Our Statement on Deceased Donor Islet Cell Availability & The ISLET Act
Curing type 1 diabetes (T1D) is Breakthrough T1D’s number one priority. As the leading global T1D research and advocacy organization, we are currently funding over $450 million in cures research in 23 countries. Everything we do is focused on making cures happen for the T1D community, sooner.
We are very excited to see the trials we fund having such positive results, including University of Chicago’s trial testing Eledon’s tegoprubart. This trial uses deceased donor islets, which come from the pancreases of deceased organ donors. We know we need more deceased donor islets available so more eligible adults can have access to islet cell transplants. To achieve this pressing goal, we have urged the Department of Health and Human Services (HHS) to reclassify deceased donor islet cells from biologics (drugs) to organs in the United States.
We are very grateful to the sponsors and supporters of the ISLET Act for their engagement on deceased donor islet cell transplantation and for helping to generate a discussion about increasing deceased donor islet cell availability.
Passing legislation is extremely difficult at best, and the ISLET Act does not require development of safety standards for reclassified deceased donor islet cell transplants that would ensure the islet cells are healthy and safe for transplant. Nor does it address insurance coverage, which could limit access to these costly treatments.
Reclassifying deceased donor islet cells as organs can be accomplished using existing HHS authority. New legislation is not needed. And so, with the full support of leading islet transplant surgeons, we’re putting 100% of our effort toward working with HHS, which has much better odds of success.
If implemented, our proposal to HHS would increase deceased donor islet availability, ensure rigorous safety standards, and address insurance coverage.
Continued research funded by Breakthrough T1D and others has shown deceased donor islet cell transplants to be safe and effective for adults with T1D who experience severe hypoglycemia and hypoglycemia unawareness. These procedures can significantly reduce—or even eliminate—the need to administer insulin.
With the science clear and patients waiting, Breakthrough T1D is calling on HHS to use its existing authority to safely expand availability of deceased donor islet cell transplants in the United States.
Deceased Donor Islet Cell Transplantation 101
Here’s what you need to know about deceased donor islet cell transplants—and our plan to expand availability to them safely and responsibly.
What Is Deceased Donor Islet Cell Transplantation?
Deceased donor islet cell transplantation is a procedure in which insulin-producing islet cells are isolated from the pancreases of deceased donors and transplanted into a person living with type 1 diabetes (T1D). These islets are infused into the liver, where they can begin producing insulin in response to blood glucose levels.
Deceased donor islet cell transplants are intended for, and only FDA-approved for, adults with T1D who experience:
- Severe hypoglycemia (dangerous low blood sugar)
- Hypoglycemia unawareness (the inability to sense when blood sugar is dropping)
These individuals often cannot safely manage their blood glucose levels with the standard therapies used to manage T1D, including continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems.
Do They Work?
Yes. For adults with T1D who experience severe hypoglycemia and hypoglycemia unawareness, deceased donor islet cell transplantation has been shown to significantly reduce—or even eliminate—the need to take external insulin.
Do Deceased Donor Islet Cell Transplants Require Immunosuppression?
Yes. Recipients of deceased donor islet cell transplants must take immunosuppressives to prevent the body from rejecting the transplanted cells. Due to the side effects of immunosuppressives, these transplants are only approved for adults whose risk from severe hypoglycemia outweighs the risks associated with immunosuppression.
Breakthrough T1D is working on alternatives to traditional immunosuppression so more people can benefit. This includes our funding of the University of Chicago’s clinical trial with Eledon’s drug, tegoprubart.
Why Aren’t They Widely Available?
Deceased donor islet cells come from organ donors. A single transplant often requires multiple pancreases as isolating islet cells is a complicated process. In addition to those challenges, the availability in the U.S. is more limited than it is in other countries due to regulatory classification.
The U.S. currently regulates deceased donor islet cells as biological drugs requiring a Biologics License Application (BLA). This is different from how whole organ transplants are regulated in the U.S. and how deceased donor islet cells are regulated in many other countries, including Canada, the United Kingdom and countries in the European Union.
Our Strategy to Increase Availability
How Is Breakthrough T1D Advocating for Expanded Deceased Donor Islet Cell Transplant Availability?
Breakthrough T1D has strongly urged HHS to reclassify and expand availability of deceased donor islet cells.
Our proposal outlines a framework to ensure safety and accessibility for the T1D community. You can read the letter here.
We are putting 100% of our energy behind this approach, and we are doing it with the full support of leading transplant surgeons and organizations.
Why Is Breakthrough T1D Pursuing This Route (HHS) as Opposed to Legislation (ISLET Act)?
This is our best chance of success. Passing legislation is extremely difficult at best, and HHS has existing authority over the regulatory framework governing cell therapies and organ transplantation. They can make this change.
Additionally, the ISLET Act does not require the development of safety standards for reclassified deceased donor islet cell transplants, nor does it address insurance coverage, which could limit access to these costly treatments.
What Are the Details of Breakthrough T1D’s Recommendations in the Letter to HHS?
Our proposal outlines a clear strategy to ensure the safety and quality of deceased donor islet cells under this reclassification. They include:
- Designating Centers of Excellence staffed with experts in deceased donor islet cell transplants
- Strict quality control to ensure islets are functional—and they work
- A way to get cells from the deceased donors’ pancreases and into people with T1D
- A strategic roadmap for adding additional centers
Why Not Work with HHS and Support the ISLET Act?
Our Breakthrough T1D legislative agenda includes several critical priorities essential to advancing our mission. This includes the ongoing renewal of the Special Diabetes Program (SDP) to fund T1D research and efforts to improve insulin affordability. These issues require legislative action, which is why we continue to focus our legislative advocacy on them.
Not every priority is best achieved through Congress. Reclassifying deceased donor islet cells as organs, for example, can be accomplished under current law through the Department of Health and Human Services (HHS), without new legislation. This is one of the key reasons we believe the path through HHS is the best path for this issue.
Simply put, we are pursuing each of our priorities through the channel where it has the greatest likelihood of success. And given how difficult it is to pass legislation, putting 100% of our effort toward working with HHS has the best chance of success.
Why Doesn’t Breakthrough T1D Recommend an Amendment to the ISLET Act?
Given how difficult it is to pass legislation, we believe our effort to reclassify deceased donor islet cells as organs is much more likely to succeed focusing on HHS.
What effect would this have on stem cell-derived islets?
Breakthrough T1D believes that stem cell-derived islets are appropriately regulated by FDA as biologic products. There is no need to change the regulatory status for manufactured islets. If adopted, our proposal would not affect them.
Through Project ACT, Breakthrough T1D is working with all parties to clear the way for future, manufactured islet therapies and to ensure they are accessible.
Breakthrough T1D’s Role in Islet Cell Therapies
Has Breakthrough T1D Supported This Work?
Yes—Breakthrough T1D has been a leader in islet cell transplantation for decades. Breakthrough T1D is currently funding over $450 million in cures research in 23 countries, including the University of Chicago trial testing Eledon’s drug, tegoprubart. Breakthrough T1D’s support goes beyond funding these clinical trials. The T1D Fund: A Breakthrough T1D Venture, invested in Eledon in 2023.
What Is Breakthrough T1D’s Strategy to Increase Access?
In 2024, Breakthrough T1D launched a flagship initiative, known as Project ACT (Accelerate Cell Therapies), designed to advance breakthroughs in T1D cell therapies, an anticipated pathway toward cures for T1D. Learn more about our work within research, medical affairs, and advocacy via the timeline at the bottom of our Project ACT landing page.
Is Breakthrough T1D Alone in These Efforts?
No. Breakthrough T1D is working with other T1D and diabetes organizations in the U.S. and around the globe, and top islet transplant surgeons. They support our efforts and agree that this is the best way forward.
How Can I Get Involved in This Work?
If you are in the United States, sign up to be a grassroots advocate here to get real time policy updates, action alerts, and tips for contacting your lawmakers.
If you live with type 1 diabetes, you can also be part of advancing treatments like this by participating in clinical trials. Visit our dedicated T1D clinical trial match tool here.
Breakthrough T1D had the pleasure of speaking with Patti Columbe, a participant in Vertex’s pivotal trial for the manufactured islet therapy zimislecel (formerly VX-880). Patti has been living with type 1 diabetes (T1D) for nearly 39 years. This is one of the most exciting T1D trials in terms of getting us closer to cures for T1D.
In this piece, we’ll talk about Patti’s T1D journey and what led to her successful enrollment in this landmark clinical trial.
The opinions, views, and experiences detailed in the interview below are those of Patti and are not representative of other participants’ experiences in this clinical trial, nor are they representative of Vertex Pharmaceuticals or Breakthrough T1D.
The conversation has been edited for length and clarity.
Q: Patti, can you tell us about your diabetes journey?
A: I was diagnosed with T1D in October 1986. I’ve lived with it for nearly 39 years. As anyone who knows this disease can tell you, it’s very hard. I’ve used every possible management system—pumps, continuous glucose monitors (CGMs), inhaled insulin, and I even have a diabetic alert dog named Otto. Despite these tools, I always struggled with low blood sugars, and I struggled with the mental burden of this disease. I was the kid at diabetes camp who had to get a glucagon injection in the middle of the night.

