Marilyn and Dr. Gerald Fishbone knew first-hand how incredible research changes the lives of people with type 1 diabetes (T1D). For more than 50 years, they were pillars in the T1D community, dedicating their lives to helping advance innovative research.
Marilyn and Gerald became deeply involved with Breakthrough T1D (then JDF and later, JDRF) in 1969 after their then-18-month-old son, Scott, was diagnosed with T1D.
“I had never heard of diabetes. No one in our family, before Scott, had diabetes,” Marilyn said in a 2021 interview with Breakthrough T1D.
Gerald passed away January 16, 2023. Marilyn, his beloved wife, passed away March 31, 2026.
After Scott’s diagnosis, Marilyn went on to found the New Haven, CT, Chapter, and remained an engaged volunteer and donor. Gerald served in many roles at Breakthrough T1D, including Chair of the Breakthrough T1D International Board of Directors (IBOD), Medical Advisor, Research Committee member, and donor.
The Fishbones created a legacy of service which is being carried forward by their children and grandchildren.
Their daughter, Lisa Fishbone Wallack, became Chair of IBOD in 2024 and had previously served as Vice Chair. She had also served as President of the Greater New England Chapter.
Their son, Scott, is a dedicated Breakthrough T1D volunteer leader who serves on the New Jersey Chapter Board and on the Chapter’s Executive Committee as Advocacy Co-chair.
“We believe in being involved and giving back,” said Marilyn. “Our kids joke that they never ate on a clean dining table because it was always covered in Breakthrough T1D fundraising envelopes. Most of the time, it was. We worked extremely hard and committed ourselves as a family to giving our time and energy to Breakthrough T1D and to inspiring others to join us.”
In 2001, the Fishbones’ then 5-year-old grandson, Harris Wallack, was diagnosed with T1D.
“His diagnosis, while still devastating, was so different. The technology available to make it safer and easier to manage T1D was drastically improved from when Scott was diagnosed,” explained Marilyn. “That is all thanks to the strategic leadership of Breakthrough T1D.”
The Fishbones are greatly missed. Their dedication and generosity to Breakthrough T1D will be felt by all members of the T1D community for decades to come.
Read more about Dr. Gerald Fishbone’s life.
Read more about Marilyn Klugman Fishbone’s life.
Breakthrough T1D is back from Barcelona! The final two days of ATTD were jam-packed with more incredible updates across all areas of our mission. Breakthrough T1D staff chaired and participated in many sessions—including Breakthrough T1D Senior Vice President of Research, Esther Latres, Ph.D.; Vice President of Medical Affairs Anastasia Albanese O’Neill, Ph.D., APRN, CDCES; Senior Director of Research, Jonathan Rosen, Ph.D.; and more.
Jump down to:
Screening
Screening is important! From preventing life-threatening diabetic ketoacidosis (DKA) at diagnosis, to giving people the opportunity to intervene with Tzield or disease-modifying therapies in clinical trials, to having more time to plan and prepare for type 1 diabetes (T1D) onset—screening and early detection are essential to our mission. And it must happen at a large scale if we’re going to achieve cures—which is why it was so heavily represented at ATTD 2026.
Vice President of Medical Affairs Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, chaired yet another panel where screening was again discussed. To repeat from our earlier recap, T1D Screening and Early Detection is a key mission priority for Breakthrough T1D. Multiple presentations illustrated how important it is, and why we need to screen more people to achieve our vision of a world without T1D.
One theme, and why it came up over and over again, is that we need more public education about screening, the benefits of early detection, and how mission-critical it is. It’s not an accident that there were many sessions that all hammered this home.
What can you do? Tell your community about screening!
Disease-modifying therapies
What are disease-modifying therapies?
Disease-modifying therapies are drugs that change the course of T1D. That means slowing or halting the progression of the disease, preventing it from ever occurring, and reversing it entirely.
There were several exciting presentations from some key Breakthrough T1D collaborators. This includes Jay Skyler M.D., MACP, FRCP, who outlined the challenges and obstacles that must be overcome at every stage in T1D in order to fulfill his personal mission to eradicate T1D before he retires. This includes disease-modifying therapies at different stages of T1D.
Another noteworthy lecture came from Chantal Mathieu, M.D., Ph.D. (who happens to be on the advisory committee for the Breakthrough T1D Clinical and Research Congress!). She focused on C-peptide, which is natural biomarker of insulin production.
Breakthrough T1D believes C-peptide should be a validated clinical trial endpoint. Dr. Mathieu agrees! Currently, metrics like HbA1c are used to evaluate the efficacy of therapies for early-stage T1D. In Dr. Mathieu’s opinion, HbA1c as a clinical trial endpoint will mean, simply, that it will be a very long time before another DMT joins Tzield as FDA an approved therapy for this disease.
And, most importantly, there are real benefits to maintaining beta cell mass and, therefore, having increased C-peptide function! We know this! And it’s up to Breakthrough T1D, academia, and others to persuade regulators that it’s time for it to be accepted as a clinical trial endpoint so there can be more options for people with T1D.
Cell therapies
Cell therapies, which place insulin-producing cells inside people with T1D, are a huge Breakthrough T1D priority, as exemplified by Project ACT.
Senior Vice President of Research, Esther Latres, Ph.D., chaired a session titled “Islet Cell Therapy: Cure for Type 1 Diabetes – How Far Are We?” This session highlighted Breakthrough T1D’s approach to cell therapies in pursuit of our ultimate goal: manufactured islets, produced at scale, transplanted without the need for chronic immunosuppression.

Cell therapies: Where are things now?
Islet transplants have been around for decades, but they require chronic immunosuppression. Additionally, deceased donor islets are in limited supply, meaning there are not enough islet cells for everyone with T1D who may want them.
Update on Eledon’s tegoprubart
Let’s start with the topic everyone is talking about: the use of Eledon’s tegoprubart with deceased donor islet cell transplants.
Breakthrough T1D is funding a trial at the University of Chicago evaluating the use of the targeted immunosuppressive therapy tegoprubart, instead of standard, broad immunosuppressants like tacrolimus, in islet cell transplants. The data so far has been very encouraging—and Principal Investigator, Piotr Witkowski, M.D., Ph.D., was at ATTD to share more.
What is Breakthrough T1D’s relationship with Eledon?
Breakthrough T1D has had a relationship with Eledon for several years, since Breakthrough T1D brought Eledon into the T1D space. Starting in 2023, Breakthrough T1D began funding a clinical trial that uses their drug, tegoprubart, with islet transplants in T1D, with initial additional support from The Cure Alliance and the Diabetes Research Institute. Additionally, the T1D Fund invested in Eledon in 2023.
All 12 recipients are off external insulin or on target to stop external insulin.
To date, 12 people with T1D have received deceased donor islet transplants and are receiving tegoprubart infusions every three weeks. All of the individuals are doing exceptionally.
Here’s some stats to back it up:
- All 12 subjects immediately experienced improved blood sugar levels and control.
- As of today, 10 of the 12 individuals are entirely off insulin therapy. The other two are on that trajectory.
- There have been zero unexpected adverse events, severe hypoglycemic episodes, thrombotic events, opportunistic infections, signs of rejection, or gastrointestinal symptoms.
- Crucially, there have been no signs of kidney and neurotoxicity, which is something that is observed in individuals who receive islet transplants and use tacrolimus.
The next step for this therapy is a study testing deceased donor islet cells in people with T1D and chronic kidney disease, funded by Breakthrough T1D.
The main takeaway? This remains a very promising alternative to current immunosuppression.
Sana’s gene-edited islets continue to make insulin
Per-Ola Carlsson, M.D., Ph.D., presented early results from a novel cell therapy approach designed to help transplanted islet cells evade immune attack while continuing to produce insulin.
The strategy uses gene editing to create hypoimmune islet cells—cells engineered to avoid detection by the immune system while maintaining their insulin-producing function. This approach is one of several next-generation cell therapy strategies being prioritized by Breakthrough T1D to help overcome one of the biggest barriers to cell replacement therapies: immune rejection.
Prior to this study, the concept had been demonstrated in non-human primates. Carlsson and his team have now taken the next step by testing the approach in a person living with T1D.
In this first-in-human study, deceased donor islets were genetically modified to become immune-evasive and then transplanted into the forearm. Because this was a phase 1 trial, the primary goal was to assess safety, while also monitoring islet function through C-peptide, a marker of the body’s own insulin production.
Importantly, the transplant included only about 5% of the number of cells typically needed to fully restore insulin production, reflecting the exploratory nature of the study.
The results so far are encouraging. After 60 weeks, there have been no severe or unexpected adverse events, meeting the trial’s primary safety endpoint. At 14 months after transplantation, the participant continued to produce detectable C-peptide, indicating that the transplanted cells remained alive and functional.
Researchers also observed that C-peptide levels temporarily declined after about one year, likely due to beta cell exhaustion, but subsequently recovered. This is an encouraging signal that the transplanted cells may be capable of regaining function.
While still very early, these findings provide important proof of concept that gene-edited, immune-evasive islet cells can survive and function in a person with T1D. If confirmed in larger studies, this approach could help move the field closer to cell therapies that work without long-term immune suppression—a major goal for the future of T1D cures.
Addressing challenges in cell therapies
Holger Russ, Ph.D. explored a key problem that must be overcome: manufacturing islets at a large scale is hard to do! He shared the processes that his team at the University of Florida uses and noted a secondary problem: we make all these cells, and a significant portion are lost post-transplantation. To solve that, he presented his work on different gene-editing strategies to keep these cells alive.
Katy Digovich, CEO of Minutia, discussed a different problem that needs to be solved: how do we monitor the transplanted islets? Minutia’s approach is to integrate gold (yes, gold) nanoparticles into the islet clusters, which allows them to safely monitor the cells and track graft survival and rejection.
All of this work is supported by Breakthrough T1D and/or the T1D Fund.
More to come soon for Vertex’s manufactured islets
Data from Vertex Pharmaceutical’s phase 1/2/3 clinical trial for zimislecel was presented by Trevor Reichman, M.D. This data is not new, but it reinforces the reasons for optimism around this manufactured islet cell therapy.
This therapy consists of fully differentiated, manufactured islet cells, infused into the portal vein using an approach similar to traditional donor islet transplantation. Participants are on immunosuppression, and the safety profile is consistent with the use of immunosuppressive medications.
Unfortunately for the T1D community, we’re not going to get new data for Vertex for some time because the data currently being collected will be used for their upcoming FDA submission. However, stay tuned for a Q&A with Vertex in the near future!
Improving Lives
Breakthrough T1D Director of Research, Jonathan Rosen, Ph.D., chaired the session “Bridging the Gap Between Academia and Industry in T1D.” This is an important priority area for us as we see ourselves as “gap fillers.” There is a pipeline from academia to industry to people with T1D, and Breakthrough T1D’s strategy is to identify these gaps and ensure that the pathway is as smooth—and complete—as possible.
This session featured several leaders from biotech illustrating how, ideally, everyone can work together to move products along. Some key points:

