t have to wait years to get them. This is why Breakthrough T1D is acting now: when the first manufactured islet cell therapy hits the market, multidisciplinary care teams around the world will be ready. These workshops—the first of many—will help accelerate the safe and effective integration of manufactured islet cell therapies into clinics.
“This way we will ensure that the medical community is ready to deliver manufactured cell therapies once they become more widely available.,” Dr. Danne said. Thanks to the hard work of the Medical Affairs team at Breakthrough T1D, this goal is in sight.

Project ACT series
This article is part of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) is shaping the future of cell therapies for type 1 diabetes (T1D). The next article in the series will focus on Project ACT’s advocacy efforts to ensure there is a regulatory pathway to approval for these therapies and that they will be covered by payers.
Read last month’s article about challenges and solutions of T1D cell therapies.
Despite significant advances in treatments for T1D, our community still has significant unmet needs. Breakthrough T1D believes that novel cell therapies will transform T1D management, and Project ACT is how we’re going to make them a reality.
First-generation cell therapies, including FDA-approved, donor-derived Lantidra® and Vertex’s manufactured islet therapy in phase 1/2/3 clinical trials, VX-880 (Zimislecel), are incredibly promising. They have some limitations, including:
- There are not enough donor-derived islets to meet the needs of everyone with T1D.
- These therapies are only available to people with severe hypoglycemia unawareness and hypoglycemic events.
- The number of people who can receive these therapies is further limited in that they must be able to tolerate chronic, broad immunosuppression.
Current research efforts at the preclinical, clinical, and manufacturing levels are working to address these challenges. The ultimate goal is a future in which manufactured islet therapies exist in large supply, survive and produce insulin in the body after implantation, and remain protected from the immune system. Learn more about Breakthrough T1D’s Cell Therapies Program and take a closer look at what researchers are doing to turn these ideas into a reality.
Clinical trials to keep an eye on
Up-and-coming cell therapies for T1D are in the clinical pipeline and working their way towards the market, including many that Breakthrough T1D has contributed to. There are some highly anticipated (and currently enrolling!) trials that we have our eyes on right now, and we hope to see data soon. Read about each study in detail below or scroll down to see a summary table.
Late last year, Vertex announced the expansion of their manufactured islet therapy, VX-880 (Zimislecel), to a phase 1/2/3 clinical trial, the final step before seeking FDA approval. This decision stemmed from groundbreaking data in the initial phases of the trial in which 11 of 12 participants reduced or eliminated the need for external insulin therapy. Currently, zimislecel is limited to people with severe hypoglycemia and requires chronic immunosuppression.
The results of the VX-880 trial are highly anticipated since this is the first time a scalable treatment for T1D has entered a final clinical testing stage, and regulatory submission is expected in 2026. Vertex is working closely with regulators to expand its manufacturing capabilities and ensure they are prepared for the therapy to hit the market.
Zimislecel would not have been possible without years of support from Breakthrough T1D and The T1D Fund: A Breakthrough T1D Venture. This includes research grants, an investment by the Fund in Semma Therapeutics (which was later acquired by Vertex), and much more.
It doesn’t stop there: Vertex is expanding their pipeline and investigating different ways to keep manufactured islets safe without standard anti-rejection immunosuppressants, including alternative immunosuppressive regimens, gene-edited immune-protected cells, and novel encapsulation devices.
Although Vertex’s T1D portfolio is progressing, the clinical development of VX-264, an encapsulated islet therapy that does not require immunosuppression, has been discontinued. While it was safe and well-tolerated in clinical trials, it did not meet efficacy and safety endpoints as measured by C-peptide, a biomarker for insulin production.
UP421 consists of islets derived from deceased donors that have been engineered to be hypoimmune, meaning they can avoid detection by the immune system without the need for immunosuppressants. Incredibly, the first person who received a partial dose of UP421, implanted in to forearm muscles, in a phase 1 clinical trial is making their own insulin, as demonstrated by increased C-peptide, without any side effects.
This is the first proof-of-concept evidence showing that this cell engineering approach can enable implanted islets to avoid immune destruction. The next step is applying this method to manufactured islets.
Breakthrough T1D is supporting research exploring similar cell engineering approaches to allow implanted islets to evade the immune system. The T1D Fund has also invested in Sana due to their distinctive hypoimmune manufactured islet replacement program, and Breakthrough T1D continues to work closely with them.
Tegoprubart is an immunotherapy that interferes with immune cell communication and dampens the immune response. This therapy is being tested in a Breakthrough T1D-funded phase 1/2 clinical trial as a novel anti-rejection immunosuppressant for people with severe hypoglycemia who have received deceased donor islets. Building on ongoing kidney transplant studies, this study will determine if tegoprubart can protect transplanted islets from rejection with fewer side effects compared to standard immunosuppressants, which is harsh on people and the implanted cells.
So far, of the first three participants, two have achieved insulin therapy independence. According to the study, tegoprubart is safer for both people and transplanted cells in comparison to standard immunosuppression, with milder side effects and greater islet survival. This study holds promise for preventing rejection of manufactured islets as well.