Q: What did you use to manage your T1D?
A: I’ve done everything—pumps, injections, CGMs, everything. But now I use multiple daily injections. I also rely on my diabetic alert dog—Otto! Otto, my service dog would often jump in my bed 20 minutes before a CGM would go off. And he consistently does that!
How did you hear about the clinical trial?
A: Having been around T1D for so long, I knew about the concept of islet transplants. I also knew that the end goal is making these cells. It was exciting but not something I thought was a reality today.
About two years ago, I heard about a clinical trial being done by Vertex. I found out about it on Instagram, actually. I then reached out to them and began the screening process.
Breakthrough T1D x Vertex Pharmaceuticals: Where it all began
Breakthrough T1D has played a role in the evolution of zimislecel for decades. In 2000, Breakthrough T1D awarded a grant to Douglas Melton, Ph.D., who developed a cutting-edge protocol to grow beta cells in a lab. Dr. Melton later founded Semma Therapeutics to turn these cells into curative therapies for T1D. In 2017, The T1D Fund made a catalytic investment in Semma, which was then acquired by Vertex in 2019—accelerating zimislecel’s development and leading to where we are today. We’ve been working closely with them ever since.
Q: What was the screening process like?
A: The screening was extremely thorough. I answered a ton of questions about my insulin usage, diet, blood type, history of hypoglycemia, and more. I even had to prove I had had T1D for all these years. They tested me for autoantibodies. They tested me for seemingly everything!
Q: How did it feel to make it into the trial?
A: Clinical trials like this are important. Clinical trials have to happen. We can’t get a cure without them, so somebody has to step up. I am lucky enough that I can step up—and I’m so glad to.
Participate in clinical trials
Clinical trials are an essential step in bringing new treatments and devices from labs to clinics. People who courageously choose to participate in clinical trials, like Patti, help drive progress for the entire T1D community. If you’re interested in participating in a clinical trial, use our Clinical Trials Matching Tool to find a trial near you. You can also connect with a Clinical Trial Education Volunteer in your area to answer any questions you may have.
Why this matters
Breakthrough T1D extends a heartfelt thank you to Patti for courageously sharing her story with us so we can truly understand why the work we’re doing is critically important. If you choose to participate in a clinical trial, like Patti, you have the potential to provide transformative benefits to the entire T1D community by accelerating the pace of clinical research. Learn more about clinical trials and how you can get involved. If you’re interested in the zimislecel clinical trial, check out the trial sites and eligibility criteria.
To accelerate T1D cures faster than ever, Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) is simultaneously advancing research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression. We will not stop until these therapies are available to everyone with T1D who wants them—and clinical trial participants like Patti are getting us there faster.
We are well on our way to seeing manufactured cell therapies like zimislecel come to life. We owe a huge thank you to our supporters and the broader T1D community for inspiring us every day to keep pushing forward until functional cures for T1D are a reality.
Disclaimer: Vertex Pharmaceuticals has donated to Breakthrough T1D.
This article was written by both Brian Herrick and Sandy Vogt.

The short version
Breakthrough T1D’s newest publication outlines what the future of beta cell replacement therapies looks like—and how we can make these therapies a reality for everyone with type 1 diabetes (T1D) who wants them through innovative clinical trial design and expanding the pool of eligible trial participants.
Breakthrough T1D, in collaboration with other leading experts in the field, recently published an article titled “Future Directions and Clinical Trial Considerations for Novel Islet β-Cell Replacement Therapies for Type 1 Diabetes” in the journal Diabetes. Breakthrough T1D Research and Advocacy staff who contributed to the publication include Sanjoy Dutta, Ph.D., Chief Scientific Officer, Esther Latres, Ph.D., Vice President of Research, and Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs.
Beta cell replacement therapies have the potential to cure type 1 diabetes (T1D) by removing the need for external insulin. While donor islet transplantation can result in insulin independence and results from early trials with manufactured islets are encouraging, people with T1D need better tools and therapies. This publication serves as a roadmap for the entire field—including researchers, product developers, industry, regulators, clinicians, and people with T1D—to address these needs and ensure that beta cell replacement therapies can get to people with T1D as quickly and safely as possible.
Read on to learn more about what the future of beta cell replacement therapies looks like.
Key Takeaways
- Currently available beta cell replacement therapies are limited to people with T1D with unstable blood sugar management, typically measured by HbA1c levels, in addition to dangerous lows (hypoglycemic events) that require immediate assistance. These therapies also require lifelong immunosuppression.
- New and improved beta cell replacement therapies are on the way, and we need to make sure that they are available for people with T1D beyond those with unstable blood sugar management and severe hypoglycemic events.
- Clinical trials for these therapies must be strategically designed to include more people with T1D given the potential for insulin independence.
- Shared decision-making between people with T1D and their care team will be critical for evaluating the risks versus benefits of a beta cell replacement therapy, especially as more options become available.
Where we are now
People living with T1D depend on external insulin to manage their condition throughout their lives. Despite advancements in diabetes technology, such as continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems, most people with T1D are unable to achieve blood sugar targets, rendering them at a higher risk for complications, reduced quality of life, and lower life expectancy.
It’s simple: we need to do more for the T1D community.
One promising avenue to meet these needs is through cell replacement therapy. Lantidra®, the first FDA-approved donor-derived islet replacement therapy for T1D, has proven to be safe and effective in eliminating severe hypoglycemia, providing insulin independence, and improving quality of life. However, this therapy is limited to people with severe hypoglycemia and requires immunosuppression to prevent rejection of the transplanted cells, which can have side effects. Even more, these donor-derived islets are in limited supply.
Alternative beta cell replacement therapies are emerging—and we can no longer limit their development to people experiencing severe hypoglycemia.
As stated in the publication:
Given the proven benefits of islet transplantation extending far beyond the amelioration of severe hypoglycemia that has been documented and the understanding of the risk profile gained over the past 20 years, consideration must be given to broadening the application for islet beta cell replacement.
Developing cell therapy strategies to meet unmet needs of the T1D population
Breakthrough T1D’s vision for the T1D community includes beta cell replacement therapies with no immunosuppression that are available to everyone who wants them. We are committed to making this a reality through our Project ACT (Accelerate Cell Therapies) initiative, which will accelerate the development of these therapies to achieve our vision as quickly as possible.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
We are entering an exciting era of beta cell replacement. Emerging therapies are addressing challenges such as cell source and scalability, resulting in the development of islets derived from sources other than donors, including manufactured islets and porcine islets. Up-and-coming therapies are also testing different transplantation sites, methods of delivery, and cell protection strategies to prevent immune rejection with the fewest side effects possible (and ideally no immunosuppression). Learn more about what scientists are doing to optimize beta cell replacement therapies.
Breakthrough T1D’s continuous support of many of these therapies, such as Vertex’s manufactured islet therapy zimislecel, has been critical to accelerating them through the clinical pipeline. Explore emerging beta cell replacement therapies in clinical trials now—and see how Breakthrough T1D’s commitment to these therapies helped make this possible.
When successful, the advent of new, safe, scalable, and effective beta cell replacement therapies will provide the T1D community with options. As these therapies are moving their way through the pipeline, we need to ensure they are being studied in a broader T1D population who stand to benefit.
So, how do we make this happen?
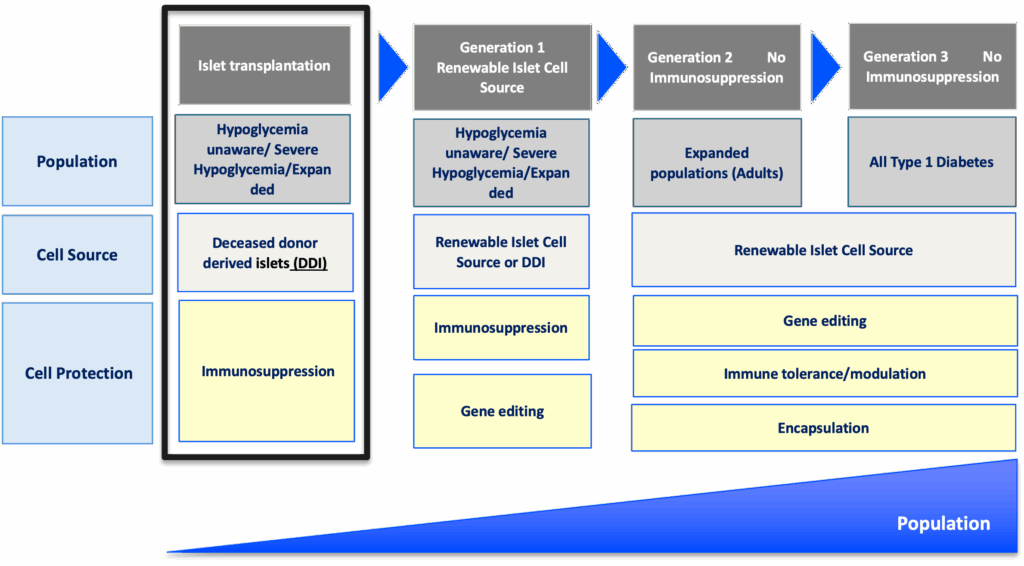
The roadmap for emerging beta cell replacement therapies
The goal
Accelerate availability of emerging next-generation beta cell replacement therapies for every person with T1D who wants them by designing clinical trials that speed their development, regulatory approval, access, and adoption.
Expanding the T1D population eligible for beta cell transplantation
Current trials testing beta cell therapies necessitating immunosuppression require participants to have elevated HbA1c levels and recurrent severe hypoglycemic events. This limits the pool of participants to people who meet the requirements and are most likely to benefit, given the side effects associated with chronic, broad immunosuppressants.
Clinical trials for emerging beta cell replacement therapies should broaden eligibility criteria so more people with T1D can participate—and experience the potential benefits. When designing new clinical trials, sponsors and regulators should consider including a broader range of HbA1c levels, clinically important or serious hypoglycemic events, and other complications.
Studies have found that the T1D community is generally open to beta cell replacement therapies as a potential solution to T1D, and people are willing to accept the associated risk versus benefit considerations for the possibility of becoming insulin independent. A Breakthrough T1D assessment also found that physicians are interested in recommending beta cell replacement therapies to people with T1D—especially if they don’t require immunosuppression.
“Clinical trials to support the development of islet cell replacement therapies need to evolve to include a broader representation of people living with T1D who could benefit from these novel therapies. This includes expanding the outcomes used to assess the benefits of cell replacement that reflect how people with T1D feel and function.”
Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs

Placing people with T1D at the center of clinical trial design
The outcomes used to assess the effectiveness of cell therapies currently in clinical trials, including those involving deceased donor islets, are acceptable for emerging beta cell replacement therapies. These include on-target HbA1c levels, absence of severe hypoglycemia, significant reduction or elimination of external insulin, and restoration of the body’s insulin production as measured by C-peptide.
Other endpoints that should be considered include CGM metric targets like time-in-range in addition to person-reported outcomes. Understanding how a beta cell therapy may affect a person’s health-related quality of life—such as diabetes distress, fear of hypoglycemia, or social and family dynamics—will be critically important for calculating the risk to benefit ratio of these therapies.
Read more about why person-reported outcomes are important for cell therapies.

“There are still significant unmet needs in the T1D community. Breakthrough T1D’s roadmap is supported by the assessment of clinically meaningful outcomes and driving research toward solutions that address key factors such as cell sources and protections strategies that will broaden the people with T1D who could benefit from emerging cell replacement therapies.”
Esther Latres, Ph.D., Vice President of Research
Innovative trial designs to accelerate development of cell therapies
Clinical trials for beta cell replacement therapies are generally based on a single-arm, open-label design—meaning there is no placebo group and both participants and researchers know which therapy is being administered. While this design can work for emerging beta cell therapies, single trials with multiple arms testing alternative transplant sites, immune protection strategies, or other methods have the potential to speed up the pace of development.
Similarly, adaptive trial designs use mid-trial interim analyses of study data to inform the remainder of the trial. This helps researchers learn what’s working (or not working) and adjust the design accordingly, with guidance from regulatory agencies, so the rest of the trial is a focused and efficient use of time and resources. Potential interim changes to trial design include reducing the number of participants required, eliminating doses, recruiting people who are most likely to benefit, or stopping the trial outright due to clear success or failure.
By applying guidance in therapeutic development and innovative trial designs to emerging beta cell replacement therapies, we can move early-stage trials along faster, thereby allowing regulators to make decisions sooner. To support quicker trials and reduce the possibility for delays, researchers, developers, and regulators around the world need to work together to achieve convergence on trial populations, endpoints, and innovative designs that will meet regional requirements.
Learning from past successes
People with T1D continue to live with unmet needs with still significant risk for long-term complications, and they need more therapeutic options. Right now, most clinical trials for beta cell replacement therapies requiring immunosuppression are limited to a small portion of the T1D population. This needs to change—especially given the potential for insulin independence. The T1D community must be put first when making decisions about beta cell replacement therapies, and Breakthrough T1D is making sure that this happens.
Adjusting how we approach clinical trials for emerging beta cell replacement therapies will be critical for ensuring we accelerate the research, development, regulatory approval, access and adoption of these novel therapies. Breakthrough T1D successfully accomplished this for AID systems—and we are confident that following a similar roadmap for cell therapies will get us to the finish line, faster.
The journey of AID systems
Learn more about the critical role of Breakthrough T1D in driving AID systems forward, recounted by Breakthrough T1D volunteer Doug Lowenstein.
Curative therapies for T1D are in reach. This roadmap, in conjunction with our Project ACT initiative, is key to bringing beta cell replacement therapies to every person with T1D who wants them.
To make this a reality, everyone needs to work together. As stated in the publication, “This requires a comprehensive strategy and a coordinated collaboration across stakeholders in every field relevant to islet cell replacement.” This roadmap is a guide for moving toward our common goal of a cure for T1D as soon as possible.
Breakthrough T1D will continue to lead the way until the T1D community can choose the beta cell replacement therapy that works best for them, regardless of blood glucose management or hypoglycemia status. Everyone deserves the chance to benefit.

Sana Biotechnology’s hypoimmune (HIP) donor-derived islets are genetically engineered to avoid immune destruction without the need for immunosuppressants.
Earlier this year, we reported on an update from the first person with type 1 diabetes (T1D) to receive a transplant of these cells, which showed that they were making insulin—without immunosuppression—after four weeks. Then, at the American Diabetes Association (ADA) 85th Scientific Sessions, we heard firsthand from Per-Ola Carlsson, M.D., Ph.D., the Principal Investigator at Uppsala University conducting the study, who provided more exciting data to a packed audience.
Now, these data have just been published in the prestigious New England Journal of Medicine in an article titled “Survival of Transplanted Allogenic Beta Cells with No Immunosuppression.” This study was supported by the Helmsley Charitable Trust, a long-time partner of Breakthrough T1D.
Read on to learn more.
Surviving and thriving
In this first-in-human study, the recipient of Sana’s HIP islets had no detectable insulin production at the time of the transplant (as measured by C-peptide). They received a small dose of islet cells—less than the eventual therapeutic dose—implanted into the forearm to evaluate safety.