- Academia, startups, and big pharma companies all have strengths and weaknesses. All roles are important and it’s crucial to understand the limitations and capabilities of each player.
- Other key players: governments and non-profit organizations. Thanks to the Breakthrough T1D Advocacy team, there has been billions of funding to T1D research over the past few decades. All these products must undergo regulatory review. All play a part!
- What Breakthrough T1D can do is identify the shared objectives. Industry and Academia are not opposing forces, and it’s up to Breakthrough T1D to ensure everyone is bringing their strengths to the table and row in the same direction.
The use of adjunctive therapies, including GLP-1s, is a priority of the Breakthrough T1D’s Improving Lives Portfolio. The use of these drugs, which is often off-label and includes semaglutide and tirzepatide, is increasing in the T1D community.
The data backs this up. Satish Garg, M.D., presented research showing these medications may support weight loss, lower insulin requirements, and improved blood glucose, specifically including better time in range. We also heard again from Viral Shah, M.D., who gave a recap of the Breakthrough T1D-funded ADJUST-T1D study, which showed that the GLP-1 receptor agonist semaglutide (Ozempic) can improve weight and glycemic outcomes in people with T1D.
It’s true: GLP-1s are increasingly being used off-label in the T1D community, as demonstrated by the Breakthrough T1D-funded work of Francisco Pasquel, M.D., MPH. Using large-scale patient datasets from electronic health records, Dr. Pasquel found that rates of GLP-1 use are highest in people living with T1D and obesity in addition to those with cardiovascular disease (CVD) or kidney disease. While this class of drugs has potential to improve weight and blood sugar management, we need larger clinical trials to confirm safety, identify less-common side effects, and get a better idea of the non-glycemic benefits that GLP-1s may have for the T1D community.
To make some headway on the benefits of GLP-1s beyond glucose management, Janet Snell-Bergeon, Ph.D., MPH walked us through cardiovascular outcomes that were measured as a part of the ADJUST-T1D study. People treated with semaglutide had significant decreases in cholesterol and blood pressure. After plugging participant data into a cardiovascular risk prediction tool, Dr. Snell-Bergeon reported that people with T1D treated with semaglutide have a 20% reduced CVD event risk after five and ten years. We need more therapies to address CVD in T1D—while large-scale clinical trials are needed, this data demonstrates the potential for GLP-1s to improve cardiovascular outcomes in people with T1D.
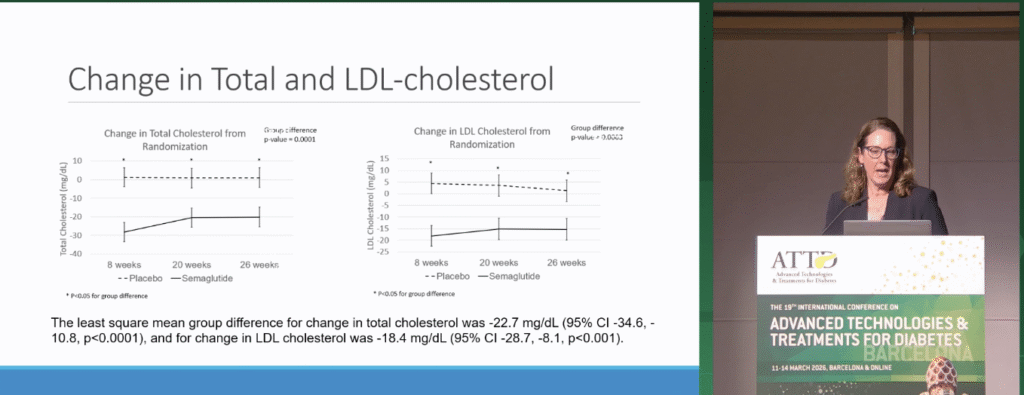
One key thing to keep in mind when it comes to GLP-1s: safe implementation. Clinicians must work hard to avoid hypoglycemia while also preventing the onset of ketones and DKA. Breakthrough T1D has led this work for years, and is currently helping promote several clinical trials, including the SUPRASS-T1D study.
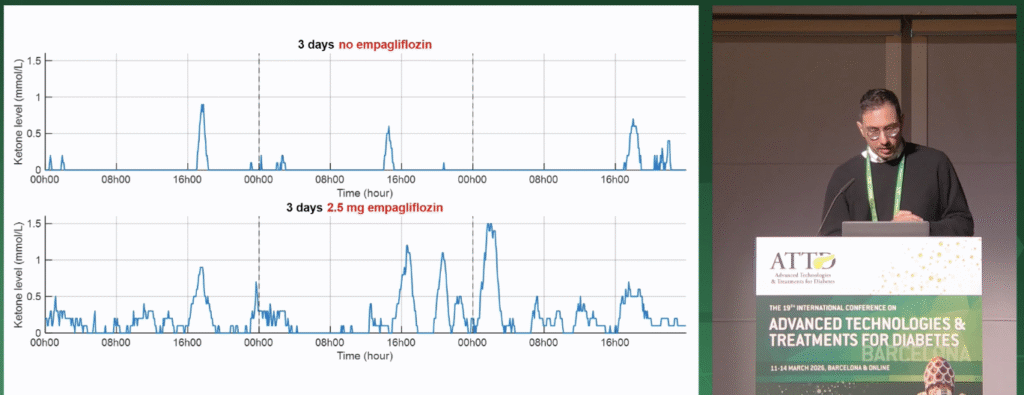
The risk of DKA also exists for a class of drugs known as SGLT inhibitors, which have proven to be effective for heart and kidney disease in type 2 diabetes (T2D) and people without diabetes, but are still being tested in T1D. Breakthrough T1D-funded researcher Ahmad Haidar, Ph.D., presented his work on changing ketone levels dependent on treatment with empagliflozin, an SGLT inhibitor, and lifestyle factors. Participants wore a continuous ketone monitor (CKM), and data showed that empagliflozin, in addition to lower-carbohydrate diets and greater physical activity, can increase ketone variability and the risk of ketosis. Dr. Haidar suggested that people with T1D may benefit from clinical time in range targets for ketones, and his studies demonstrate the potential of tracking ketone levels to predict and avoid DKA.
Thomas Danne wins an award

Breakthrough T1D Chief Medical Officer Thomas Danne, M.D., Ph.D., received the Children with Diabetes (CWD) President’s Award for Innovation. The award acknowledges a healthcare professional who exemplifies extraordinary leadership in using innovation and advancements in technology for devices that help people with diabetes thrive. An internationally recognized leader in endocrinology and pediatric diabetes, Dr. Danne has spent decades advancing research, clinical care, and global collaboration in the field.
“Breakthrough T1D is incredibly proud of Dr. Danne’s achievements and this well-deserved award for innovation and leadership in diabetes health care,” said Aaron Kowalski, Ph.D., Breakthrough T1D CEO. “Thomas’ many contributions to driving breakthroughs in type 1 diabetes therapy development and care have had a tremendous impact on the global T1D community and are instrumental in moving our mission forward, faster.”
Adios from Barcelona!
The next scientific conference we’ll be covering is the Annual American Diabetes Association (ADA) Scientific Session, which takes place from June 5-8 in New Orleans.
And, don’t forget, Breakthrough T1D will be hosting our first ever Clinical and Research Congress in Philadelphia in October! Over two and a half inspiring days, clinicians, researchers, scientists, healthcare professionals, industry leaders, and the T1D community will explore cutting-edge advances in T1D discovery science, immunotherapies, automated insulin delivery, islet cell therapies, psychosocial care, health equity, and more. Designed to foster collaboration and spark innovation, CRC 2026 will provide translational insights, interactive learning, and meaningful dialogue, all with one shared goal: improving lives and driving toward a world without T1D. Register today—and submit abstracts!
This article was written by Brian Herrick and Sandy Vogt, Ph.D.
Greetings from Barcelona, Spain! From March 11–14, 2026, the world’s leading type 1 diabetes (T1D) researchers, healthcare professionals, and industry partners gathered to share groundbreaking advances in T1D science—and Breakthrough T1D–supported work (and staff) were front and center.
Below is everything you need to know from the first two days of this remarkable conference, including key takeaways from panels and sessions chaired by Breakthrough T1D staff and supported researchers. Featured participants included Breakthrough T1D CEO Aaron Kowalski, Ph.D.; Chief Scientific Officer Sanjoy Dutta, Ph.D.; Thomas Danne, M.D., Ph.D., Chief Medical Officer; Vice President of Medical Affairs Anastasia Albanese O’Neill, Ph.D., APRN, CDCES; and many more.
Screening
T1D Screening and Early Detection is a key mission priority for Breakthrough T1D—and multiple presentations illustrated how important it is, and why we need to screen more people to achieve our vision of a world without T1D.
The landscape of T1D screening today is incredibly exciting—and has come a very, very long way in recent years. Across the globe, the number of people being screened, which includes kids and adults, is growing significantly. In addition to the U.S., screening numbers are on the rise across Europe, Australia, Qater, Israel, and now in Central America.
So—what are some big takeaways on screening?
Global screening programs
EDENT1FI, a European screening initiative supported by Breakthrough T1D, presented data from around the world on screening for early detection. One of the presenters was Breakthrough T1D VP of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, who shared our screening strategy and underscored the importance of international consensus recommendations for screening and monitoring that can be used in clinical care. This session outlined the global scope of screening that Breakthrough T1D is supporting.

Breakthrough T1D is funding a truly global network of screening programs. From ELSA in the United Kingdom to ASK and TrialNet in the U.S. to FR1DA in Germany, all these centers are working together to build the case for general population screening. They’re doing this by improving outcomes for people with detected autoantibodies, providing follow-up monitoring, enrolling eligible individuals in clinical trials (including Breakthrough T1D-funded INNODIA), offering FDA-approved therapies like teplizumab (Tzield), and learning the best ways to educate and inform people of the benefits and logistics of screening and follow-up monitoring.
Education and communication are key. One incredible learning from Italy and the United Kingdom is how people learn about screening. For the majority of individuals, it’s through school and through social media. It’s up to all of us to help spread the word. Learn more about our screening programs here.
Another key takeaway from the ELSA study: no one who learns that they have T1D autoantibodies regrets having that knowledge. However, there are gaps around how we can help people with that information and how we can better support families who have detected T1D early.
Free, voluntary general population screening for T1D and Celiac Disease is now law in Italy. Francesca Ulivi from the Italian Diabetes Foundation shared what Italy is learning about how they are increasing the number of people screening—and to add to an earlier note, education is key.
Clinical adoption of T1D screening
In 2024, Breakthrough T1D led the way with the publication of Consensus Guidance for Monitoring Persons with Islet Autoantibody-Positive Pre-Stage 3 Type 1 Diabetes. This report contains guidelines for what to do when T1D autoantibodies are detected—but it does not touch on the bigger question: who to screen?

To make the case for general population screening, Breakthrough T1D convened a group of international experts to establish guidance for the clinical adoption of screening for early detection. This paper is expected to be published later this year and will be a clinical roadmap for general population screening—so more people can realize the benefits.
Breakthrough T1D Chief Scientific Officer, Sanjoy Dutta, Ph.D., also spoke to our organization’s vision for expanding general population screening.
Breakthrough T1D is funding a series of pilot screening programs across the country. These centers aim to develop and test diabetes center workflows for screening and monitoring for T1D. We are learning a lot from these, as presented by Carla Demeterco-Berggren, M.D., who runs a Breakthrough T1D-funded screening pilot in San Diego. Some key takeaways:

- With proper monitoring, DKA is almost completely preventable. Her center has had 0 people who had autoantibodies detected present with DKA at diagnosis.
- Successful T1D screening takes an integrated approach within the hospital setting, from hospital leadership to the nurses running the diabetes clinics.
- More importantly—it takes a team! Screening is just one step. Monitoring is where DKA prevention happens. And it takes a team to do that.
- The more people we screen, the more people can benefit!
Disease-modifying therapies
Disease-modifying therapies (DMTs) have the potential to change the course of T1D by slowing, halting, or reserving its progression. Teplizumab (called Tzield in the U.S.) was the first DMT approved by the FDA in November 2022 for individuals with early-stage T1D, delaying the onset of clinical, symptomatic T1D and the need for insulin therapy by a median of two years. Since its approval, and thanks to increased education and awareness around screening, hundreds of people have had the opportunity to benefit from teplizumab.
Breakthrough T1D and Tzield
Learn more about the first disease-modifying therapy for T1D and how Breakthrough T1D helped guide it through every stage of therapy development.
The changing landscape of available T1D therapies requires healthcare professionals (HCPs) to consider how to best work within existing care structures to support their patients who are using therapies like teplizumab. In an informative session led by Breakthrough T1D-funded researchers Prof. Colin Dayan, BSc, MBChB, Ph.D., FRCP from Cardiff University, Michael Haller, M.D. from the University of Florida, and Prof. Olga Kordonouri from Children’s Hospital Auf der Bult in Hannover, Germany, these superstar endocrinologists walk us through practical considerations for use of teplizumab in clinics and real-world experiences from the perspective of both teplizumab recipients and caregivers.

- People considering teplizumab should work with their diabetes care team to ensure they meet pre-assessment, vaccination, and other requirements and are able to tolerate intravenous access for the duration of the infusion.
- Teplizumab requires 14 consecutive infusion days, and people may have the opportunity to finish the infusion process at home with a home healthcare aide after the first five days in an infusion clinic. Everyone is consistently monitored for safety both during and after the infusion process.
- Multidisciplinary staff, diverse training, and specific infrastructure are required for successful teplizumab infusion, monitoring, and follow-up. This includes infusion nurses, on-call clinicians, pharmacy coordination, thorough documentation, after-hours care, support for travel and lodging for families coming from afar, and centers that have the capacity to deliver teplizumab for 14 days straight.
- In a survey of 47 people who received teplizumab in the U.S., 49% had a family history of T1D, 47% were initially misdiagnosed with type 2 diabetes, and 36% had other autoimmune conditions.
- In the same survey, 98% of people reported that their rationale for choosing teplizumab was to delay T1D onset. While most respondents were grateful to receive the therapy and would recommend it to others, people still check blood sugars often and worry about what to eat.
Cell therapies
While the current standard of care for T1D has taken immense strides in the recent past, one thing is abundantly clear: people with T1D are not doing well enough. Per data presented by Breakthrough T1D-funded researchers Jason Gaglia, M.D., Trevor Reichman, M.D., Jennifer Sherr, M.D., and Jay Skyler, M.D. at Vertex Pharmaceuticals’ Industry Session, globally, about 25% of people with T1D are hitting their ideal HbA1c target of under 7%. Automated insulin delivery (AID) systems—which have been transformative for T1D care—are improving outcomes, but the majority of people are not achieving their ideal goals. This means that most people with T1D are more at risk for the most common complications of the condition, including heart disease, kidney disease, and diabetic eye disease.
Among the individuals who are achieving ideal HbA1c goals, an acute complication begins to emerge: severe hypoglycemic events.
This paints a pretty clear picture: we need better therapies to help people do better, and cell therapies have the potential to fill the gaps between how people are currently doing and where they need to be to prevent long-term and acute complications.
Currently, deceased-donor islet transplants are an option, but they are not an option for the majority of people with T1D due to a scarcity of deceased-donor islet cells (it can take 4 donor pancreases to get enough cells for one person) and the drawbacks of chronic immunosuppression.

The future Vertex and the field at large is driving toward is one where manufactured islets are mass produced at scale, which solves the scarcity problem, and are used with novel techniques to keep them safe from the immune system—including gene-edited cells and alternatives to modern immunosuppression (like Eledon’s tegoprubart).
One other note: someone in the audience inquired as to why more people in the U.S. do not receive islet transplants. The panelists were in agreement that it is due to the current regulatory environment for deceased-donor islet cells, which classifies them as biologics, not organs. This reinforces Breakthrough T1D’s efforts to urge the Department of Health and Human Services to reclassify deceased-donor islet cells as organs—which it has the legal authority to do now.
Improving Lives
All the major T1D tech companies were present and accounted for at ATTD. Omnipod, Abbott, Tandem, Minimed, Dexcom, and more highlighted their current devices and how they can improve diabetes management.
We’re not going to cover the specifics of these devices, but one theme was consistent throughout the presentations: people utilizing AID systems and continuous glucose monitors (CGM) have better outcomes.
But there were some exciting developments around adjunctive therapies, the use of artificial intelligence, and a new approach: gene therapy.
The use of Artificial intelligence (AI) is a hot topic at ATTD. This topic was presented by longtime Breakthrough T1D collaborator Boris Kovatchev, Ph.D., in the opening lecture and Giacomo Cappon, M.D., Marc Breton, Ph.D., and others in a plenary session chaired by Breakthrough T1D CEO Aaron Kowalski, Ph.D.