The T1D Fund has made several investments in Eledon to support this effort as it sees the potential to address the key unmet need of safe and effective immunosuppression for people who receive islet replacement therapies.
Cell Pouch™ is an implantable bio-hybrid organ that provides a specialized environment for transplanted islet cells by allowing them to access oxygen and nutrients provided by blood vessels, called vascularization.
The first cohort of a phase 1/2 clinical trial enrolled participants with severe hypoglycemia who received deceased donor islets within Cell Pouch in addition to standard immunosuppressants. Of the six enrollees, five remain insulin therapy independent from one year to more than five years. Cohort B is currently evaluating a higher-capacity Cell Pouch that can accommodate 50% more islet volume, and the trial will soon advance to Cohort C to further test safety and efficacy of the system.
Most excitingly, Sernova recently announced that following the conclusion of the ongoing clinical trial, they will initiate a new trial to test Cell Pouch implanted with manufactured islets—paving the way towards a scalable solution to T1D.
Breakthrough T1D has supported the development of Cell Pouch™ and continues to work with Sernova.
SR-02 is a manufactured islet cell therapy implanted onto the omentum, a fatty, protective layer around organs. This therapy is in a phase 1/2 clinical trial for people with severe hypoglycemia and requires immunosuppression. The trial is evaluating safety and insulin production as measured by C-peptide.
Seraxis is also working on another manufactured islet therapy (SR-03) that has been gene-edited so that anti-rejection immunosuppressants are not needed. They are hoping to initiate a new clinical trial for SR-03 in 2026.
The T1D Fund has invested in Seraxis to aid in the development of these therapies based on their distinctive and promising islet replacement approach.
CTX211 is another manufactured islet therapy that has been gene-edited to evade the immune system so that recipients do not have to take immunosuppressants. In an ongoing phase 1/2 clinical trial, these cells are implanted within a specialized device to help keep the cells alive in the body, and investigators are evaluating safety as well as insulin production measured by C-peptide. Results are expected this year.
Breakthrough T1D was a long-time supporter of ViaCyte, which initially developed the manufactured islets and partnered with CRISPR Therapeutics to genetically modify them. ViaCyte was acquired by Vertex in July 2022, and now CRISPR Therapeutics is the sole owner of this therapeutic platform.
- OPF-310 (Otsuka Pharmaceutical): encapsulated islets derived from pigs in phase 1/2 clinical trials
- ENC-201-CED (Encellin): donor-derived islets in a proprietary encapsulation device implanted subcutaneously in a phase 1 clinical trial
- Gastrin: a phase 1/2 trial testing if gastrin, a naturally occurring hormone involved in pancreatic development, is safe and helps retain or grow islets following donor-derived islet transplantation
Recruiting cell therapies clinical trials
| Therapy | Primary Outcome(s) | ID | Location | Phase |
| VX-880 (Zimislecel) | Safety, insulin independence, and absence of severe hypoglycemic events | NCT04786262 | US/Canada/UK/EU | 1/2/3 |
| VX-880 (Zimislecel) | Insulin independence in people with T1D and a kidney transplant | NCT06832410 | Canada | 2 |
| UP421 | Safety | NCT06239636 | Sweden | 1 |
| Tegoprubart + donor islets | Insulin independence | NCT06305286 | US (Chicago, IL) | 1/2 |
| Cell Pouch™ + donor islets | Safety | NCT03513939 | US (Chicago, IL) | 1/2 |
| SR-02 | Safety, C-peptide | NCT06651515 | US (Pennsylvania) | 1/2 |
| CTX211 | Safety, C-peptide | NCT05565248 | Canada | 1/2 |
| OPF-310 | HbA1c<7% and absence of severe hypoglycemic events | NCT06575426 | US (Chicago, IL) | 1/2 |
| ENC-201-CED ENCRT + donor islets | Safety | NCT06408311 | Canada | 1 |
| Gastrin + donor islets | Insulin independence, absence of severe hypoglycemic events, and HbA1c<6.5% | NCT03746769 | US (Duarte, California) | 1/2 |
Where do we go from here?
The emergence of cell therapies for T1D in clinical trials is incredibly exciting for the T1D community. Advancements using deceased donor-derived islets are paving regulatory pathways for manufactured islet therapies—which are curing people with T1D in clinical trials—to make their way towards the market.
“One of the greatest effects that manufactured islet cell therapies will have for the T1D community is being able to think about their type 1 diabetes less,” explained Nicholas Mamrak, Ph.D., a scientist at Breakthrough T1D. This may soon be a reality for a subset of people with T1D as we drive towards the approval of a first-generation manufactured islet replacement therapy, reducing the day-to-day burden of managing blood glucose and insulin dosing.
The ultimate goal is to make sure manufactured islet therapies are available to everyone with T1D—ideally without the need for immunosuppression. This is the objective of Breakthrough T1D’s Project ACT.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Before cell therapies can become available for everyone, they must first receive regulatory approval. A key part of Project ACT is streamlining and advancing regulatory pathways for the treatment of T1D. To help achieve this, Advocacy and Regulatory leadership teams at Breakthrough T1D are working on a new publication capturing how day-to-day lives have changed for people with T1D who have received cell therapies. By using personal experiences to share what benefits of cell therapies matter most to patients, we can help regulators understand the powerful impact that islet replacement therapies can have.