C-peptide
C-peptide is an easily measurable biomarker of insulin production and islet function.
After 12 weeks, researchers found that the transplanted islets were successfully avoiding destruction by the immune system—meaning they are surviving AND making insulin!
The recipient had decreased HbA1c levels and stable insulin production between seven days and 12 weeks post-transplant, although, as expected with the small dose of cells used in this pilot study, they still required external insulin therapy. They experienced some non-serious side effects possibly related to the surgical transplant procedure, but not the islet cells themselves. Overall, the recipient is doing well and in good health—an exciting step for the T1D community.
Why this matters
To put it plainly: this is a huge step toward cures for T1D.
Immunosuppression is a major barrier to cell therapies because of the side effects associated with them. This is the first proof-of-concept evidence that we can genetically engineer islets to avoid immune destruction—eliminating the need for chronic, broad immunosuppressants. This means that more people may have the opportunity to benefit.
Importantly, the person in this study received only a fraction of the islet cells that would eventually be used therapeutically. Researchers are just beginning to understand the full potential of engineered HIP islets. Because more islet cells means more insulin production, there’s the possibility of insulin therapy independence if higher doses of HIP islets are implanted—something that will be addressed in future studies.
Thanks to this study, we’ve gained more evidence that forearm muscles may be a possible minimally invasive site for islet transplantation. This transplant site also provides researchers with the advantage of visualizing the islets—and making sure they’re alive—using magnetic resonance imaging (MRI), which isn’t possible with the current transplant site in the hepatic portal vein.
Excitingly, Sana is working to apply this technology to manufactured islets, opening the doors to producing HIP islets at large scale. They expect to begin phase 1 clinical trials for this next-generation HIP islet therapy as early as 2026.
Cell therapies without immunosuppression are a Breakthrough T1D priority
Cell replacement therapies—especially those that do not require immunosuppression—are a priority of Breakthrough T1D’s Cell Therapies Program and the goal of our Project ACT (Accelerate Cell Therapies) initiative.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
To accelerate the development of cell therapies that do not require immunosuppression, The T1D Fund: A Breakthrough T1D Venture invested in Sana to help advance their HIP technology platform. Breakthrough T1D continues to support creative and promising approaches that may eliminate the need for immunosuppression with cell replacement therapies, and we are hopeful and excited about what the future may hold.
This wouldn’t have been possible without supporters like you. Thank you for all that you do to drive Breakthrough T1D’s mission, and together we celebrate each step towards a world without T1D.
We’ve made major progress in the development of cell replacement therapies for type 1 diabetes (T1D) over the past few decades. We know that manufactured islets can be safely implanted into people and produce insulin. Yet, there is more work to do to advance cell therapy research and bring these therapies to the larger T1D community. Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) will make manufactured cell replacement therapies that do not require broad immunosuppression a reality, faster.
Project ACT
To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
The Breakthrough T1D x Stem Cell Network Partnership
To drive innovation in manufactured islet therapies, Breakthrough T1D, Breakthrough T1D Canada, and the Stem Cell Network (SCN) have partnered to support four new projects led by Canadian researchers. The organizations issued a joint Request for Applications and together will maximize resources to drive high-impact research into manufactured cells. This partnership is a novel and meaningful part of Breakthrough T1D’s global Project ACT effort to power high-impact cell therapies research.

“Accelerating cell therapies is a central focus of Breakthrough T1D’s research strategy to drive toward cures for type 1 diabetes,” said Breakthrough T1D Vice President of Research Esther Latres, Ph.D. “We’re excited to join Stem Cell Network and Breakthrough T1D Canada in funding these outstanding cell therapy researchers and projects that can build on the current momentum to overcome barriers and advance cell therapies that can benefit all those who live with type 1 diabetes.”
These projects will receive support from May 2025 to April 2027. They are a part of a broader SCN investment totaling more than $33 million to support 36 regenerative medicine research projects and clinical trials.
Read on to learn more about the exciting, newly funded projects.
A closer look at the projects
The project
Combining manufactured islets and vasculature for a better islet replacement product
Fueling Biotechnology Partnerships Award
The team
Dr. Tim Kieffer (UBC), Dr. James Shapiro (University of Alberta), Dr. Takanori Takebe (Cincinnati Children’s Hospital), & Lunar Therapeutics (Vancouver, BC)
Current cell therapies for T1D, while often effective, are hampered by reliance upon donor-derived cells and poor cell survival after transplant, necessitating large doses of cells and repeat procedures. This ambitious new project will address both the source of islet cells and the low cell survival rates associated with islet transplantation by accelerating Lunar Therapeutics’ preclinical development of a manufactured islet replacement product. Takebe’s lab describes it as “complex miniature organs” for T1D.
This product will consist not only of insulin-producing cells, but also endothelial cells, which line blood vessels. Endothelial cells will support islet cell survival and engraftment upon transplantation.

Organoid
An organoid is a three-dimensional tissue grown in the lab that resembles an organ.
To accomplish this objective, Lunar Therapeutics will bring together Canadian expertise in manufactured islets and clinical islet transplantation led by Drs. Timothy Kieffer and James Shapiro. The team will also include U.S.-based Dr. Takanori Takebe, who specializes in designing complex organoids composed of various cell types. Using technologies developed across each laboratory, this multidisciplinary team will work to address challenges in islet cell transplantation.
The project
Using naturally-derived gels to optimize cryopreservation (extreme cold storage) of manufactured islets
Impact Award
The team
Dr. Marya Ahmed & Dr. James Shapiro (University of Alberta)
The implantation of manufactured islets into people with T1D can restore insulin production, eliminating the for external insulin and improving quality of life. However, after islet cells are derived from donors or manufactured in the lab, they must be stored before being used to treat a person with T1D. Currently, the storage and transportation of islet cells is difficult, and the only storage method is freezing at low temperatures in the presence of chemical solutions that help with the freezing process. However, these solutions cause cell death during thawing and may also cause allergic reactions in people after transplantation.
This project will address this gap in the field by aiming to develop non-toxic, naturally derived gels to optimize islet freezing and storage. The gel-based products will be evaluated for large-scale commercial production. The success of this project will provide new intellectual property that will be of interest to researchers and companies in regenerative medicine in Canada and across the globe.
The project
Using blood vessels to create a better encapsulation device for islet replacement therapies
Impact Award
The team
Dr. Corinne Hoesli (McGill), Dr. André Bégin-Drolet (Laval), Dr. Richard Leask (McGill), Dr. Andras Nagy (Sinai Health, Toronto), Dr. Steven Paraskevas (McGill)
Manufactured islets offer a potentially unlimited source of islets for transplantation. Since manufactured islets carry unique risks compared to donor-derived islets, containment within a device could allow retrieval if off-target growth ever occurs. However, encapsulation devices that have been tested in clinical trials so far and have shown minimal success, mainly because blood supply to the cells is limited by the device barrier. In this project, the team proposes to develop a device where the manufactured islets are placed around pre-established vessels that can improve islet cell survival and speed of insulin responses via improved blood supply. In this project, they will optimize their device design and conduct advanced preclinical studies.
This project could lead to better survival and function of manufactured islets, so they can keep producing insulin. The project may also pave the way for other engineered human-scale encapsulation devices, also sometimes called bioartificial organs.
The project
Using manufactured cells to create a human T1D immune system model in a petri dish
Impact Award
The team
Dr. Megan Levings, Dr. Bruce Verchere, Dr. Francis Lynn & Dr. Peter Zandstra (UBC)
There are many new treatments on the horizon for T1D, including those that block autoimmunity or replace insulin-producing cells. However, a major barrier to these therapies is the lack of an easy-to-use model in which their effects on human cells can be tested before advancing to human trials. The standard preclinical model is to test therapies in small animal models of T1D, but this has significant limitations since it is nearly impossible to replicate the human immune system. In fact, diabetes has been “cured” hundreds of times in a mouse model, which has not translated to humans.
To overcome this barrier, Dr. Levings and her team will establish a new a model that recreates human T1D autoimmunity in the lab. The model will use manufactured cells to create the three types of cells that are involved in the disease: insulin-producing cells and two different types of immune cells. Using the model, cells can then be combined in different ways to recreate what usually happens during autoimmunity.
A model of human T1D that can be generated in the lab will help test potential treatments and prompt new questions about why T1D develops, and how to prevent it. Thus, this research has the potential to support the further development of innovative therapies that may offer new approaches to prevent or treat people with T1D.
Putting it all together
Curing T1D is the north star of Breakthrough T1D. These partnerships will help us work together toward our shared goal of a world without T1D—through innovation, forward-thinking cell therapy research, and the best and brightest scientists.

Project ACT series
This article is the last part of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) is shaping the future of cell therapies for type 1 diabetes (T1D).
Read last month’s article about the role of Advocacy in cell therapies for T1D.
Cell therapies, which place insulin-producing cells back into the bodies of people with T1D, are making major headway in clinical trials—and people are becoming insulin independent. In past installments of this article series, we’ve explored how we got to this point at every step in the pipeline: from advocating for funding, to conducting research in laboratories, to clinical testing in humans, and to working with regulators and insurers.
But we’re not at the finish line yet. Approval of manufactured cell therapies isn’t enough to make sure that people who want these cells can get them. So, how do we fill this gap?
Medical Affairs is the backbone of clinical adoption
The answer is Medical Affairs—Breakthrough T1D’s newest mission pillar—which aims to support the clinical adoption of manufactured cell therapies. As we are soon approaching the regulatory submission of a first-generation manufactured cell therapy, Medical Affairs initiatives will be especially important in making sure that healthcare professionals (HCPs) have the tools they need to bring these therapies to clinics. By partnering with global experts and HCPs to increase knowledge and clinical competencies, the Medical Affairs team is working to ensure the future delivery of manufactured cell therapies to eligible people with T1D.
Preparation is key
Since manufactured cell therapies for T1D are not yet FDA-approved, we are at a critical moment where preparation is essential. This is why the Medical Affairs team is planning now for the eventual arrival of first-generation manufactured cell therapies. They are partnering with advanced clinical teams to determine best practices to ensure that clinics have the right resources and skillsets. They are anticipating obstacles—and finding solutions—to the adoption of manufactured cell therapies. Then, when these therapies become available, HCPs will be ready and waiting to deliver them to people with T1D.
Assembling expert clinical teams and facilities
For people with T1D to have access to manufactured cell therapies, comprehensive expert clinical care teams need to be at the ready. To make this possible, we are starting at the very beginning: in training programs.
New, updated programs and fellowships must incorporate specialized education around cell therapies (such as optimized immunosuppression regimens and pre- and post-transplant education) for the next generation of HCPs. At a time when there are too few endocrinologists and certified diabetes care and education specialists, there’s no better time than now to encourage trainees to enter the field. We need to ensure that T1D care teams have trained professionals who are up to pace with the newest cell therapy protocols.
In line with these efforts, the Medical Affairs team is also working to recognize expert multidisciplinary Centers of Reference, which are a network of T1D treatment centers that have the personnel, resources, facilities, and skills to deliver manufactured cell therapies to people with T1D with a consistent approach. These centers will act as training hubs and share outcomes to continuously improve and establish best practices as manufactured cell therapies are incorporated into clinics.
Guidelines, statements, workshops, and more
The Medical Affairs team is working with experts in the field on publications that will guide care teams to further support the standardized integration of manufactured cell therapies into clinical practice.