One of the more interesting areas discussed was the use of digital twins—virtual models of a person with T1D. These models allow researchers and clinicians to test potential changes in diabetes management in a simulated environment before talking through and implementing those adjustments in real life. The potential is big, but there are still risks. AI-generated “hallucinations,” e.g. 2 + 2 = 7, can occur, and any therapeutic recommendations need human review. Even so, the promise of these tools for T1D is significant.
Dr. Kovatchev and Dr. Breton also highlighted how AI is already being used in clinical trials. Their talks focused on AID As a Network (AIDANET), an AI-based AID system. This fully closed-loop setup—using a Dexcom G6, a Tandem t:slim insulin pump, and a smartphone—first learns from insulin pump and CGM data. Once it understands a person’s patterns, it takes over and fully automates insulin delivery. No mealtime announcements required.
Dr. Breton shared data from a Breakthrough T1D–funded study using AIDANET, and the results were striking. People who already had lower HbA1c levels saw modest improvements, while those starting with higher HbA1cs experienced significant gains. In this fully closed-loop, no-bolus approach, AIDANET was better than standard use across nearly every outcome for the 34 people in the clinical trial—and overall, it delivered superior glycemic control.
How does it achieve this? Essentially, AIDANET performs about as well as usual care during the day and then outperforms it at night.
Breakthrough T1D continues to fund this work, including the AIDANET clinical trial in adolescents and young adults that Dr. Breton presented. The takeaway so far: AIDANET delivers equivalent control for people who are already well managed—and even better outcomes for those with higher HbA1c levels.
And here’s something to look out for: there is an ongoing clinical trial testing AIDANET at home in over 50 people with T1D. The data should come out soon—and should be another proof point in support of the use of AI in T1D.
Gene therapy in T1D
Longtime Breakthrough T1D collaborator Jeremy Pettus, M.D., was in attendance to share information from Kriya Therapeutics, a T1D Fund portfolio company. Kriya is pursuing a gene therapy for T1D, and it’s an exciting new approach to achieving glycemic control.
Gene therapies explained
Gene therapies are still fairly new—there are 7 approved today, and they primarily are used for rare diseases. They work by using a safe, non-pathogenic virus to deliver genetic information into the nucleus of a cell. This process does not alter the individual’s chromosomes or genetic makeup but can correct genetic abnormalities or change the behavior of cells to treat disease.
Applications in T1D
Kriya’s approach is to modify muscle cells to secrete insulin. This is very different from, for example, beta cell therapies, which place insulin producing beta cells into people with T1D. In Kriya’s therapy, there are no new beta cells or transplanted cells that require immunosuppression—it’s just enabling muscle cells to make insulin.
The administration of this therapy is fairly straightforward. For 2-3 months in advance of the procedure, the recipient takes immunomodulatory drugs. Then, they undergo a procedure where they receive intramuscular injections in both thigh muscles. Then, the person goes home. After 2-3 months, the cells are fully up to speed and making insulin, which has the potential to help control blood sugar levels and reduce insulin therapy requirements.
Clinical trials
In large animal models, the data is very encouraging—after receiving the therapy, blood glucose levels were normalized on the order of years. Based on this data, clinical trials are expected to begin in people this year.
This phase 1 clinical trial, called the Progress Study, will assess dosage and safety, and the phase 2 trial will expand to assess broader safety and efficacy.
This is a new and innovative approach supported by the T1D Fund. If you’re interested in participating in the clinical trial, stay tuned to our clinical trials page, which is regularly updated with opportunities like this.
Medical Affairs
Breakthrough T1D Chief Medical Office Thomas Danne, M.D., Ph.D. ,outlined the goals of our Medical Affairs team—which is to improve the world’s ability to accelerate implementation of current and future breakthroughs so that, ultimately, people with T1D can have better quality of life.

The current state of endocrinology globally is a growing concern. For example, we don’t have enough endocrinologists and diabetes educators to take on of the growing and aging population of people with T1D. Clinical adoption for teplizumab, the first approved DMT for T1D, has been slow. We must build the infrastructure today to ensure that in the future, there isn’t a gap between the breakthroughs in the lab becoming breakthroughs for people with T1D.
Dr. Danne outlined the vision for a training program that will entice a new generation of endocrinologists as the field evolves and new methods of treating T1D are available. Breakthrough T1D and ATTD are supporting a certificate program for an interventional diabetologist, which includes training in immunology, using immune therapies to treat individuals in stage 1 and stage 2, education on cell therapies for those in stage 3 and stage 4, and the other advancements in the pipeline. He also spoke about other work the Medical Affairs team is involved in including the Centers of Reference, an international collaboration creating a roadmap for implementing manufactured cell therapies into clinics once they become available.
More to come!
We have even more exciting updates to share with you. Keep an eye out for our recap of days 3 & 4 of ATTD, coming next week!
This article was written by Brian Herrick and Sandy Vogt, Ph.D.
This article was written by Alecia Wesner (pictured above, right), a Participant Advisory Council (PAC) member, a Clinical Trial Education Volunteer, and long-time Breakthrough T1D supporter. Alecia’s story details her lived experiences with type 1 diabetes, participation in clinical trials, and her journey to joining the PAC. The views expressed by the author are her own and are not necessarily representative of Breakthrough T1D or our leadership, employees, or supporters.
Where it all began

I was diagnosed with type 1 diabetes (T1D) in 1979 in suburban Philadelphia. I had just finished kindergarten, learned to ride a bike, and spent summer days pedaling the block. I was thirsty all the time. I was a new little cyclist and it was summer, of course I was thirsty! One day I drank an entire quart of iced tea, within mere minutes, at a neighbor’s house. That was the red flag. My pediatrician sent us to the local hospital, and multiple nurses and a doctor held me down as they repeatedly struggled to find a vein for a blood draw. My Dad took me for pancakes afterwards—definitely not ideal without insulin. Later, there was a phone call and my Dad left the house. I saw him sitting outside with his head in his hands. It was the first time I saw him cry. My Mom pulled me away from the window.
I spent the next week and a half in a children’s ward of a Philadelphia hospital. My parents were trained to care for me, and I practiced giving insulin shots on an orange while arts and crafts kept me busy. I spoke to my parents every night on a pay phone, whispering, while crying that I wanted to come home. My parents would say the other children in the ward needed me to be brave. This was long before continuous glucose monitors (CGMs) or even portable blood glucose meters. My parents learned to check my glucose by testing my urine with chemicals in test tubes, a non-exact way to measure glucose range. When I came home, neighbors were anxious because many believed diabetes was contagious.
A designer’s perspective on T1D clinical trials

Clinical trials and research have been central to my life. Clinical trials are essential to proving a concept, where ideas are tested rigorously and then refined.
My training as an industrial designer has always been about the intersection of people and systems. Whether designing a lighting fixture, a user interface, or an educational program, I think first about human needs, usability, and empathy.
That design lens informs how I view research. A study protocol is not just a list of procedures. It is a system that has users, interfaces, and outcomes. Participants are the users. Devices and consent forms are the interfaces. The data is the outcome. When design thinking is applied to research, studies become more accessible, clearer, and better aligned with the realities of daily life.
A defining experience
For those of us living with type 1 diabetes, research is not just academic—it is the path to a better quality of life. I have participated in nine clinical trials and numerous studies. I tested early automated insulin delivery (AID) systems over many years, assessing software and hardware which led to commercially available AID systems. I tested an implantable continuous glucose monitor, and I took part in studies that examined complications and diabetes biomarkers. Each trial taught me about technology, the human body (specifically mine), and how research truly impacts lives.
The experience that defined my clinical trial pathway was in my mid-20’s. I developed retinopathy. Due to a great macular specialist, I received aggressive laser treatments for years. It was scary, especially as a young professional starting a career in product development and visual design. The treatments that preserved my vision were possible because others had volunteered for research for years, even decades, before me. That reality gave me enormous gratitude, and it also gave me a sense of responsibility. Participating in research is how I have been able to embrace a life philosophy of “Do good, feel good” and ultimately pay it forward. Clinical trials and research studies are how we learn and design better solutions for everyone who follows.
Joining the Participant Advisory Council

My designer’s perspective led me to join the Breakthrough T1D Participant Advisory Council (PAC). I had been involved with Breakthrough T1D for many years as a Clinical Trial Education Volunteer, an advocate, and former board member. The PAC offered a new opportunity to bring lived experience directly into the research process.
On the council, we review trial designs, advise on participant burden, critique consent language, and recommend practical changes in the hope of lowering barriers to participation. My concern initially was that I’d be a token voice, but we are not. We are advisors who help researchers see the experience through the eyes of the people who will take part in the studies—people who may ultimately benefit from the data gained.
One of the most meaningful ways I contributed as a PAC member was in June 2025, when I spoke on a panel about disease-modifying therapies (DMTs) at the American Diabetes Association Scientific Sessions in Chicago. The panel focused on disease-modifying therapies designed to preserve C-peptide (a biomarker for insulin production) and slow or stop the progression of T1D. I represented the lived experience, and I brought the perspective of a long-term clinical trial participant and an educator who helps others understand and navigate research opportunities.
Using my voice to spread awareness about clinical trials

There were numerous questions to be addressed. One question asked how patients view trials targeted at specific subsets of the T1D population compared to all open trials. My answer was that subsets are still part of the whole community, and a win in one group can expand to broader benefits. At the same time, being excluded from any study can feel deeply personal. People who have made the decision to participate and then hear they do not qualify may feel a sense of loss and frustration. That is where clear communication and education matter. Explaining why inclusion and exclusion criteria exist, offering information about other potential trials, and keeping lines of communication open helps people stay engaged rather than feeling shut out.

Recruitment, especially for new onset trials, is another challenge the panel addressed. Speed matters because opportunities for early intervention are time-sensitive. Effective recruitment requires a network approach. Endocrinologists and clinicians are critical, but so are community organizations, advocacy groups, and peer networks. Many newly diagnosed people search for help and connect with the T1D community to find resources. When trusted peers share information about trials, it not only demystifies the process and reduces the emotional burden for families facing a new diagnosis—it opens doors.
We also discussed patient-relevant endpoints in DMT trials. Clinical metrics matter, but they do not capture everything people live with. Participants prioritize outcomes that reduce daily burden, delay disease progression, improve long-term health, lower the risk of severe hypoglycemia, preserve endogenous insulin, enhance glycemic control, potentially reduce lifetime treatment costs, and sustain hope. These are the practical changes that matter in everyday life.
Education and implementation were the final themes I emphasized. Clinicians and patients alike need clear information about DMTs and about how early interventions would be implemented in real-world settings. Linking DMTs to screening efforts ensures that people in the early stages of T1D are identified and connected with options. Volunteers, advocacy groups, and community programs are essential for amplifying success stories and helping clinicians and patients understand how to access emerging therapies.

“Research is deeply personal”
All of this ties back to design. When we include lived experience in research design, the results are more humane, relevant, and practical. We reduce friction that prevents people from participating, and we improve outcomes by focusing on the needs of real users. My perspective as both a person who lives with T1D and an industrial designer helps me see where small design decisions have large effects.
Looking forward, I remain committed to this work because research is deeply personal. It saved my vision. It offers better tools and brighter possibilities. It is also a way to honor those who volunteered before me and to give others the chance to benefit. Whether I am advising on study protocols, speaking on national panels, or teaching people how to find trials, I see it as part of the same lifelong commitment: to make life with T1D easier, to improve the design of treatments and systems, and to help guide us toward the ultimate goal: cures.