None of this would have been possible without the T1D community’s continued generosity and support, as we all work together to move the needle forward.
Have an impact by participating in clinical trials
Without clinical trials, we would never know if new therapies developed by scientists in the lab could make a difference in people’s lives. This is where the T1D community comes in—by volunteering to participate in clinical trials, you become uniquely positioned to help drive biomedical research forward. Moreover, by participating, you help not only yourself, but everyone with T1D. Find a clinical trial near you and see if you are eligible to participate. Connect with a Clinical Trial Education Volunteer in your area, who can answer any questions you may have.

Project ACT series
This article is part of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) will shape the future of cell therapies for type 1 diabetes (T1D). The next article in the series will discuss the progress of cell therapies in clinical trials.
We’ve made major progress in the development of cell replacement therapies for type 1 diabetes (T1D) over the past couple of decades. We know that manufactured islets, such as Vertex’s VX-880 (now Zimislecel), can restore insulin therapy independence and glucose control when implanted into people with T1D.
However, there is more work to do.
We need to make sure the cells survive and function, ideally without immunosuppression, and ensure that these therapies are accessible to everyone with T1D. Read on to learn more about where there’s room for improvement and what we’re doing about it.
Optimizing manufactured islets
Priority #1: Cell source
Current external cell sources that are not manufactured in the lab, such as those from deceased donor pancreases or a person’s own cells, are in extremely limited supply. The only FDA-approved cell therapy for T1D, Lantidra®, requires donor cells, and it can take up to three pancreases to get enough islets for one transplant. This is unsustainable and limits the number of people who can get this therapy.
We can consistently generate an unlimited source of islet cells in the lab. This way, we can make enough insulin-producing cells for everyone with T1D and have a single cell source to test and compare multiple strategies to protect them.
Breakthrough T1D is funding an initiative at the Advanced Regenerative Manufacturing Institute (ARMI) to scale up Dr. Jeffrey Millman’s protocol to generate unlimited manufactured islets in a reliable, automated, and reproducible way. Breakthrough T1D is also building a partnership with the Cedars-Sinai Biomanufacturing Center to accomplish this goal.
Priority #2: Cell survival
Implanted islets that move through the bloodstream can cause an inflammatory reaction, resulting in cell death. Alternatively, those that are implanted in devices that are cut off from the blood and immune system are unable to get nutrients and oxygen, again leading to cell death.
It doesn’t have to be the pancreas, but islets need access to nutrients and oxygen so that they can survive for long periods of time and produce insulin.
Breakthrough T1D is funding research to develop scaffolds, which are specialized biomaterials that islet cells can stick to and get nutrients and oxygen to help them survive. Similarly, islets implanted in encapsulation devices, such as Sernova’s Cell Pouch™, can access nutrients and oxygen while having the added benefit of being protected from the immune system. There are also various Breakthrough T1D-funded clinical studies that are investigating different places in the body for manufactured islets, including the omentum and abdominal wall.

Omentum
The omentum is a fatty tissue layer that surrounds and protects the organs in your abdomen. Researchers are testing it as a new implantation site for manufactured islets.
Priority #3: Cell protection
Like organ transplants, manufactured islet therapies from an external source are recognized by the immune system as “non-self,” leading to immune rejection. Currently available cell therapies require broad immunosuppressants that may come with unwelcome side effects, including increased risk of infection and malignancy and toxicity to kidneys, nerves, and islet cells themselves.
By swapping standard immunosuppressives with options that have less complications, more people with T1D will be able to access these therapies.
Research and clinical studies funded by Breakthrough T1D approach immune protection from many different angles. This includes genetically engineered islets that can evade immune detection, encapsulation devices, and immunomodulatory therapeutics that can dampen the immune response.
The future of manufactured islets and T1D cures
There’s a lot of promising solutions in the pipeline. What’s next? Unlocking access.
“We recognize that approval of cures is not a life-changing breakthrough if people do not have access to the therapy itself,” said Aaron Turner-Phifer, Senior Director of Health Policy at Breakthrough T1D. “Building on the experience gained from past breakthroughs, we are working now, across our Mission teams, to identify and remove any potential barriers to people accessing cell therapies.”

“While our work is just beginning, we’ve already conducted market analysis to identify clinical and payer barriers to give us clarity on where to start. We are directly engaging policymakers and health plans to educate them on T1D cell therapies. We’ve also launched data projects to begin to generate the types of data required to positively inform future policy and coverage decisions.”
Aaron Turner-Phifer
In the coming years, it’s likely that first-generation manufactured islet therapies will be available to people with T1D with severe hypoglycemia unawareness and will require broad immunosuppression.
Later, advancements in cell survival and immune protection—combined with the advent of more tolerable immune suppression approaches—will open the doors for more people with T1D to access these life-changing therapies.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Without continuous support from the T1D community and its supporters, we would never have gotten this far. Breakthrough T1D looks forward to a future where manufactured islet therapies are a reality for everyone with T1D, and we will not stop until we get there.