Clinical guidelines
Clinical guidelines are recommendations for clinicians about how to care for people with T1D. They are informed by a systemic review of evidence by experts to assess benefits versus risk of care options.

Consensus statements
Consensus statements are collective opinions of an expert panel representing the multidisciplinary care team and voice of people with T1D. These statements are developed when evidence is still being gathered.
To drive these efforts, the Medical Affairs team hosts workshops, convening international experts to collaborate and discuss ideas. Using established methodologies, these projects ensure that the final document reflects expert consensus. One of the latest workshops assembled islet cell transplantation professionals from around the world to begin developing a consensus on the clinical roadmap of cell replacement therapies—the first step of many toward clinical adoption.
Read more about the most recent Medical Affairs workshops, including one geared toward creating Centers of Reference. See a recent example of clinical guidelines for monitoring early-stage T1D.
Opportunities to learn
Through decades of research, we’ve learned a lot about T1D—and the newest therapies and treatments are as innovative and complex as ever. To keep up with the rapidly changing pace of innovation, the Medical Affairs team has put together opportunities for HCPs learn about cutting-edge advancements.
These HCP resources offer accredited, free, live or on-demand education around the latest T1D research. There is also information on upcoming conferences, published guidelines, and clinical trial opportunities, in addition to an email newsletter with regular updates. Accredited HCP education is essential to ensure that clinicians have the most up-to-date knowledge that they can share with the T1D community—an important step in clinical adoption. Cell therapy resources are currently under development and expected in the next year.
Clinical trial education and awareness
Clinical trials are a key step in ensuring that new and effective T1D treatments and cures, including manufactured cell therapies, can safely make their way to the T1D community. The Medical Affairs team is working to foster a culture of research participation by ensuring that HCPs have access to information on current clinical trials and can help to educate the T1D community on their importance.
The ultimate goal is to increase awareness about clinical trial opportunities and support shared decision-making between people with T1D and their diabetes care team about clinical trial participation. The bottom line? HCPs and the T1D community must be equipped with information to enable them to make informed choices about participating in cell therapy clinical trials.

Medical Affairs help turn groundbreaking science into real care—by supporting doctors, educating communities, and making sure cell therapies for type 1 diabetes reach the people who need them most.”
The bigger picture: Project ACT across the pipeline
In this series, we’ve explored how manufactured cell therapies are moving through every step of the pipeline. We took a deep dive into the challenges that researchers are addressing to optimize manufactured cells. We surveyed the different cell therapies that are in currently clinical testing. We reviewed how advocacy shapes funding, regulatory decisions, and access to cell therapies. Finally, we considered how to ensure the clinical adoption of manufactured cell therapies.
This is where Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) comes in. The ultimate goal is to accelerate manufactured cell therapies that do not require immunosuppression—all the way from the first discovery research in the lab to the adoption of cell therapies in clinics.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
At every step of the way, we are moving cell therapies through the pipeline as fast as possible. Breakthrough T1D is preparing for the day when first generation manufactured cell therapies are available, and we are working toward a future with next generations of therapies that do not require immunosuppression. Thanks to concerted efforts by Breakthrough T1D’s Research, Advocacy, and Medical Affairs teams, each day we move closer to manufactured cell therapies becoming a reality for everyone with T1D.
Key Takeaways
- Breakthrough T1D helped organize a meeting in European Parliament to bring the unmet needs of the type 1 diabetes (T1D) community to the forefront and discuss how we can accelerate cures, especially cell therapies, in the EU.
- This meeting was attended by several Breakthrough T1D leadership and staff.
- Building long-term partnerships with the European Union (EU) Institutions will allow us to work together toward global T1D cures.
A meeting of the minds

This past week, Breakthrough T1D helped organize an event hosted by Member of European Parliament Tomislav Sokol, Ph.D., titled “Accelerating Breakthroughs to Address Unmet Needs in Type 1 Diabetes.” This meeting, a significant coming-together of Breakthrough T1D and European policymakers, focused on the role of the EU in addressing the needs of the T1D community and accelerating T1D breakthroughs.
The purpose of this meeting was to raise awareness of T1D and the urgent need for the accelerated development and approval of breakthrough therapies in the EU. Conversations between Breakthrough T1D and European policymakers homed in on barriers and opportunities to advancing cures—including cell therapies and disease-modifying therapies—in the EU to get them into the hands of people with T1D, faster. This was an important step in establishing an open dialogue between Breakthrough T1D and the EU Institutions about working together to address T1D globally.
“This event in the EU Parliament allows us to engage with important decision-makers to ensure that the research and policy environments are oriented in a way to accelerate development of T1D breakthrough therapies in the EU as we also do in other countries,” explained Campbell Hutton, Senior Vice President of Global Advocacy at Breakthrough T1D.
Attendees
Several Breakthrough T1D leadership and staff members attended the meeting, including Thomas Danne, M.D., Ph.D., Chief Medical Officer, Global; Sanjoy Dutta, Ph.D., Chief Scientific Officer; Lynn Starr, Chief Global Advocacy Officer, Carmen Hurtado del Pozo, Director, European Research; and Campbell Hutton, Senior Vice President of Global Advocacy.
Several Members of the European Parliament (MEP) in addition to host Tomislav Sokol, Ph.D., were in attendance. Other attendees included people with a lived experience of T1D, health staff from EU Member States, researchers in the EU, and other European diabetes organizations.
Dr. Dutta delivered a talk on the role of breakthrough therapies in transforming T1D. Dr. Danne moderated a panel to provide insight about unlocking the potential of cell therapies breakthroughs in the EU with T1D cell therapy researchers: Professor Lorenzo Piemonti, M.D., Director of the Diabetes Research Institute at Vita-Salute San Raffaele University and Associate Professor Francoise Carlotti, Ph.D., Head of the Islet Research Lab at Leiden University Medical Center. Finally, Lynn Starr closed with remarks about our shared global responsibility to work toward breakthrough T1D therapies.

When and where it took place
The event took place on June 5, 2025, in Brussels, Belgium, at the European Parliament.
Driving toward T1D cures in the EU and beyond
T1D is on the rise around the world, including in Europe. Recent publications by Breakthrough T1D staff and leadership brought attention to the rise in incidence and global T1D burden. We need to act now in conjunction with governments around the world—like the EU—to address the unmet needs of everyone around the world affected by T1D.

T1D cures, including cell therapies, are advancing through the clinical pipeline. Bringing awareness of T1D to the forefront—and educating key people on the progress we’re making toward cures—will be incredibly important for driving T1D research forward in the EU. This represents a critical opportunity for the EU to accelerate cell therapies faster than ever. As a global organization, Breakthrough T1D is collaborating with the EU government to help make this possible.
As the largest global funder of T1D research, Breakthrough T1D has provided funding to researchers across the world in addition to the EU. Right now, Breakthrough T1D is supporting €56 million in European initiatives, including 31 clinical trials—representing 19% of our funded research (including the U.K.). Breakthrough T1D has expert teams in research, medical, regulatory, and advocacy in Europe, meaning we are uniquely positioned to provide guidance on how the EU can strengthen its T1D efforts and collaborate on a global scale to drive T1D breakthroughs, especially in cell therapies.