If you or someone you know may be interested in joining the Participant Advisory Council, please reach out to Michelle Simes-Kennedy at MSimes-Kennedy@BreakthroughT1D.org.
Learn more about clinical trials
Visit Breakthrough T1D’s clinical trials web page to learn more about how you can get involved in clinical trials. Use our clinical trial matching tool to find recruiting trials near you that you may be eligible for. Connect with a Clinical Trial Education Volunteer in your area to better understand the process and get your questions answered.
What’s happening?
Continuous ketone monitoring (CKM) is on its way. A new paper in The Lancet Diabetes & Endocrinology, titled “International expert recommendations on the application and utility of continuous ketone monitoring for people with diabetes,” was published by Breakthrough T1D and expert collaborators, spearheaded by our Chief Medical Officer, International, Thomas Danne, M.D., Ph.D. This publication summarizes the recommendations for effective use CKM use in people with type 1 diabetes (T1D) at risk of developing diabetic ketoacidosis. Read on to learn more about this technology, guidelines for its use, and what this means for the future.
Ketones and DKA: What you need to know
When there is not enough insulin in the body, glucose cannot be used for energy, and the body uses fat for fuel instead. Excessive breakdown of fat ultimately causes ketones to build up in the blood. Very high levels of ketones in the blood can lead to diabetic ketoacidosis (DKA)—a life-threatening condition that requires immediate medical treatment.
Too often, people are diagnosed with T1D only after they have experienced DKA. Yet, even for people who are aware that they have T1D, DKA can and does happen. There are many reasons that it can occur, including the onset of illnesses, like the flu. Currently, people with diabetes use blood and urine strips to test for ketones—but even so, surveys show that very few people measure their ketones regularly, and DKA rates remain unacceptably high.
Given the current options, it remains very difficult to predict when ketone levels may be rising, and people lose opportunities to intervene early to prevent DKA altogether. So, how can we fill this gap?
The answer: continuous ketone monitoring.
CKMs are coming—but they’re not here yet
CKMs are in the works, and we can expect them to become a reality sooner rather than later. Similar to continuous glucose monitors (CGMs), CKMs will continuously measure ketone levels in the body and can alert users if their ketone levels are rising. This ability to track ketone levels in real-time could have substantial benefits in preventing DKA—and more.
Examples of people with T1D who may benefit from CKM
- Insulin pump users, including automated insulin delivery (AID) users, who are at higher risk for DKA due to possible infusion-set or infusion-site failures
- People with T1D and complications, such as chronic kidney disease or cardiovascular disease, who have an increased risk of DKA
- Pregnant women with pregestational or gestational diabetes, who may have increased resistance to insulin and therefore higher rates of DKA
As of today, there are academic groups and early-stage companies working on CKM. Abbott also has a glucose-ketone sensor (CGM-CKM) in development. Other companies, including Tandem, Beta Bionics, Sequel Med Tech, and Ypsomed, are planning to integrate Abbott’s ketone-glucose sensing device into their AID systems once it is available. We’re also keeping an eye a company called SIBIONICS, which currently offers a wellness-focused CKM that has demonstrated strong ketone-tracking data, although it is not available as a medical device yet.
Breakthrough T1D strongly supports the development of CKM to help protect people with T1D from the dangers of DKA. We previously funded five projects—two of which are co-funded by the Helmsley Charitable Trust—to support the development of CGM-CKM. Now, we’re getting ahead of the curve for CKM so we can learn how to use this technology to answer clinical questions about T1D.
We are currently looking for and investing in research projects to understand how ketone monitoring may improve outcomes for people living with T1D from multiple angles, including DKA prevention, understanding disease progression and pathophysiology, learning from clinically meaningful ketone patterns, and more. The goal is to accelerate the generation of evidence surrounding the use of CKM while these devices are on the horizon. This way, we can get a better idea of the kinds of benefits this technology can bring to the T1D community.
Snapshot of CKM recommendations for people with T1D
We feel optimistic that CKMs will become a reality soon. The next question is: how should they be integrated into T1D care? To answer this question, Dr. Danne and an international Breakthrough T1D consensus group published a report about how these novel technologies can be effectively used to help the T1D community.
The experts agreed on the following recommendations:
- CKMs should include trend arrows, akin to those on a CGM, and reflect rates of change of around 0.4 mmol/L per hour.
- Due to the existence of alarm fatigue, alarms should sound if ketone concentrations rise above the threshold of ≥3.0 mmol/L.
- The terminology for ketone levels should be: Normal, Elevated, High, Urgent High.
- All CKM users should be provided with a blood ketone meter to use if they are experiencing the symptoms of High or Urgent High ketones that do not match the CKM reading.
- All individuals who wear a CKM should receive education on what elevated ketones mean and what actions to take.
These are just a few of the highlights. Check out the full publication for a more in-depth look at what the experts recommend.
Why CKM guidelines are important
CKMs have the potential to transform T1D care by stopping dangerous, life-threatening DKA before it ever occurs. It is critically important that healthcare professionals (HCPs) and people living with T1D know how to most effectively use this technology to maximize its benefits.
This publication does just that. It’s a set of established guidelines, put together by experts from around the globe, to help standardize the integration of CKM into clinics once they become available. Both HCPs and the T1D community can refer to these recommendations as this technology is rolled out—and potentially becomes the standard of care—to start realizing the benefits of CKM as soon as possible.
Even more, this document can be used as a resource in future conversations surrounding regulatory decisions and payer coverage. Ultimately, we are preparing all stakeholders for a future that we know will be here soon—one where CKM is a valuable addition to daily T1D management.

“Experts from 12 countries agree: continuous ketone monitoring can be a game-changer. By detecting risk earlier and guiding faster action, CKM has the potential to reduce DKA, strengthen confidence for people with T1D, and transform how we deliver safe, proactive diabetes care.” – Thomas Danne, M.D., Ph.D., Chief Medical Officer, International
How this fits into our Medical Affairs goals
CKMs are coming—how can we ensure they make their way into clinics around the globe? By preparing the T1D community in advance of their arrival, we are setting the stage for CKMs to come. That way, once they are available, HCPs and people with T1D will already know what to do.
In essence, this is clinical adoption: the cornerstone of our Medical Affairs unit. We are preparing the right people at the right time by providing resources, education, and guidelines for a new technology. We are getting our ducks in a row before the new technology is here—so that we are 100% ready when it is.
Thanks to Dr. Danne and the expert consensus panel, we can be ready-as-ever for CKMs to materialize. We look forward to a future in which DKA is easily prevented and the T1D community can fully experience the benefits of continuous ketone monitoring.
How quickly a year goes by—and a lot has happened for type 1 diabetes (T1D) in 2025! In these last few weeks leading up to the new year, Breakthrough T1D would like to take a moment to reflect on how far we’ve come in the past 365 days. Read on for the greatest T1D accomplishments in 2025—and the exciting things we’re looking forward to in 2026.
A special note for our donors
Breakthrough T1D would like to give a special thank you to our donors. Each and every one of us —staff, volunteers, T1D community members, researchers, advocates, clinicians, and more—are able to accomplish what we can because of you. Our shared commitment to a world without T1D is driven by your generosity, and truly no words can adequately express our appreciation for your continued support of our mission. Thank you for all that you do!
Check out our Top Advances 2025 video featuring Breakthrough T1D CEO Aaron Kowalski, Ph.D., and read on for more details!
Cell therapy milestones
First things first: cell therapies. One of the biggest breakthroughs that had the T1D community buzzing in the last year was Sana Biotechnology’s donor-derived islets that are genetically engineered to be invisible to the immune system. These cells are still making insulin 6+ months after the first person was treated—without immunosuppression! Sana—a portfolio company of the T1D Fund: A Breakthrough T1D Venture—published this accomplishment in The New England Journal of Medicine.
On the manufactured cell therapies front, Vertex Pharmaceuticals continues to hit milestones with zimislecel (formerly VX-880)—a therapy that Breakthrough T1D has had a hand in for decades. Promising results from the phase 1/2/3 clinical trial were published in The New England Journal of Medicine, revealing that 10 of 12 participants (83%) are insulin therapy independent. The study is ongoing.
Tegoprubart, an immunomodulatory drug made by the T1D Fund portfolio company Eledon, is being investigated as an alternative to standard immunosuppressants to help prevent transplanted islets from rejection. In a Breakthrough T1D-funded trial, six individuals who have received a donor islet transplant and tegoprubart are now insulin therapy independent. Preliminary data suggest that tegoprubart is more tolerable and has milder side effects compared to standard immunosuppressants—paving the way for easier post-transplant management. The study is ongoing, and nine people total are enrolled. The promising results from this trial opened the doors to another Breakthrough T1D-funded study that will include people with T1D and chronic kidney disease (CKD)—a population that was previously excluded from islet transplants due to side effects associated with standard immunosuppressants.
Cell Pouch is an implantable device that provides a livable environment for transplanted islets to help them survive. The ongoing phase 1/2 trial investigating Cell Pouch with donor-derived islets is on track to meet its endpoints for safety and tolerability, and people are achieving insulin therapy independence. The final cohort of this trial, which is expected to initiate before the end of this year, will use manufactured islets provided by Sernova’s partner Evotec. Even more, this cohort will receive Eledon’s tegoprubart instead of standard immunosuppressants—accelerating progress on two fronts at once. Breakthrough T1D has been supporting the development of Cell Pouch over many years, and we look forward to seeing data from the final cohort.
Vertex and large-scale manufacturing
While zimislecel is in the works, Vertex has forged collaborations to manufacture these cells at a large scale once they are approved. They’ve partnered with Lonza to build a dedicated large-scale facility for eventual commercial production of zimislecel. They’ve also licensed technology from TreeFrog to scale up production capabilities. We call that planning ahead!
Civica’s low-cost, long-acting insulin available on January 1, 2026
Thanks to a huge win for insulin affordability, the first day of 2026 will kick off with a bang. Civica Rx’s insulin glargine-ygfn (which is interchangeable with Lantus®), will be available on January 1, 2026, for no more than $55 for five pens. Anyone with a prescription—regardless of insurance status—will be able to purchase it at a pharmacy. Breakthrough T1D has been working with Civica for nearly three years to make this possible.
Positive outlook for disease-modifying therapies
Building on results from a promising study funded by Breakthrough T1D, Eli Lilly launched two new pivotal clinical trials for baricitinib, a JAK inhibitor that is already approved for other autoimmune diseases. The trials will test whether baricitinib—which is taken as a once-daily oral pill—can delay the progression of stage 2 to stage 3 T1D (BARICADE-DELAY) or preserve beta cells in newly diagnosed stage 3 T1D (BARICADE-PRESERVE). Enrollment will open in 2026.
Another disease-modifying therapy is coming down the pike: ATG. Following from prior studies, the phase 2 MELD-ATG clinical trial found that low-dose ATG has the potential to preserve insulin-producing beta cells in children and young adults between five and 25 years old, and it was generally well-tolerated. In line with ATG’s potential, SAB Biotherapeutics—a company with funding support from the T1D Fund—is developing a next-generation ATG therapy.
Tzield, the first disease-modifying therapy approved to delay stage 3 T1D in people eight years and older in stage 2 T1D (before insulin therapy is required), has been accepted into the FDA Commissioner’s National Priority Voucher (CNPV) program for accelerated review. If approved, this could lead to expanded use of Tzield for people with newly diagnosed stage 3 T1D (when insulin therapy is required)—and this would be the first time people at this stage could have a therapy option besides insulin. We look forward to hearing more soon.
Right now, Tzield is only approved for use in delaying progression from stage 2 to stage 3 T1D for kids and adults eight years and older. The PETITE-T1D clinical trial is trying to change that. Interim results showed that Tzield is safe and well-tolerated in children under eight with stage 2 T1D, with no new safety issues reported. The study is ongoing and final results are expected in 2026.
Tzield in action: Breakthrough T1D’s Chris Dunn
Chris Dunn is a parent of four children, two of whom live with T1D. Knowing that immediate family connections are a risk factor for developing T1D, Chris and her children without T1D participated in screening. Much to her surprise, Chris learned that she was in stage 2. Chris knew that Tzield could potentially delay her need for insulin therapy, so she made the informed decision to try it.
A big win for kidney disease, plus new trials, devices, and more to improve the lives of people with T1D
Bayer shared data from the phase 3 FINE-ONE clinical trial, which investigated finerenone (Kerendia®)—a drug that is already approved for type 2 diabetes—for CKD associated with T1D. The results showed that finerenone significantly reduces a marker of kidney damage, marking the first treatment in nearly 30 years to achieve positive outcomes for CKD in people with T1D. This data will be submitted for regulatory review to expand finerenone’s indication to include T1D.
The Breakthrough T1D-funded phase 2 ADJUST-T1D trial investigated whether the GLP-1 receptor agonist semaglutide (Ozempic®) could benefit people living with T1D and obesity or overweight who are using an automated insulin delivery (AID) system. Thirty-six percent of people in the trial treated with semaglutide met the primary endpoints for glycemic and weight control compared to 0% in the placebo group, and the drug was well-tolerated and safe. These data—which were published in The New England Journal of Medicine Evidence—show that GLP-1 receptor agonists have the potential to help people with T1D manage both blood sugar and weight.
Building on the growing interest in GLP-1s for T1D, Eli Lilly launched two pivotal clinical trials for tirzepatide (Mounjaro® or Zepbound®), a dual GLP-1/GIP receptor agonist. These trials—which are enrolling now—will investigate whether tirzepatide can reduce blood sugar levels in people living with T1D and obesity or overweight over a period of 40 weeks (SURPASS-T1D-1) or 20 months (SURPASS-T1D-2). Industry investment in GLP-1s for T1D is a promising avenue toward a future where this class of drugs may be approved for use in members of the T1D community.
AID systems are getting better each year, and approximately 10 such systems are now approved for use in the United States. Here are a few advances in diabetes technology that stood out:
- Dexcom’s G7 15-day sensor—the longest-lasting FDA-approved wearable continuous glucose monitor (CGM) on the market—is now available for people in the U.S. 18 and over.
- Sequel Med Tech’s twiist AID system, which uses FDA-cleared Tidepool Loop technology, is now available in the U.S.
- Tandem’s SteadiSet 7-day infusion set has been cleared by the FDA and is expected to launch in the U.S. in 2026.
- Abbott’s Freestyle Libre 3 Plus sensor has integrated with twiist and T:slim X2 insulin pumps.
- Medtronic unveiled the largest real-world dataset pulled from users of the MiniMed 780G AID system.
To better understand the mental health needs of the T1D community, Breakthrough T1D and the Helmsley Charitable Trust co-hosted a Psychosocial Roundtable, convening mental and behavioral health experts across the T1D care spectrum. The discussion revolved around challenges and gaps in psychosocial care for people with T1D and how we can work together to address them. Thanks to the expert opinions of the attendees, Breakthrough T1D has a clear path forward for tackling these issues and supporting the mental and emotional wellbeing of the T1D community through research and awareness.
Making headway in rebalancing the immune system in T1D
Sanofi has entered into a licensing agreement with EVOQ Therapeutics, a company with support from Breakthrough T1D, to help develop and commercialize its NanoDisc technology. This technology has the potential to retrain the immune system to stop attacking beta cells. Industry partnerships like these are key to accelerating T1D research.
The Nobel Prize in Physiology or Medicine was awarded to three immunologists—Fred Ramsdell, Ph.D., Mary E. Brunkow, Ph.D., and Shimon Sakaguchim M.D., Ph.D.—for their discoveries in immune tolerance, meaning the immune system’s ability to distinguish between self and non-self. Building on these breakthroughs, Dr. Ramsdell co-founded Sonoma Biotherapeutics, a T1D Fund-backed company, to restore immune tolerance in autoimmune diseases like T1D. Another year, another researcher with ties to Breakthrough T1D winning the most prestigious award in science!
Launch of Breakthrough T1D’s Medical Affairs department
A noteworthy organizational change is marked by the launch of Breakthrough T1D’s Medical Affairs unit, which aims to bridge the gap between access to and adoption of T1D therapies, devices, and treatments. The team—which continues to expand—is tackling challenges associated with slow clinical adoption of T1D therapies. In less than a year, the team has already launched healthcare professional (HCP) education and resources and hosted two international workshops at Breakthrough T1D HQ focused on preparing the clinical workforce to bring next-generation cell replacement therapies to the T1D community. This is just the beginning, and we can’t wait to see what else our Medical Affairs unit will accomplish!
Continuous ketone monitoring consensus guidelines published
Continuous ketone monitoring (CKM) has substantial benefits for the T1D community, including preventing diabetic ketoacidosis. CKMs are coming—and to prepare, Breakthrough T1D’s Chief Medical Officer, International Thomas Danne, M.D., Ph.D., spearheaded an effort with expert collaborators to publish consensus guidelines for use of CKM in T1D. These guidelines will help HCPs integrate CKM into clinics and establish best practices for their use in the T1D community.
Tackling T1D on a global scale
Breakthrough T1D, in partnership with the Helmsley Charitable Trust and Roche and Sanofi’s Global Health Unit, launched ALIGN-T1D. This is a new global alliance uniting philanthropic, industry, and community leaders to strengthen government-led T1D care networks in low-middle income countries, integrating diagnosis, insulin access, monitoring, and education.
Breakthrough T1D helped organize an event at the European Parliament to highlight unmet needs for the T1D community and discuss how to accelerate cures—especially cell therapies—in the European Union. In a similar vein, an event co-hosted by Breakthrough T1D in Brussels, Belgium sparked conversation about overcoming challenges surrounding cardiovascular disease in T1D. Building long-term partnerships with EU Institutions and international diabetes organizations will allow us to work together towards global T1D breakthroughs.
Breakthrough T1D, in partnership with Friends of Mewar and UNICEF, participated in the Udaipur Type 1 Diabetes Summit: Advancing Access, Equity, and Action, in Udaipur, Rajasthan, India. The event gathered government leaders, health experts, community advocates, and international partners to develop a collaborative roadmap for strengthening T1D care across India.
Breakthrough T1D, Breakthrough T1D Canada, and the Stem Cell Network partnered to support four new projects led by Canadian researchers to drive high-impact research into manufactured cells and next-generation cell replacement therapies. This partnership is a novel and meaningful part of Breakthrough T1D’s global Project ACT effort to power high-impact cell therapies research.
The Australian government committed $50M over five years to the T1D Clinical Research Network (T1DCRN), a highly impactful research network driven by Breakthrough T1D Australia. This funding will support T1D research in prevention, precision medicine, and cures.
The Grand Challenge, a partnership between Breakthrough T1D UK, the Steve Morgan Foundation, and Diabetes UK, is funding 23 research projects to advance new treatments and cures for T1D. Their funding is supporting 189 researchers and collaborators across 49 institutions in eight countries. Recent breakthroughs from the past year include uncovering why T1D could be more aggressive in young children and developing a new insulin-glucagon molecule that could reduce dangerous hypoglycemic events.
Breakthrough T1D publications across the map
Breakthrough T1D Research and Advocacy staff, in collaboration with other leading experts in the field, authored a paper titled “Future Directions and Clinical Trial Considerations for Novel Islet Beta Cell Replacement Therapies for Type 1 Diabetes” in the journal Diabetes. This publication outlines what the future of beta cell replacement therapies looks like—and how we can make these therapies a reality for everyone with T1D who wants them through innovative clinical trial design and expanding the pool of eligible trial participants.
“The Urgent Need for Breakthrough Therapies and a World Without Type 1 Diabetes” was authored by Breakthrough T1D staff and leadership and published in Diabetes Therapy. This commentary exposes the many persistent challenges of living with T1D, despite incredible progress in recent years. The publication is a call to action for the entire T1D community—researchers, clinicians, advocates, people with T1D, regulators, and all other stakeholders—to work together to overcome barriers at all levels to make cures for T1D a reality sooner rather than later.
Breakthrough T1D Advocacy staff and collaborators published a paper titled “Perceptions of the Benefits and Risks of Novel Therapies for Type 1 Diabetes: A Qualitative Study” in the journal Diabetes Therapy. This Breakthrough T1D-funded study found that people with T1D and their caregivers are largely willing to try next-generation therapies, drawn by the promise of reduced reliance on insulin and freedom from constant disease management. These findings highlight the importance of ensuring that the perspectives of people with T1D and their caregivers are used to guide regulatory decisions around the benefits and risks of a new therapy.
Breakthrough T1D Advocacy staff and collaborators published an article called “We Are on the Verge of Breakthrough Cures for Type 1 Diabetes, but Who Are the 2 Million Americans Who Have It?” in The Journal of Health Economics and Outcomes Research. This study identifies key T1D demographics in the U.S. and predicts how these demographics may change over the next decade. These data will help inform elected officials and payers about how healthcare policies may ultimately affect the T1D community—ensuring that the policies in place are having the greatest impact they can.
An analysis by Avalere Health, which was supported by Breakthrough T1D, found that research funded by the Special Diabetes Program (SDP) has yielded more than $50 billion in federal healthcare savings. This study supports the fact that the SDP has a strong return on investment, both clinically and economically, and demonstrates its ever-important role in bringing advanced therapies to the T1D community and improving health outcomes. Even more, SDP funding was recently extended through January 30, 2026, with retroactive funding back to October 1, 2025.
Consensus guidance on population screening for T1D coming soon
Breakthrough T1D’s Vice President of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, spearheaded an effort to establish a consensus on T1D screening guidance. These guidelines—which will be published soon—push for population-level T1D screening and provide guidance for HCPs to effectively integrate T1D screening into their clinics. The publication will outline the benefits, harms, and methods of T1D screening, who should be screened and how often, and how to effectively communicate screening results.
Breakthrough T1D announces first Clinical and Research Congress in October 2026
In a historic first, Breakthrough T1D is hosting our very own Clinical and Research Congress next fall! This meeting will bring together clinicians, researchers, scientists, HCPs, industry leaders, and the T1D community to explore cutting-edge advances in T1D research, clinical care, health equity, and more. The goal is to foster collaboration and spark innovation to drive our mission forward faster than ever.
Bonus advance: The first Barbie doll with T1D
In December 2023, Mattel approached Breakthrough T1D with a partnership opportunity for the first Barbie with T1D. Less than two years later, in July 2025, Barbie with type 1 diabetes was launched to a most enthusiastic reception from Delegates at Breakthrough T1D’s Children’s Congress. This groundbreaking global collaboration reflects a shared commitment to ensuring that the millions of people living with T1D are seen, heard, and empowered. It’s working—the dolls have been in high demand since launch!
One word: WOW! That’s a lot of exciting things that YOU helped make possible. Thank you for your support of Breakthrough T1D and everything you do to drive our mission forward. Each year, each month, each day, we get closer to a world without T1D. This wouldn’t be possible without our donors, community members, staff, leadership, and every single heart and mind around the globe affected by the work we do each day. Cheers to a successful 2025—and here’s to another year of accomplishments!
What’s happening?
Today, Breakthrough T1D co-hosted an event with leading international diabetes organizations to discuss cardiovascular disease (CVD) and type 1 diabetes (T1D)—where we are, challenges that remain, and how we can work together in Europe and beyond to address this need for the T1D community.
The focus of the event: Doing more for CVD and T1D