Type 1 diabetes is a critical disease in Europe, and I was pleased to host an event for the T1D community and my colleagues in the European Parliament to learn about the unmet needs in T1D and how we can work together to accelerate breakthrough therapies in Europe to address those needs.”
What Breakthrough T1D leadership is saying

“This meeting is critically important to bringing the unmet needs of the T1D community into the spotlight in the EU. Global advocacy for curative T1D research is essential to achieving Breakthrough T1D’s mission, and continued collaboration with EU policymakers will get us there faster.”
Lynn Starr
Chief Global Advocacy Officer
“International efforts to accelerate global adoption of T1D cures will become increasingly important as newer, emerging cell therapies become available to people with T1D. Conversations like these with EU policymakers is bringing this urgent need to the forefront.”
Thomas Danne, M.D., Ph.D.
Chief Medical Officer, Global


“Cell therapies are accelerating through the clinical pipeline faster than ever. We need to act now on a global scale to ensure that people with T1D around the world can access these transformative therapies. This meeting is a significant step toward that goal.”
Sanjoy Dutta, Ph.D.
Chief Scientific Officer
Accelerating global action is paramount to our mission

Global problems require global solutions. This meeting served as a critical launching point for a continued partnership with the EU to fill gaps and address unmet needs for the T1D community. Building relationships and fostering long-term partnerships is critically important for reaching our common goal of bringing cures to people with T1D as soon as possible.
These important conversations between Breakthrough T1D and the EU government align with our Project ACT (Accelerate Cell Therapies) initiative to accelerate the development of cell therapies that do not require immunosuppression—for everyone with T1D in every country. In addition, through international Centers of Reference, Breakthrough T1D’s Medical Affairs team is developing expert clinical care centers that will be trained and ready to provide cell therapies to people with T1D once they become available. We are at the forefront of global action to prepare the world for curative cell therapies.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
We are driving toward a future in which everyone with T1D—no matter where they are—has access to therapies, treatments, and care, bringing us closer to achieving our mission of a world without T1D. The more people we have working toward our mission, the faster we will get there.

Project ACT series
This article is part of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) is shaping the future of cell therapies for type 1 diabetes (T1D). The next and final article in the series will focus on how Breakthrough T1D’s Medical Affairs team is working to bridge the gap between access to and adoption of cell therapies.
Read last month’s article about cell therapies for type 1 diabetes in clinical trials.
Right now, promising new manufactured cell therapies for type 1 diabetes are making their way to and through the clinical pipeline. Testing cell therapies in clinical trials helps us understand if they are safe and effective, and the data from these trials is ultimately used in decisions about whether a therapy should be approved and covered by insurers. From basic research to clinical trials, and from regulatory approval to access in the clinic, bringing cell replacement therapies to people with T1D as quickly as possible requires consistent advocacy and clear, thoughtful, and efficient policies from beginning to end.
Advocacy takes teamwork
This is where Breakthrough T1D’s Advocacy team comes in. They work on both sides of product approval—before and after—advocating for funding and policies that support research, development, approval, and access. This includes educating key players on the latest T1D science, modernizing and expanding approaches to manufactured cell therapy clinical trials, and working with payers to ensure access through insurance.
The ultimate goal is to work together—regulators, researchers, clinicians, policymakers, people with T1D, insurers, and industry—to accelerate the development and approval of manufactured cell replacement therapies, especially those that do not require broad immunosuppression. Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) will make this a reality faster.
Read on to learn about the key areas towards which Breakthrough T1D’s Advocacy team is working to advance manufactured cell replacement therapies.
Advocating for funding and policies supporting cell therapy research
To ensure that there is government funding to support T1D research—including manufactured cell therapies—Breakthrough T1D’s Advocacy team regularly engages with policymakers in Washington, D.C. Importantly, we advocate for continuous renewal of the Special Diabetes Program (SDP), a federally funded initiative that contributes to T1D research at the National Institutes of Health (NIH). This funding is a critical accelerator of manufactured cell therapies research.
Special Diabetes Program
The SDP expires in September 2025. Sign up to be an advocate today and help us garner support for this incredible program.
Through other initiatives, such as Government Day and Children’s Congress, we convene advocates of all ages in Washington, D.C. to share the impact of life with T1D and research updates with Members of Congress. This work includes hundreds of meetings with Members of Congress and their staff each year and engagement with the Diabetes Caucus in the House and Senate. These lawmakers play major roles in making decisions about government policies and funding. Engaging with them about the realities of T1D and the importance of research into potential cures like manufactured cell therapies is essential to ensuring the continuity of government funding that keeps this critical research going.
Ensuring that manufactured cells are safe—and that there are enough for everyone
Before manufactured cell therapies can be put into people, researchers use animal models to study the safety of a new therapy and determine whether it has the potential to work in humans. This can be a time-consuming and costly process. Breakthrough T1D is developing a roadmap for researchers, encouraging focus on the most critical challenges for manufactured cell therapies. The goal is to expedite these studies and get manufactured cells into people faster.
Another critical step is how the cells are made and making sure there are enough manufactured cells for everyone. The challenge is finding the best way to manufacture islets in large quantities while keeping them safe and functional. Breakthrough T1D is partnering with the best and brightest scientists at the Advanced Regenerative Medicine Institute and the Cedars-Sinai Biomanufacturing Institute to make this possible.
Understanding risks and benefits from the perspectives of people with T1D
For decision-makers—which includes regulators that approve products, health insurers, and policymakers—to make the best recommendations possible about manufactured cell therapies, they must consider the perspectives of people living with T1D. Importantly, this includes what risks someone with T1D is willing to accept for the possibility of curing T1D. This is especially important considering the significant unmet needs that remain for the T1D community, which are not always well understood by policymakers who are removed from the daily realities of living with T1D.
Unmet needs and demographics of the T1D community
Breakthrough T1D published two papers in recent months that fully illuminate the true scale of type 1 diabetes (T1D) in America, the unmet needs of the global T1D community, and the barriers we must overcome to achieve our vision of a world without T1D.
As a new kind of therapy, the way we think about the clinical studies that test cell replacement therapies needs to change. Clinical trials up to this point have only included a small subset of the T1D population, but as we have gained more experience with these therapies, we’ve learned that the trials should include as many people with T1D who could benefit as possible. This is especially important given the great potential of manufactured cell therapies to cure T1D. Further, how we measure benefits in clinical trials needs to expand to better consider the perspectives of people who will receive these treatments.
Breakthrough T1D is spearheading a Patient Preference Study to better understand and quantify how people living with T1D consider tradeoffs between risks and benefits when deciding whether to receive a manufactured cell transplant. This involves gathering perspectives from both people with T1D and their caregivers to understand how they view different aspects of cell replacement therapies that may influence their willingness to try it. The results, expected in fiscal year 2026, can be shared with regulators and policymakers to incorporate into their decisions regarding manufactured cell therapies for type 1 diabetes.
Educating insurers and advocating for access
Once a manufactured cell therapy becomes approved, the next challenge to tackle is access through insurance. To achieve our vision, people must have affordable insurance coverage of manufactured cell therapies, and ideally people should be able to choose between multiple cell therapy options. Our Advocacy team is already engaging with insurers and government payers to make them aware of manufactured cell therapy research progress for T1D. We are working toward establishing the pathway to coverage for these therapies.
To support these efforts, Breakthrough T1D uses data-driven approaches. For example, a recent study determined key demographics and the health insurance status of the T1D population. Our Advocacy team is using this data to better understand the population that can benefit from manufactured cell therapies. Through continued data collection and analyses, we can further advance the pathway to coverage for manufactured cell therapies.
Advancing progress through Project ACT
Breakthrough T1D is working with stakeholders across the entire T1D spectrum—from researchers, to regulators, to payers, to clinicians, to people with T1D—to ensure that progress is moving along quickly and efficiently for new cell therapies for type 1 diabetes. These efforts are based on a model that has already proven successful, leading to the development and adoption of Artificial Pancreas systems.
With the launch of Project ACT, every day we move closer to our goal of manufactured cell therapies becoming a reality for everyone with T1D.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Breakthrough T1D’s Advocacy team regularly engages in conversations with decision-makers who play a major role in influencing when, and if, manufactured cell therapies will become available to people with T1D. Breakthrough T1D is bringing the T1D community into the conversation, changing the way we think about clinical trials, and opening the doors of manufactured cell therapies to as many people with T1D as possible.
More manufactured cells, faster clinical trials, accessible therapies—we are identifying challenges, working together to tackle them, and accelerating manufactured cell therapies for type 1 diabetes faster than ever.
Breakthrough T1D’s newest mission pillar, Medical Affairs, is bridging the gap between access to and adoption of T1D therapies. The establishment of this program is essential to Project ACT (Accelerate Cell Therapies): Breakthrough T1D’s initiative to accelerate the development of manufactured islet cell replacement therapies that do not require immunosuppression. The goal is to make sure that people with type 1 diabetes (T1D) can get these therapies as soon as they hit the market.
The field is moving quickly: people are becoming insulin-independent in cell therapy clinical trials. We are advancing towards the submission of the first-generation manufactured islet cell therapy that requires immunosuppression, Vertex’s zimislecel (VX-880). We are at a critical moment and need to act now to ensure that healthcare providers (HCPs) are ready to bring manufactured islet cell therapies into clinical settings.
Enter Medical Affairs
This is where Medical Affairs comes in. The team, led by Thomas Danne, M.D., Chief Medical Officer International, is working with the medical community to anticipate obstacles to getting manufactured islet therapies into clinics and find ways to overcome them now.
To accomplish this, Breakthrough T1D recently hosted two cell therapy workshops, convening multidisciplinary, international experts in islet cell transplantation to discuss a clinical roadmap for manufactured islet cell therapies—and how to ensure that clinical teams are in place and prepared to provide these therapies to people with T1D who qualify. By preparing now, we can get this first-generation therapy into the hands of people with T1D as soon as possible after regulatory approval.
The cell therapy workshops were hosted by Breakthrough T1D’s Thomas Danne, M.D., Chief Medical Officer International, and Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, Vice President of Medical Affairs.
Read on to learn more about the cell therapy workshops, the attendees, and what each accomplished.
Workshop #1: Who may benefit most from manufactured cell replacement therapies
Key Takeaways
- T1D leaders are working on a five-year roadmap for the clinical adoption of manufactured islet cell therapies.
- It will include key criteria and evidence to help clinicians determine who is the best fit for manufactured islet cell therapies.
- The guidelines are prioritizing shared decisions between people with T1D and their care team to optimize outcomes and maximize long-term health.
Attendees
The first of the cell therapy workshops, held in late April, convened transplant surgeons, T1D clinicians and researchers, a member of Vertex’s leadership team, a member of Breakthrough T1D’s Participant Advisory Council to represent people with a lived experience of T1D, and Breakthrough T1D Vice President of Research Esther Latres, Ph.D.
The purpose of this workshop was to start developing a five-year roadmap to help guide the T1D care community to support the adoption of manufactured islet cell therapies in clinical care. The evidence-based recommendations will be vetted by a larger group of clinical experts, diabetes organizations, and people with lived experience with T1D to ensure there is broad agreement. The consensus document will ultimately be published to expand its reach.
This process will summarize the essential evidence that will help HCPs decide who may benefit the most from manufactured islet cell replacement therapies. These decisions will take into account the perspective of people with T1D and differences in age, hypoglycemia unawareness, or kidney health, to name a few. This can help HCPs better understand the benefits versus risks for manufactured cell therapies on a person-by-person basis—making sure that each clinical decision is made jointly to prioritize long-term health.
Workshop #2: Pilot workshop to develop international Centers of Reference for T1D cell therapy
Key Takeaways
- Centers of Reference are expert T1D care centers that are preparing to bring manufactured islet cell therapies to the T1D community.
- This workshop was the first step to understanding what kind of education, training, and resources are needed for Centers of Reference to be effective.
- Attendees discussed how to make sure people T1D who receive manufactured cell therapies at Centers of Reference will have the best clinical experience possible.
Attendees
The second cell therapy workshop, held in early May, convened clinicians from various global medical institutions, including University of Minnesota Medical Center, University of Wisconsin Health Transplant Center, the Penn Rodebaugh Diabetes Center, University of Chicago Medicine, IRCCS Ospedale San Raffaele (Italy), Institute of Transplantation, Newcastle upon Tyne (United Kingdom), and University of Alberta (Edmonton, Alberta, Canada).
Additional attendees from Breakthrough T1D included CEO Aaron Kowalski, Ph.D., Vice President of Research Esther Latres, Ph.D., the Medical Affairs team, a volunteer, and a member of the Participant Advisory Council, who is a person living with T1D.
The objective of this workshop was to take the first step toward creating Centers of Reference for T1D manufactured cell therapies. “The initial purpose is to accelerate readiness of healthcare professionals to deliver manufactured islet cell therapies once they become available,” explained Dr. Danne. “…making such a treatment a success needs teamwork. Accredited Centers of Reference will not only deliver advanced T1D treatments but also serve as a training hub for professionals aspiring to become experts.”
This workshop focused on better understanding what potential Centers of Reference need to be successful. The attendees covered a range of topics: what an ideal T1D care team might look like, the education and training required for experts in T1D manufactured islet cell therapy, and career development for early-stage T1D professionals.
The goal is to prepare expert clinicians—who are already doing islet cell transplants—to bring manufactured islet cell therapies into clinical practice at their institutions and others, once they have regulatory approval. These centers will serve as a benchmark for best practices in T1D manufactured cell therapy, establishing a network of expert teams to make sure that everyone who can benefit from manufactured cell replacement therapy is given the opportunity to consider it.
What the experts are saying