This event, titled “Type 1 Diabetes & Cardiovascular Disease: From Data to Solutions,” took place today on November 14, 2025, in Brussels, Belgium. Breakthrough T1D co-hosted this event in conjunction with the International Diabetes Federation (IDF) Europe and the International Society for Pediatric and Adolescent Diabetes (ISPAD). The goal was to bring together different stakeholders to address a critical concern: the lack of therapies for cardiovascular disease for people living with T1D.
The speakers at the event covered a range of topics. Jonathan Rosen, Ph.D., Director of Research at Breakthrough T1D spoke about the often-overlooked link between CVD and T1D. Others touched upon lessons we can learn from (T2D) diabetes and CVD, the gaps in evidence that still exist, new frontiers in CVD care for people living with autoimmune diseases, and the inclusion of T1D in CVD therapy development.
Attendees
From Breakthrough T1D:
- Sanjoy Dutta, Ph.D., Chief Scientific Officer
- Jonathan Rosen, Ph.D., Director of Research
- Carmen Hurtado del Pozo, Director of European Research
- Hilda Ahnstedt, Program Officer of European Research
- Alessandro Bisio, M.D., Director of Medical Affairs, International
Other attendees:
- Speakers from the European Medicines Agency
- Member of the European Parliament: Elena Nevado del Campo (EPP, Spain)
- People living with T1D
- Researchers
- Industry leaders
Why this matters

People living with T1D have a high risk of developing heart complications, despite advances in T1D care. At this time, there are only a few options available to lower this risk. In the United States and Europe, people with T1D have access to blood pressure medications and lipid-lowering medications (for example, statins) that can reduce the risk of developing CVD.
Other therapies, like the SLGT inhibitors empagliflozin or dapagliflozin, are the standard of care in the U.S. to treat heart failure in people without diabetes or with T2D. This class of drugs has revolutionized treatment and significantly reduced the rates of heart failure in these populations. However, people with T1D were excluded from these clinical trials—despite the fact that heart disease remains a critical concern for the T1D community. While not strictly approved, these drugs are occasionally prescribed off-label to reduce the risk of heart failure in people with T1D. Greater accessibility will require regulatory approval, and these options are not currently available for people living in Europe.
Despite the availability of some treatments that can reduce the risk of heart complications, there are no available cardioprotective therapies in the U.S. or Europe that can prevent CVD in people with T1D. More targeted, innovative therapies and treatment strategies are needed to further reduce the risk of—or ideally prevent altogether—cardiovascular complications in the T1D population.
There is a clear unmet need for the T1D community: the risk of CVD is high, and there are not enough options to treat or prevent it. That’s where events like these come in. They are a call to action for collaborative research efforts, better data integration, new and innovative ideas, and global cooperation towards a shared goal of better heart health for people living with T1D. The growing prioritization of CVD is evidenced by the European Commission’s cardiovascular health plan—which includes the Joint Action on Cardiovascular Diseases and Diabetes—showcasing how stakeholders in Europe are taking charge to accelerate breakthroughs for CVD and T1D.
What we’re saying
“Cardiovascular disease remains the leading cause of death for people with type 1 diabetes, yet targeted therapies and guidelines are still lacking. In collaboration with IDF Europe, ISPAD, and aligned with the European Commission’s cardiovascular plan, this event brings together researchers, clinicians, patient advocates, regulators, and industry partners to drive collaborative solutions and accelerate progress toward better prevention and treatment strategies.”
Addressing the challenge of cardiovascular complications is a cornerstone of our Improving Lives strategy. Breakthrough T1D is actively supporting clinical trials for therapies that can reduce or prevent CVD in people with T1D, and we are working on identifying new projects and researchers to fund to further accelerate these efforts.
We are committed—on a global scale—to making sure people with T1D live the best lives possible, which includes having accessible therapies that reduce the risk of heart complications. Events like these bring together the right people that can make this happen in Europe and beyond.
Bonjour from Montreal, Canada, where the latest and greatest in type 1 diabetes (T1D) research was presented at the International Society for Pediatric and Adolescent Diabetes (ISPAD) 51st Annual Conference. ISPAD is a yearly highlight of the T1D conference calendar, and this year was no exception. Scientists, clinicians, researchers, industry members, people with diabetes, and more were on hand to provide updates in cures, improving lives, access, and more—with much of it being supported by Breakthrough T1D.
Let’s take a look at the highlights.
About ISPAD

ISPAD is one of Breakthrough T1D’s peer organizations. They are dedicated to childhood and adolescent diabetes all over the world. In addition to collaborating in scientific advancements, Breakthrough T1D and ISPAD also fund several grants together—including providing support for clinical fellows from around the world to learn how to care for people with T1D from top-tier clinicians.
Tzield is being studied in kids under 8
Tzield has been in the news for a variety of reasons lately. Sanofi—the manufacturer of Tzield—recently received a voucher from the FDA for the use of Tzield in symptomatic, stage 3 T1D (for which it is not yet approved), potentially expediting the regulatory review process.
Tzield is currently approved for children and adults aged eight years and older in stage 2 T1D to delay the onset of stage 3 T1D. An ongoing study is investigating if Tzield can delay the progression of T1D from stage 2 to stage 3 in a new demographic: kids under eight.
At the ISPAD conference, researchers presented an interim analysis from the PETITE-T1D study, which is investigating if Tzield is as safe and tolerable in kids under eight in stage 2 T1D as it is in older individuals.
So far, 23 children have received a 14-day course of Tzield. To date, the safety and tolerability profile of Tzield is similar to that of older individuals who receive the drug. Two children in the study have progressed to stage 3 T1D.
This study is ongoing and we look forward to seeing more data.
Key takeaway
Tzield can delay T1D onset by an average of 3 years. This important study is determining if a new population—kids with T1D under eight years old—can benefit, and the data so far is encouraging.
Breakthrough T1D taking the stage

Breakthrough T1D leadership was on site to present and chair sessions.
- Chief Scientific Officer Sanjoy Dutta, Ph.D., gave an overview of Breakthrough TD’s mission strategy and chaired a session jointly with ISPAD.
- Chief Medical Officer, Global, Thomas Danne, M.D., was there to discuss Time in Normoglycemia, which is blood sugars between 70 and 140 mg/dl, and how technology can help kids achieve it.
- Vice President of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, served as faculty for the 21st Annual ISPAD Science School for Health Professionals that took place immediately before the ISPAD Conference.
- Community Engagement & Communications Director, Global Access & Global Responsibility, Renza Scibilia, hosted a symposium on advancing diabetes care in complex settings.
Technology improves outcomes
A motif that was repeated by many, many investigators: automated insulin delivery (AID) systems help people do better. This spans several different technologies and systems: Tandem Control IQ, Minimed 780G, Cam APS, and OmniPod. Here’s why:
- The near-constant adjustments made by these systems are always working to meet glycemic targets.
- They can help people get closer to their glycemic targets with less user input.
- They lower the mental burden of T1D, helping people less engaged with managing their T1D do better.
Key takeaway
It is widely established that AID systems lead to better outcomes. It’s important to continue to ensure that everyone has access to these systems and that they work better with less user input. They must become the standard of care across the world for kids with T1D.
The importance of clinical trials for stage 3 T1D
Stage 3 is when people are typically diagnosed with T1D. At this stage, beta cell function has not been completely eliminated. Individuals are still making insulin—not enough, but some. Decades of research by Breakthrough T1D and others has demonstrated clear benefits to holding onto beta cell function for as long as possible.
Clinical trials for individuals in stage 3 are few and far between. While there are exceptions (like the recently announced BARICADE-PRESERVE), we need more trials because people in stage 3 T1D can benefit from therapies to keep these cells alive and producing insulin for longer.
Key takeaway
Prioritizing clinical trials for stage 3 T1D—and recruiting for them—can be a bridge between better glycemic control today and future cures.
See you in 2026