“We need to build consensus and teamwork. When manufactured cell therapies exist, it’s going to take significant coordination between endocrinologists, transplant surgeons, and people with T1D to ensure as many people as possible are benefiting from these therapies.”
Jon Odorico, M.D.
Professor of Surgery and Director of Pancreas and Islet Transplantation at University of Wisconsin Health Transplant Center
“We’re trying to solve access and awareness. There’s a definite gap between primary care endocrine diabetes specialists and transplant specialists…there’s so much more that we have to fill in for [people with T1D].”
Helen Nelson, BSN, RN, CCTC, CPTC
Program Manager, Organ Allocation/Clinical Triage and Pancreas Transplant Program at University of Wisconsin Health Transplant Center


“It’s going to be a significant problem if we have a cure but no one has access to it because no one can deliver it. We must work together—transplant surgeons, endocrinologists, researchers, everyone. It’s like building Cape Canaveral in anticipation of sending rockets into space.”
Peter Senior, MBBS, Ph.D.
Islet Transplant Endocrinologist, Professor in the Department of Medicine, and Director of the Alberta Diabetes Research Institute at the University of Alberta, Canada
“There is amazing excitement around creating cell therapies. People are excited about it. We must ensure that organizations like Breakthrough T1D bridge the gap between research and the T1D population so that there is no difference between an individual’s reality and what therapies are available.”
James Shaw, M.D., Ph.D.
Transplant Endocrinologist and Professor of Regenerative Medicine for Diabetes at the Institute of Transplantation, Newcastle upon Tyne, United Kingdom

This is just the beginning
Manufactured islet cell therapies are coming. We need teamwork to get these therapies into clinics so people with T1D don’t have to wait years to get them. This is why Breakthrough T1D is acting now: when the first manufactured islet cell therapy hits the market, multidisciplinary care teams around the world will be ready. These workshops—the first of many—will help accelerate the safe and effective integration of manufactured islet cell therapies into clinics.
“This way we will ensure that the medical community is ready to deliver manufactured cell therapies once they become more widely available.,” Dr. Danne said. Thanks to the hard work of the Medical Affairs team at Breakthrough T1D, this goal is in sight.