2026 is going to be a big year—with many more T1D conferences to cover. This includes, for the first time, our very own conference: the 2026 Breakthrough T1D Clinical and Research Congress.
Stay tuned for the 2025 Top Advances piece in the coming weeks to see everything we’ve accomplished this year as we look towards the next one.
To drive our mission forward, Breakthrough T1D financially supports type 1 diabetes (T1D) research through training awards and grants to academic scientists or clinicians, investments in promising companies, conference support, and more. Here, we take a deeper dive specifically into our training awards offered to early-stage researchers to understand the impact and outcomes that these awards have on individual careers and T1D research as a whole.
Key Takeaways
- Breakthrough T1D conducted a survey of researchers and clinicians who received training awards from 2015 to 2024, totaling $62,513,719 in funding.
- The purpose was to understand the impact of the training awards on career decisions and the research and clinical outcomes stemming from the awards.
- Most Breakthrough T1D award recipients stayed in diabetes research or clinical practice after the award ended.
- Training award recipients reported publications, patents, honors, clinical trials, collaborations, resources, and mentoring of new trainees arising from Breakthrough T1D training awards.
- Recipients gave positive feedback on the awards, and many continue to be involved in Breakthrough T1D beyond applying for funding.
Why did we conduct this survey?
Training awards are a key part of Breakthrough T1D’s grants portfolio. These awards support early-career scientists and clinical researchers to explore promising ideas and encourage trainees to follow career paths that drive T1D research forward. Our approach is designed to provide opportunities to support trainees from an early postdoctoral stage (after receiving a higher degree) to transitioning to an independent lab and career as a T1D researcher.
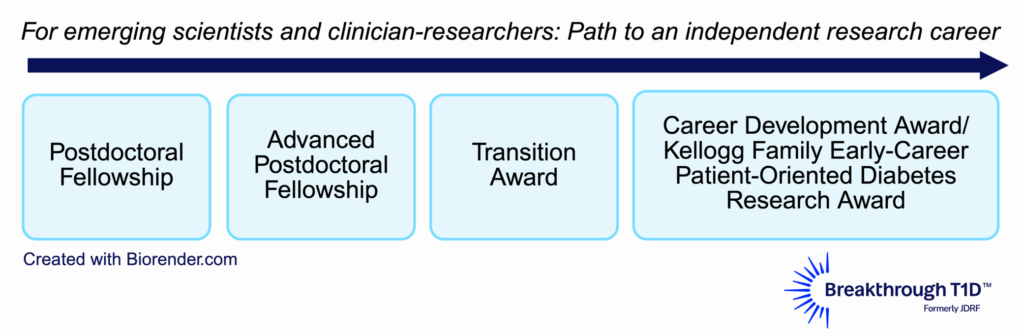
Breakthrough T1D previously conducted surveys analyzing the impact and outcomes of training awards completed between 1974 to 2014, revealing that these awards encouraged long-term pursuit of a career in T1D. The current survey included an expanded analysis on training awards completed between 2015 to 2024. The results provide a broader understanding of how our financial support impacts research outcomes, career paths, and additional funding secured by awardees. This way, we can ensure that our funds for trainees are being used in the best way possible, supporting the best and brightest researchers and ideas that drive our mission forward now and in the future.
Survey methodology
From 2015 to 2024…
$62,513,719
Dollar amount of Breakthrough T1D funds invested in training awards
269
Number of training awards granted
230
Number of unique award recipients
Types of training awards surveyed
| Training award | Number of awards granted between 2015-2024 | Number of awards evaluated based on survey responses |
|---|---|---|
| Postdoctoral Fellowship | 155 | 80 |
| Advanced Postdoctoral Fellowship | 48 | 35 |
| Career Development Awards | 36 | 26 |
| Kellogg Family Early Career Patient-Oriented Diabetes Research Awards | 8 | 4 |
| Transition Awards | 22 | 17 |
| Total | 269 | 162 |
Of the total training awards funded between 2015 to 2024, this survey is a reflection of 135 respondents (60%) covering 162 awards. Respondent data was analyzed, mapped, and reported.
Impacts on career trajectories
17
Number of countries where respondents are located
76%
Percent of respondents who remained in academia after award completion
79%
Percent of respondents still working in diabetes (including T1D and type 2 diabetes) in both research and/or clinical practice
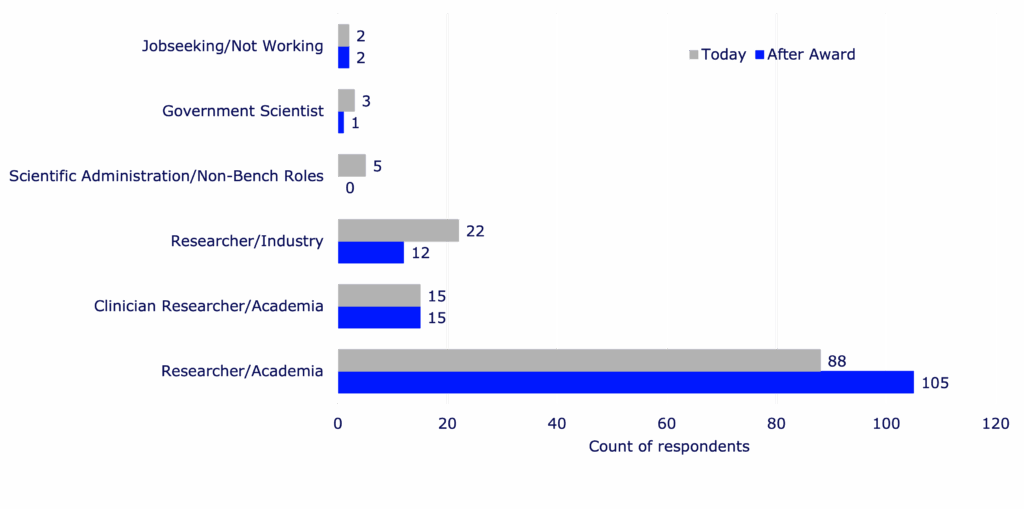
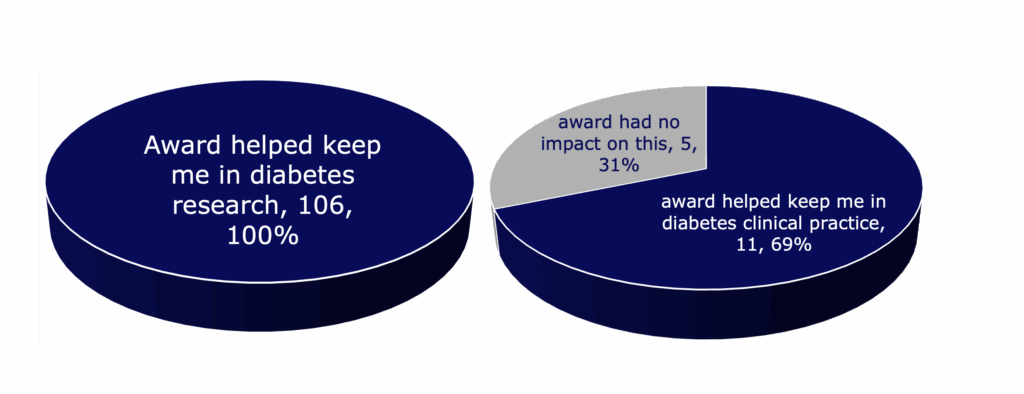
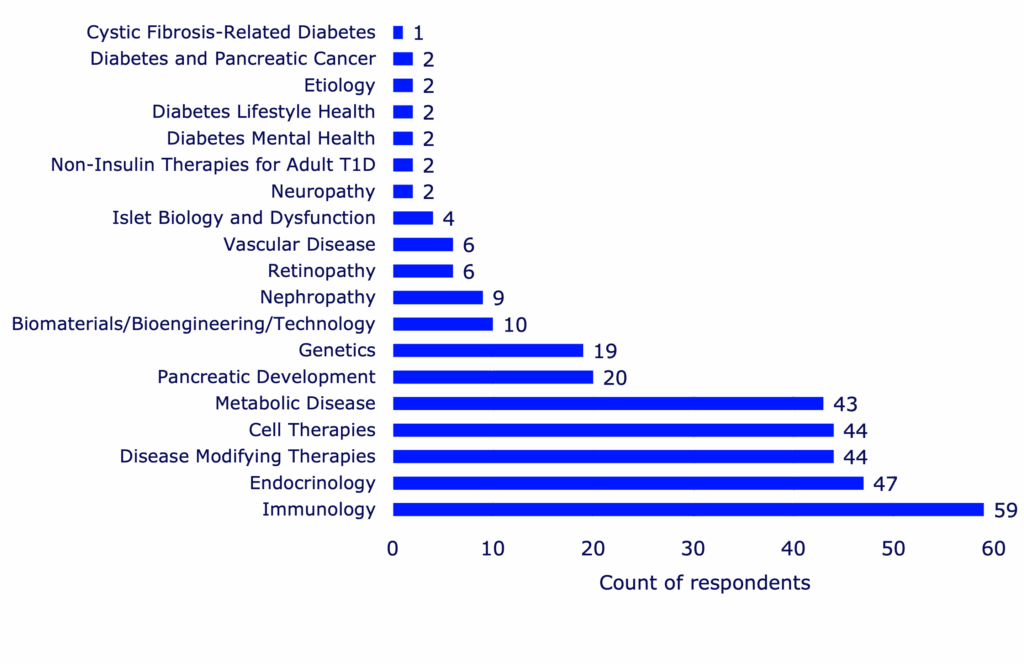
Essential findings: Career trajectories for training award recipients
- Researchers take on more senior positions post-Breakthrough T1D award, including tenure-track roles.
- Every respondent working in diabetes research reported that the training award influenced their decision to continue to focus on diabetes.
- Those who received more advanced training awards were more likely to stay in the diabetes field.
- The main reasons researchers and clinicians stayed in diabetes are that the awards support training, networking, and publications.
- If given the option, most respondents chose the Breakthrough T1D training award over other training award options, primarily due to prestige and better award terms.
Impacts on research outcomes
Research supported by training awards resulted in a variety of outcomes that could benefit the larger scientific community and drive T1D research forward. Readouts of successful research include peer-reviewed journal publications, clinical trials, patents, collaborations, resources, grant review requests, and research honors or recognitions.
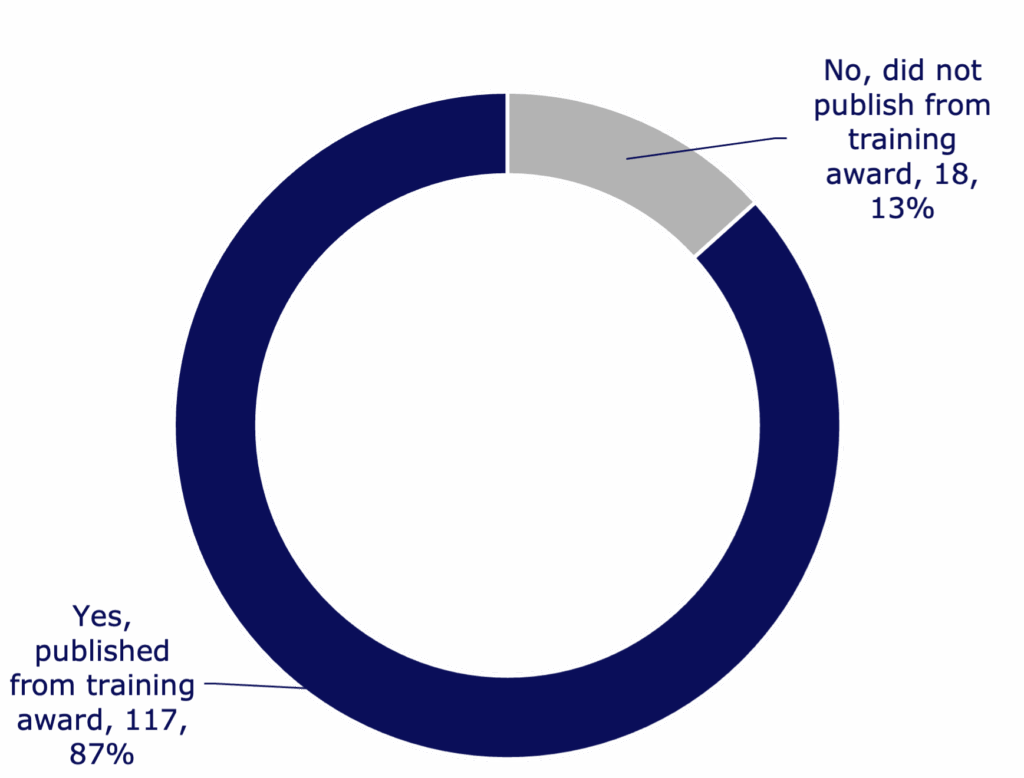
463
Number of published papers reported by respondents
176
Number of journals that respondents published in
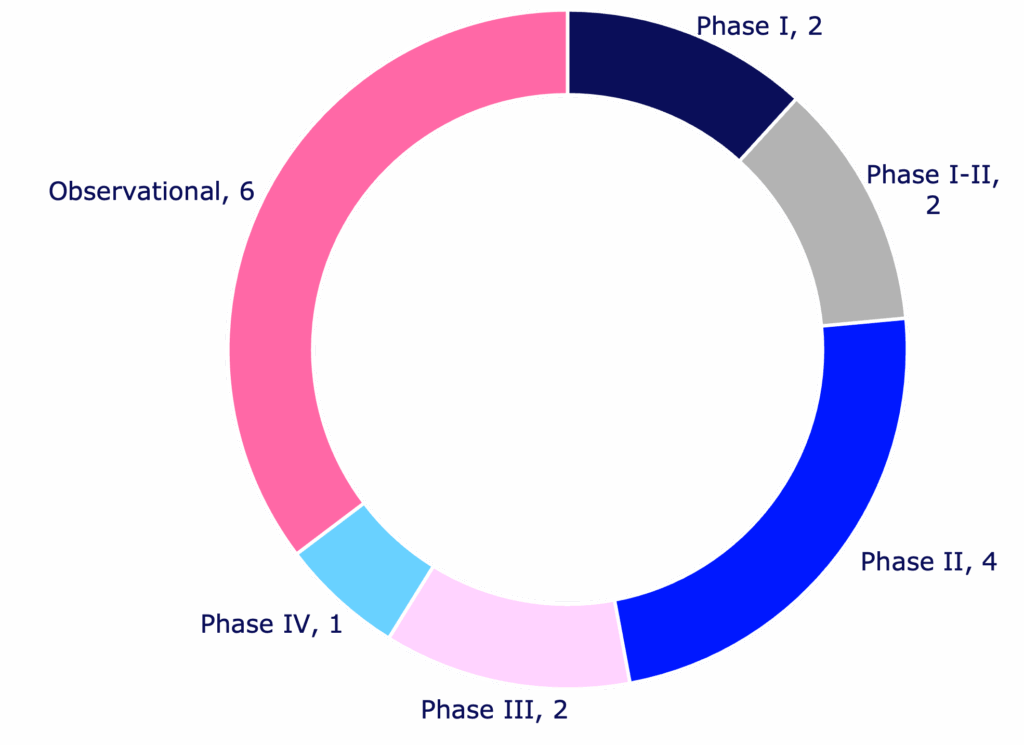
41
Number of patent-related activities reported from training awards
57%
Percent of respondents who formed at least one and often multiple collaborations throughout the training award
57
Number of different shareable resources developed by award recipients
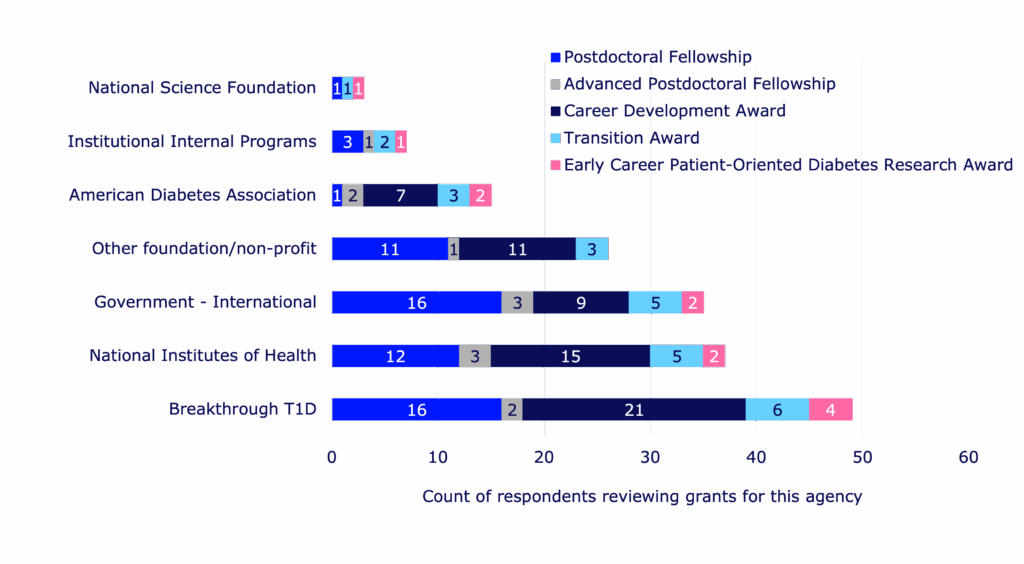
39%
Percent of respondents who serve as grant reviewers for Breakthrough T1D
31
Percent of respondents who reported receiving at least one research honor
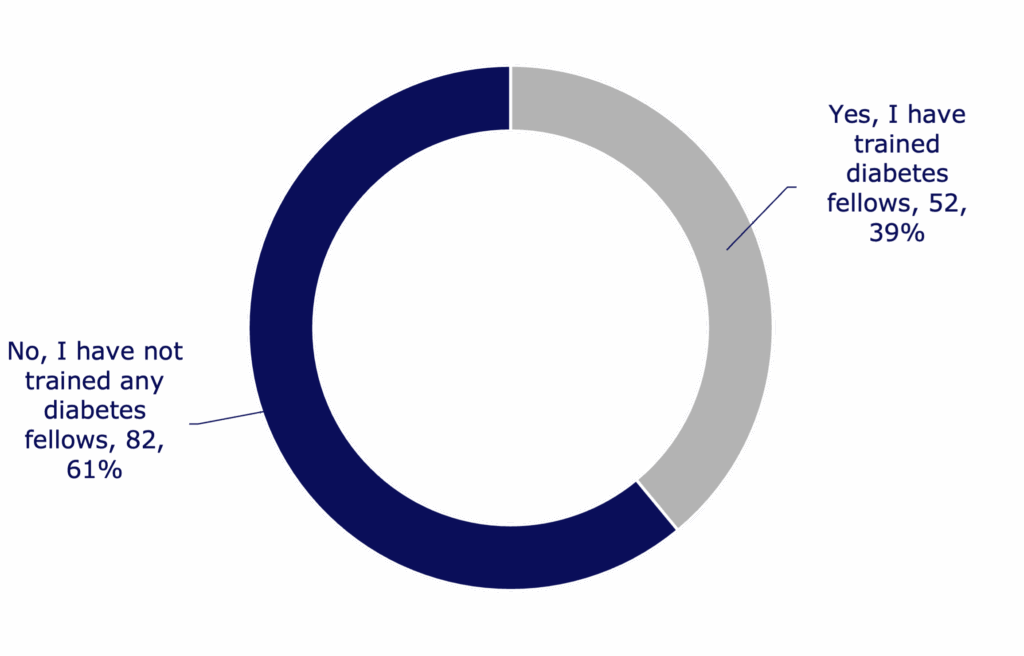
91
Total number of research fellows trained by respondents
84%
Percent of next-generation trainees in academic research or clinical practice
Essential findings: Research outcomes stemming from awards
- Respondents from every single type of training award report having trained a next-generation diabetes research fellow.
- Recipients of Career Development Awards published the most papers and formed the most collaborations.
- Recipients of Postdoctoral Fellowships were the least likely to form a collaboration, revealing a potential opportunity for Breakthrough T1D to help facilitate more collaborative connections amongst this awardee group.
- Awardees reported holding various professional roles in the diabetes community, including consultants to nonprofits and industry, conference planners, program leaders, and manuscript reviewers.
- The most prominent honor reported was a recognition or excellence award.
Impacts on securing additional funding
Breakthrough T1D training awards help early-career researchers continue an engaging and successful career in diabetes research. This is an important factor in securing further funding after the awards end, which helps researchers and clinicians maintain the ability to continue critical research that helps drive our mission forward.
78%
Percent of respondents who reported receiving additional research funding since the completion of the training award
$327,880,689
Dollar amount of additional research funding secured by respondents from various funding sources
Essential findings: Additional funds secured for research
- For each training award, recipients secured at least five times the training award amount in additional funding post-award.
- Postdoctoral fellows exhibited the greatest return on investment—meaning they secured the greatest amount of additional funding after the award ended.
Overall impacts on early career development

Essential findings: Impact on early career development as a whole
- Early-stage trainees prioritize the ability to test a promising research idea that others would not support.
- Later-stage trainees prioritize getting promotion or tenure and generating data to secure additional research funding.
Training awards are key to achieving our mission
Based on these data, it’s clear that Breakthrough T1D’s training awards impact early-career researchers and clinicians across the board: from career development, to training the next generation, to securing additional research funding, to publishing more papers.
This is exemplified by feedback from trainees. Some reported that Breakthrough T1D’s training awards are the best in their class and are critical for T1D research, including the ability to execute niche projects that other funding agencies would not support. Most respondents reported being immensely grateful for their training awards. See what some of the awardees have to say:

“Unequivocally, the award launched my career as a diabetes researcher.”
–Danielle Dean, Ph.D., Postdoctoral Fellowship Award recipient
“I am now fortunate to have mentees below me (one of whom received a Career Development Award). So not only did this award help me, but it helped establish an enduring source for new trainees interested in diabetes research.”
–Jeremy Pettus, M.D., Kellogg Family Early Career Patient-Oriented Diabetes Research Award recipient


“[I have made] International collaborations … research institutions and biotech companies to advance therapy development… partnerships across Europe, Asia, and the U.S.”
–Luis Arnes, Ph.D., Advanced Post Doctoral Fellowship and Transition Award recipient
Not only do these awards help retain talent in diabetes research, but they also help awardees maintain connections with Breakthrough T1D as a whole—outside of seeking financial support. Training award recipients have presented research updates at local events, participated in Breakthrough T1D fundraising activities, and supported our advocacy efforts. We are helping to build the next generation of T1D researchers, advocates, T1D community supporters, and more—all working toward the same goal of a world without T1D.
This survey has put into context where our money is going—and it’s going to the right places. Helping to train the next generation of scientists and clinicians is key to the success of our mission. We’re seeing significant returns on our investments, embodied by trainees securing substantial funding for ongoing T1D research, the creation of shareable resources that will help drive research forward and increase collaboration, and the success of our awardees in securing desirable jobs and leadership positions.
We put our time and money into identifying the best-and-brightest scientists and ideas, encourage them to stay in the field, and guide them as they pass the torch to the upcoming talented pool of young T1D researchers. We support scientists so they can do the important work that will help people with T1D live better lives—and maybe one day live without T1D. And, most importantly, we wouldn’t be able to do this without YOU: our supporters, our donors, and the entire T1D community that allows us to do what we do every day—improve the lives of people with T1D as we drive towards a cure. Thank you.
The full-length report was put together with the assistance of Breakthrough T1D consultant Kim Hunter-Schaedle.
There is always breaking, inspiring news in the world of type 1 diabetes (T1D) research—and that news was front and center at the 2025 European Association for the Study of Diabetes (EASD) Meeting.
This annual event is one of the largest diabetes conferences in the world, and Breakthrough T1D staff, partners, funded researchers, and more were there to participate, share new data, and stay current on the state of T1D and the path to cures.
At EASD 2025, we heard the best and the brightest in T1D present on every area of our Mission priorities, from advances in cell therapies to building consensus around general population screening for T1D.
Check out the highlights below!
Long-awaited updates on two disease-modifying therapies: ATG and verapamil
Excitingly, the results from the MELD-ATG and Ver-A-T1D clinical trials were presented at EASD, co-chaired by our very own Sanjoy Dutta, Ph.D., Chief Scientific Officer at Breakthrough T1D. Each of these studies were made possible through a collaboration between INNODIA, Breakthrough T1D, the Helmsley Charitable Trust, industry, the European Commission’s Innovative Medicines/Health Initiative (IMI/IHI), and investigators around the world. Each of these studies incorporated the INNODIA master protocol, an effort to align on clinical trial design and evaluation for investigational therapies for newly diagnosed T1D.
The phase 2 MELD-ATG study, presented by Chantal Mathieu, M.D., Ph.D., investigated whether a minimal low dose of anti-thymocyte globulin (ATG) could preserve beta cells in children, adolescents, and young adults (5-25 years old) newly diagnosed with clinical stage 3 T1D. ATG works by blocking the immune cells that destroy beta cells. Here’s a summary of the recently published data:
- Using an adaptive clinical trial design, the study examined multiple doses of ATG at once, eventually moving forward with 0.5 mg/kg and 2.5 mg/kg in comparison to placebo.
- The minimal effective low dose for ATG is 0.5 mg/kg. This dose had fewer side effects compared to the higher doses, and it was generally well-tolerated.
- Participants who received low dose ATG demonstrated clinically significant higher C-peptide levels during the treatment period compared to placebo. This was accompanied by lower HbA1c levels.
The phase 2 Ver-A-T1D study, presented by Thomas Pieber, M.D., investigated whether verapamil could preserve beta cells in adults (18-44 years old) with newly diagnosed stage 3 T1D. Verapamil is a blood pressure medication that may also reduce beta cell stress, and initial clinical studies suggest that it may protect beta cells in newly diagnosed T1D. The key findings presented are:
- At 12 months, there was no statistically meaningful difference in C-peptide levels between those who were treated with verapamil versus placebo. This was accompanied by no differences in insulin dose or continuous glucose monitor (CGM) metrics. There was a trend toward better C-peptide preservation in the treatment arm compared to placebo but missed statistical significance marginally.
- Verapamil was generally safe and well-tolerated, with no unexpected adverse events but some reported mild and reversible cardiac disorders.
- The study investigators concluded that future studies testing verapamil in T1D require a larger population and longer duration—and that verapamil may be a candidate to combine with immunomodulatory disease-modifying therapies.
- The Ver-A-Long extension study will follow people who continued with verapamil treatment after this trial ended.
Key takeaways
The horizon is bright for ATG—investigators have now identified the lowest dose needed to preserve beta cells in newly diagnosed children and young adults with T1D, with the fewest side effects possible. BT1D and the T1D Fund are supporting the development of a next generation ATG with a manageable/lower side effect profile compared to the medication used in the current trial. On the other hand, the future is open for verapamil—more studies are needed to determine if it can delay beta cell loss and T1D progression. A biotechnology company, TiXiMed, is developing a next-generation verapamil (TX-100), with a more favorable pharmacological profile.
Emerging consensus for screening the general population for T1D
Breakthrough T1D spotlight: Early detection and screening
Breakthrough T1D’s Vice President of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, spearheaded an effort to assemble a working group of nearly 30 experts to establish a consensus on T1D screening guidance. This session—which Dr. Albanese-O’Neill chaired—included presentations by some of these experts, who gave the audience a sneak peek on how to integrate population-level screening for T1D into clinical practice.
In this session, Marian Rewers, M.D., Ph.D., Anette Ziegler, M.D.,and Chantal Mathieu, M.D., Ph.D., provided an overview of soon-to-be-published consensus screening guidance for the general population. These presentations highlighted an urgent need for population-level screening given the rising global incidence of T1D and the known benefits of early detection, including time to prepare and potentially delay disease progression.