Project ACT series
This article is part of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) is shaping the future of cell therapies for type 1 diabetes (T1D). The next article in the series will focus on Project ACT’s advocacy efforts to ensure there is a regulatory pathway to approval for these therapies and that they will be covered by payers.
Read last month’s article about challenges and solutions of T1D cell therapies.
Despite significant advances in treatments for T1D, our community still has significant unmet needs. Breakthrough T1D believes that novel cell therapies will transform T1D management, and Project ACT is how we’re going to make them a reality.
First-generation cell therapies, including FDA-approved, donor-derived Lantidra® and Vertex’s manufactured islet therapy in phase 1/2/3 clinical trials, VX-880 (Zimislecel), are incredibly promising. They have some limitations, including:
- There are not enough donor-derived islets to meet the needs of everyone with T1D.
- These therapies are only available to people with severe hypoglycemia unawareness and hypoglycemic events.
- The number of people who can receive these therapies is further limited in that they must be able to tolerate chronic, broad immunosuppression.
Current research efforts at the preclinical, clinical, and manufacturing levels are working to address these challenges. The ultimate goal is a future in which manufactured islet therapies exist in large supply, survive and produce insulin in the body after implantation, and remain protected from the immune system. Learn more about Breakthrough T1D’s Cell Therapies Program and take a closer look at what researchers are doing to turn these ideas into a reality.
Clinical trials to keep an eye on
Up-and-coming cell therapies for T1D are in the clinical pipeline and working their way towards the market, including many that Breakthrough T1D has contributed to. There are some highly anticipated (and currently enrolling!) trials that we have our eyes on right now, and we hope to see data soon. Read about each study in detail below or scroll down to see a summary table.
Late last year, Vertex announced the expansion of their manufactured islet therapy, VX-880 (Zimislecel), to a phase 1/2/3 clinical trial, the final step before seeking FDA approval. This decision stemmed from groundbreaking data in the initial phases of the trial in which 11 of 12 participants reduced or eliminated the need for external insulin therapy. Currently, zimislecel is limited to people with severe hypoglycemia and requires chronic immunosuppression.
The results of the VX-880 trial are highly anticipated since this is the first time a scalable treatment for T1D has entered a final clinical testing stage, and regulatory submission is expected in 2026. Vertex is working closely with regulators to expand its manufacturing capabilities and ensure they are prepared for the therapy to hit the market.
Zimislecel would not have been possible without years of support from Breakthrough T1D and The T1D Fund: A Breakthrough T1D Venture. This includes research grants, an investment by the Fund in Semma Therapeutics (which was later acquired by Vertex), and much more.
It doesn’t stop there: Vertex is expanding their pipeline and investigating different ways to keep manufactured islets safe without standard anti-rejection immunosuppressants, including alternative immunosuppressive regimens, gene-edited immune-protected cells, and novel encapsulation devices.
Although Vertex’s T1D portfolio is progressing, the clinical development of VX-264, an encapsulated islet therapy that does not require immunosuppression, has been discontinued. While it was safe and well-tolerated in clinical trials, it did not meet efficacy and safety endpoints as measured by C-peptide, a biomarker for insulin production.
UP421 consists of islets derived from deceased donors that have been engineered to be hypoimmune, meaning they can avoid detection by the immune system without the need for immunosuppressants. Incredibly, the first person who received a partial dose of UP421, implanted in to forearm muscles, in a phase 1 clinical trial is making their own insulin, as demonstrated by increased C-peptide, without any side effects.
This is the first proof-of-concept evidence showing that this cell engineering approach can enable implanted islets to avoid immune destruction. The next step is applying this method to manufactured islets.
Breakthrough T1D is supporting research exploring similar cell engineering approaches to allow implanted islets to evade the immune system. The T1D Fund has also invested in Sana due to their distinctive hypoimmune manufactured islet replacement program, and Breakthrough T1D continues to work closely with them.
Tegoprubart is an immunotherapy that interferes with immune cell communication and dampens the immune response. This therapy is being tested in a Breakthrough T1D-funded phase 1/2 clinical trial as a novel anti-rejection immunosuppressant for people with severe hypoglycemia who have received deceased donor islets. Building on ongoing kidney transplant studies, this study will determine if tegoprubart can protect transplanted islets from rejection with fewer side effects compared to standard immunosuppressants, which is harsh on people and the implanted cells.
So far, of the first three participants, two have achieved insulin therapy independence. According to the study, tegoprubart is safer for both people and transplanted cells in comparison to standard immunosuppression, with milder side effects and greater islet survival. This study holds promise for preventing rejection of manufactured islets as well.
The T1D Fund has made several investments in Eledon to support this effort as it sees the potential to address the key unmet need of safe and effective immunosuppression for people who receive islet replacement therapies.
Cell Pouch™ is an implantable bio-hybrid organ that provides a specialized environment for transplanted islet cells by allowing them to access oxygen and nutrients provided by blood vessels, called vascularization.
The first cohort of a phase 1/2 clinical trial enrolled participants with severe hypoglycemia who received deceased donor islets within Cell Pouch in addition to standard immunosuppressants. Of the six enrollees, five remain insulin therapy independent from one year to more than five years. Cohort B is currently evaluating a higher-capacity Cell Pouch that can accommodate 50% more islet volume, and the trial will soon advance to Cohort C to further test safety and efficacy of the system.
Most excitingly, Sernova recently announced that following the conclusion of the ongoing clinical trial, they will initiate a new trial to test Cell Pouch implanted with manufactured islets—paving the way towards a scalable solution to T1D.
Breakthrough T1D has supported the development of Cell Pouch™ and continues to work with Sernova.
SR-02 is a manufactured islet cell therapy implanted onto the omentum, a fatty, protective layer around organs. This therapy is in a phase 1/2 clinical trial for people with severe hypoglycemia and requires immunosuppression. The trial is evaluating safety and insulin production as measured by C-peptide.
Seraxis is also working on another manufactured islet therapy (SR-03) that has been gene-edited so that anti-rejection immunosuppressants are not needed. They are hoping to initiate a new clinical trial for SR-03 in 2026.
The T1D Fund has invested in Seraxis to aid in the development of these therapies based on their distinctive and promising islet replacement approach.
CTX211 is another manufactured islet therapy that has been gene-edited to evade the immune system so that recipients do not have to take immunosuppressants. In an ongoing phase 1/2 clinical trial, these cells are implanted within a specialized device to help keep the cells alive in the body, and investigators are evaluating safety as well as insulin production measured by C-peptide. Results are expected this year.
Breakthrough T1D was a long-time supporter of ViaCyte, which initially developed the manufactured islets and partnered with CRISPR Therapeutics to genetically modify them. ViaCyte was acquired by Vertex in July 2022, and now CRISPR Therapeutics is the sole owner of this therapeutic platform.
- OPF-310 (Otsuka Pharmaceutical): encapsulated islets derived from pigs in phase 1/2 clinical trials
- ENC-201-CED (Encellin): donor-derived islets in a proprietary encapsulation device implanted subcutaneously in a phase 1 clinical trial
- Gastrin: a phase 1/2 trial testing if gastrin, a naturally occurring hormone involved in pancreatic development, is safe and helps retain or grow islets following donor-derived islet transplantation
Recruiting cell therapies clinical trials
| Therapy | Primary Outcome(s) | ID | Location | Phase |
| VX-880 (Zimislecel) | Safety, insulin independence, and absence of severe hypoglycemic events | NCT04786262 | US/Canada/UK/EU | 1/2/3 |
| VX-880 (Zimislecel) | Insulin independence in people with T1D and a kidney transplant | NCT06832410 | Canada | 2 |
| UP421 | Safety | NCT06239636 | Sweden | 1 |
| Tegoprubart + donor islets | Insulin independence | NCT06305286 | US (Chicago, IL) | 1/2 |
| Cell Pouch™ + donor islets | Safety | NCT03513939 | US (Chicago, IL) | 1/2 |
| SR-02 | Safety, C-peptide | NCT06651515 | US (Pennsylvania) | 1/2 |
| CTX211 | Safety, C-peptide | NCT05565248 | Canada | 1/2 |
| OPF-310 | HbA1c<7% and absence of severe hypoglycemic events | NCT06575426 | US (Chicago, IL) | 1/2 |
| ENC-201-CED ENCRT + donor islets | Safety | NCT06408311 | Canada | 1 |
| Gastrin + donor islets | Insulin independence, absence of severe hypoglycemic events, and HbA1c<6.5% | NCT03746769 | US (Duarte, California) | 1/2 |
Where do we go from here?
The emergence of cell therapies for T1D in clinical trials is incredibly exciting for the T1D community. Advancements using deceased donor-derived islets are paving regulatory pathways for manufactured islet therapies—which are curing people with T1D in clinical trials—to make their way towards the market.
“One of the greatest effects that manufactured islet cell therapies will have for the T1D community is being able to think about their type 1 diabetes less,” explained Nicholas Mamrak, Ph.D., a scientist at Breakthrough T1D. This may soon be a reality for a subset of people with T1D as we drive towards the approval of a first-generation manufactured islet replacement therapy, reducing the day-to-day burden of managing blood glucose and insulin dosing.
The ultimate goal is to make sure manufactured islet therapies are available to everyone with T1D—ideally without the need for immunosuppression. This is the objective of Breakthrough T1D’s Project ACT.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Before cell therapies can become available for everyone, they must first receive regulatory approval. A key part of Project ACT is streamlining and advancing regulatory pathways for the treatment of T1D. To help achieve this, Advocacy and Regulatory leadership teams at Breakthrough T1D are working on a new publication capturing how day-to-day lives have changed for people with T1D who have received cell therapies. By using personal experiences to share what benefits of cell therapies matter most to patients, we can help regulators understand the powerful impact that islet replacement therapies can have.
None of this would have been possible without the T1D community’s continued generosity and support, as we all work together to move the needle forward.
Have an impact by participating in clinical trials
Without clinical trials, we would never know if new therapies developed by scientists in the lab could make a difference in people’s lives. This is where the T1D community comes in—by volunteering to participate in clinical trials, you become uniquely positioned to help drive biomedical research forward. Moreover, by participating, you help not only yourself, but everyone with T1D. Find a clinical trial near you and see if you are eligible to participate. Connect with a Clinical Trial Education Volunteer in your area, who can answer any questions you may have.