T1D autoantibodies
Autoantibodies are proteins that may signal that the body’s immune system is attacking insulin-producing cells in the pancreas, and they can easily be measured by a blood test. If a person has two or more persistent autoantibodies, it’s very likely they’ll develop T1D.
The presenters discussed benefits, harms, and methods of T1D screening; who should be screened and how often; and how to effectively communicate screening results. Some notable takeaways:
- Early detection of T1D—prior to clinical onset and at the time of diagnosis or later—has enormous benefits, including prevention of diabetic ketoacidosis, opportunities to delay progression with approved therapies or experimental therapies in clinical trials, early control of hyperglycemia that can reduce the risk of complications, and more.
- Providing support during the screening process will be important, such as mitigating pain associated with obtaining blood samples or reducing anxiety.
- Every initial screening result must be confirmed with another test before diagnosis.
- Infrastructure and policy must be in place before population-level screening can be implemented into clinical practice, including established processes for confirming positive test results, monitoring people with diagnosed early-stage T1D, and referring people to specialists.
- General population screening is suggested starting at 2-4 years old, and again at 6-8 years old and 10-15 years old if negative for autoantibodies.
- Healthcare providers (HCPs) need to be knowledgeable on key aspects of the screening process—and be able to effectively communicate what screening results mean.


Breakthrough T1D spotlight: Pediatric screening for T1D
To increase pediatric T1D screening in the UK, Breakthrough T1D is co-funding the ELSA (EarLy Surveillance of Autoimmune disease) study with Diabetes UK. At EASD, Parth Narendran, FRCP, Ph.D., presented how the data from this study was used to assess the acceptability and feasibility of general population screening for presymptomatic T1D in children who are 3-13 years old. The research team found that an initial finger-prick dried blood spot test—which can be performed at home or school and mailed to the team—was acceptable as an initial autoantibody screen, followed by a confirmatory blood test. Most families found out about this screening opportunity through social media, school, or their general practitioner—and none regretted participating, regardless of the outcome. This study will continue with an expanded population of children who are 2-17 years old.
Key takeaways
Consensus guidance from T1D experts suggests that population-level screening for T1D, starting at a young age, will be critically important for detecting T1D early so families have ample time to prepare and potentially delay disease progression. This consensus guidance will soon be published in a peer-reviewed journal, with the goal of integrating T1D screening into clinical practice. Breakthrough T1D is committed to expanding T1D early detection on a global scale.
Cell therapies are coming
Cell Therapies replace destroyed beta cells with external cells that make insulin and protect them so that they can function for a very long time. It’s one of our primary strategies to achieve cures for T1D—as exemplified by Project Accelerate Cell Therapies.
Our goal is to grow these cells in a laboratory and discover a way to keep them safe inside the body without the need for chronic immunosuppression. EASD provided several reminders that these therapies are not science fiction—they’re working in clinical trials, and we must continue to push these first-generation therapies—and future generations—across the finish line.
Vertex and zimislecel
Attendees at EASD were treated to another presentation demonstrating the efficacy of Vertex’s zimislecel, a manufactured islet cell replacement therapy. Michiel Nijhoff, M.D., presented some highlights from the FORWARD study, all of which were previously presented at other diabetes conferences and published in the New England Journal of Medicine [subscription required] this summer.
This chart does an excellent job illustrating the drastic improvement in glucose control in the year following transplantation of the cells. As does this stat: the 12 individuals in the first phase of the study went from 55.5% time in range and 36.1 units of insulin per day at the start of the study to 95% time in range and 0 units of insulin per day after one year.
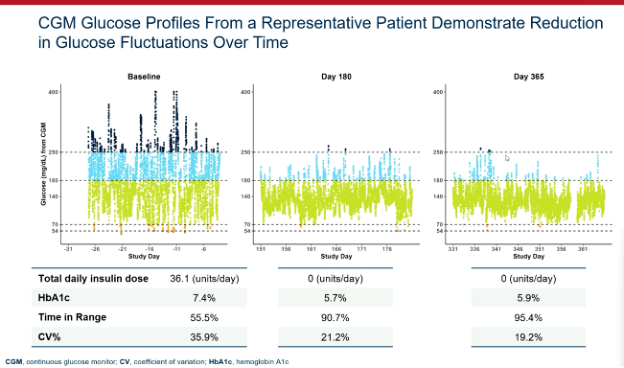
Breakthrough T1D’s support for Doug Melton, Ph.D.—whose proprietary lab-created beta cells are now being advanced by Vertex—goes back decades, both via research grants and an investment from the T1D Fund: A Breakthrough T1D Venture. We can’t wait to see more data when the Phase 1/2/3 trial is completed in the coming months.
Multiple strategies moving forward
Several other investigators shared data on their approaches to bringing scalable cell therapies to people with T1D. This includes:
- Matthis Hebrok, Ph.D., a longtime Breakthrough T1D collaborator, showed his research on hypoimmune, gene-edited islets. These cells are genetically engineered to avoid the immune system.
- Maria Nostro, Ph.D., another Breakthrough T1D-funded researcher, is using a different protection strategy: macrophages. Macrophages are a type of white blood cell that we have learned are present in the developing pancreas. The idea here is that we can implant the macrophages alongside the beta cells during islet transplant and it will help protect and vascularize the cells. This research is still pre-clinical but represents another “shot on goal” to keep these cells safe.
- Orizuru Therapeutics (a T1D Fund Company) presented on OZTx-410, an investigational cell therapy composed of human manufactured islets derived from adult cells that were reprogrammed to precursor cells and eventually islets. This is one of the first cell therapies for T1D derived from reprogrammed adult cells, and this therapy is suitable for manufacturing at large scale. Earlier this year, these cells were implanted into a person with T1D as multiple “sheets” in the abdominal wall. Orizuru expects to share results in the coming months.
- Allarta, a company with Breakthrough T1D funding, shared pre-clinical data on their hydrogel, which allows islet transplants without the use of immunosuppression. Their product is showing efficacy in animal models, and they are progressing towards human clinical studies.
Breakthrough T1D spotlight: Personal journeys and emerging T1D therapies
Breakthrough T1D’s Chief of Global Medical Affairs, Thomas Danne, M.D., Ph.D., hosted an interactive workshop for participants to better understand the personal journeys of people with T1D through videos from T1D community members and their families. Dr. Danne—along with other Breakthrough T1D staff and in conjunction with Eelco J.P. de Koning, M.D., Ph.D., and Valera Sordi, Ph.D.—fielded questions from the audience and covered topics spanning immunosuppression, how islets are derived from donor pancreases, T1D screening, and more.


Key takeaways
Cell therapies are real and advancing, with promising results in clinical trials, including dramatic improvements in glucose control. Also moving forward are ways to keep them safe and happy without the use of immunosuppressants.
Continuous ketone monitoring is coming – for good reason!
Diabetic ketoacidosis (DKA) is a serious and, unfortunately, too-common complication of life with T1D. This dangerous condition happens when the body does not have enough insulin. DKA is life-threatening and costly.
Breakthrough T1D-funded research Jennifer Sherr, M.D., Ph.D., presented this, outlining that this is a problem and that there is room for improvement in identifying DKA and agreeing on the interventions. We need better mitigation strategies, and if we get them, people will do better.

Breakthrough T1D Medical Affairs: CKM
Breakthrough T1D has prioritized the development of continuous ketone monitors, which we believe can be lifesaving. Our Medical Affairs team is working to build consensus around these and how they can be useful in the clinic and in the lives of people with T1D.
Key takeaways
Peter Bergstal gave a great summary, where he noted the CKM can be incredibly useful if we hit a few key checkpoints. These include developing an accurate CKM, receiving regulatory approval, finding the people most likely to benefit, and they’re covered by payers.
T1D Index 3.0 is here
The T1D Index—a computational tool launched in 2022 that measures the human, public health, and economic impact of T1D around the world—was developed by Breakthrough T1D in collaboration with Life for a Child, the International Diabetes Foundation (IDF), and the International Society for Pediatric and Adolescent Diabetes (ISPAD). Renza Scibilia, part of the Global Responsibility team at Breakthrough T1D, presented about exciting new upgrades for the T1D Index version 3.0. Some important updates:
- The newest version of the T1D Index has been updated with new published and unpublished data to ensure it remains the most comprehensive picture of T1D in the world.
- The new-and-improved dashboard allows for easier usability for researchers, healthcare professionals, and T1D community members to explore the data and computationally model the effects of care and diagnosis.
- The next version of the T1D Index in the works will further break down T1D statistics, focusing on regions and states within countries.
- The T1D Index is critically important for global advocacy and collaboration efforts by providing evidentiary data that support initiatives to change the trajectory of T1D.
9.5 million
The number of estimated people living with T1D across the globe.
1.5 million
The number of people at all ages living with T1D in the United States—the country with the highest prevalence of T1D.
Key takeaways
The T1D Index 3.0 is the most updated, accurate version yet, offering the best possible global coverage of T1D statistics and an enhanced dashboard to simulate changes in T1D incidence and prevalence.
The path to fully closed-loop automated insulin delivery systems
Hybrid closed-loop automated insulin delivery (AID) systems—a continuous glucose monitor (CGM) paired with an insulin pump, requiring mealtime and physical activity announcements—are estimated to be used by hundreds of thousands of people today. While these devices have been transformative for the T1D community, user input is still a daily burden, and fully closed-loop systems aren’t widely available for T1D yet. In this session, Moshe Phillip, M.D., Katrien Benhalima, M.D., Ph.D., and Charlotte Boughton, M.D., Ph.D., explored the benefits of currently available AID systems and fully closed-loop AID systems on the horizon.
- Based on clinical trial data and real-world evidence, hybrid closed-loop systems improve time-in-range (TIR) and reduce HbA1c levels without increasing the risk of hypoglycemia. This translates to better outcomes and quality of life for people with T1D.
- Different AID systems will work better for different people, depending on preferences for blood glucose management. Because there have been limited head-to-head trials comparing two or more AID systems, there is no evidence that any one system is definitively better than another.
- Right now, only one AID system is licensed for use in pregnancy (CamAPS FX). More research is needed to determine safety and efficacy of additional AID systems for use during pregnancy, and real-world evidence of women who become pregnant while using different AID systems may provide some clues.
- A clinical trial for the CamAPS HX fully closed-loop AID system in the U.K. increased TIR by 50% compared to 36% in people using standard pump therapy with a CGM—amounting to three additional hours each day of blood glucose in target range. Participants reported improved mood and sleep, less stress, and reduced diabetes burden. Similar improvements in blood glucose and daily life were reported in adolescents.
- Next-generation AID systems under investigation include two or more hormones for optimal glycemic control, artificial intelligence, and “digital twin” simulation tools.
Key takeaways
Fully closed-loop AID systems are coming, but they’re not here yet. Initial clinical studies using fully closed-loop systems in T1D demonstrate significant improvements in blood glucose control and quality of life. In the meantime, hybrid closed-loop systems offer substantial benefits for people with T1D—and there are many commercially available systems to choose from based on individual preferences.
Updates on heart and eye complications
Reducing T1D complications remains a major research focus area around the globe. Robert Humphrey presented on the LENS trial, which found that fenofibrate—a generic and affordable cholesterol-lowering medication—can reduce the progression of diabetic retinopathy in people with T1D and type 2 diabetes. A follow-up study found that fenofibrate is a cost-effective option for diabetic retinopathy in the U.K., especially for T1D. In conjunction with these studies, the recruiting Breakthrough T1D-funded Protocol AF trial is further investigating fenofibrate in preventing progression of diabetic retinopathy in people with T1D.
Rebecka Bergdal presented on a study examining the relationship between cumulative exposure to glycemia or lipids and heart failure, one of the leading causes of cardiovascular mortality in T1D. The study found that both cumulative glycemic exposure (defined as time spent with HbA1c levels > 7%) and cumulative lipid exposure (LDL, triglycerides, and cholesterol) are independently associated with increased risk for heart failure. The publication highlighting these findings includes a call to action for healthcare providers to help people with T1D minimize high blood sugar and lipid exposure, and support and encourage the best diabetes management possible to reduce risk for heart complications.
Key takeaways
Exciting clinical trials to prevent worsening of diabetic retinopathy in people with T1D are ongoing. To reduce the risk of heart complications, including heart failure, people with T1D and healthcare providers should aim for minimal exposure to high blood sugar and lipids, and more work is needed to find solutions for cardiovascular disease in T1D.

“We saw significant progress in each area of our Mission at EASD,” said Sanjoy Dutta, Ph.D., Chief Scientific Officer. “The MELD-ATG results are a noteworthy step forward for disease-modifying therapies; the screening consensus is a much-needed stake in ground around general population screening; we are moving closer to urgently needed adjunctive therapies; and cell therapies continue to perform in clinical and pre-clinical studies. It’s an incredibly exciting time in T1D and we will continue to support this work—and more—to bring cures to our community.”
The next big T1D meeting is ISPAD 2025, which will take place November 5-8 in Montreal, Canada. Stay tuned for more news!
This article was written by Sandy Vogt and Brian Herrick.

