
Greetings from Barcelona, Spain! From March 11–14, 2026, the world’s leading type 1 diabetes (T1D) researchers, healthcare professionals, and industry partners gathered to share groundbreaking advances in T1D science—and Breakthrough T1D–supported work (and staff) were front and center.
Below is everything you need to know from the first two days of this remarkable conference, including key takeaways from panels and sessions chaired by Breakthrough T1D staff and supported researchers. Featured participants included Breakthrough T1D CEO Aaron Kowalski, Ph.D.; Chief Scientific Officer Sanjoy Dutta, Ph.D.; Thomas Danne, M.D., Ph.D., Chief Medical Officer; Vice President of Medical Affairs Anastasia Albanese O’Neill, Ph.D., APRN, CDCES; and many more.
Screening
T1D Screening and Early Detection is a key mission priority for Breakthrough T1D—and multiple presentations illustrated how important it is, and why we need to screen more people to achieve our vision of a world without T1D.
The landscape of T1D screening today is incredibly exciting—and has come a very, very long way in recent years. Across the globe, the number of people being screened, which includes kids and adults, is growing significantly. In addition to the U.S., screening numbers are on the rise across Europe and now in Japan and Central America.
So—what are some big takeaways on screening?
Global screening programs
EDENT1FI, a European screening initiative supported by Breakthrough T1D, presented data from around the world on screening for early detection. One of the presenters was Breakthrough T1D VP of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, who shared our screening strategy and underscored the importance of international consensus recommendations for screening and monitoring that can be used in clinical care. This session outlined the global scope of screening that Breakthrough T1D is supporting.

Breakthrough T1D is funding a truly global network of screening programs. From ELSA in the United Kingdom to ASK and TrialNet in the U.S. to FR1DA in Germany, all these centers are working together to build the case for general population screening. They’re doing this by improving outcomes for people with detected autoantibodies, providing follow-up monitoring, enrolling eligible individuals in clinical trials (including Breakthrough T1D-funded INNODIA), offering FDA-approved therapies like teplizumab (Tzield), and learning the best ways to educate and inform people of the benefits and logistics of screening and follow-up monitoring.
Education and communication are key. One incredible learning from Italy and the United Kingdom is how people learn about screening. For the majority of individuals, it’s through school and through social media. It’s up to all of us to help spread the word. Learn more about our screening programs here.
Another key takeaway from the ELSA study: no one who learns that they have T1D autoantibodies regrets having that knowledge. However, there are gaps around how we can help people with that information and how we can better support families who have detected T1D early.
Free, voluntary general population screening for T1D and Celiac Disease is now law in Italy. Francesca Ulivi from the Italian Diabetes Foundation shared what Italy is learning about how they are increasing the number of people screening—and to add to an earlier note, education is key.
Clinical adoption of T1D screening
In 2024, Breakthrough T1D led the way with the publication of Consensus Guidance for Monitoring Persons with Islet Autoantibody-Positive Pre-Stage 3 Type 1 Diabetes. This report contains guidelines for what to do when T1D autoantibodies are detected—but it does not touch on the bigger question: who to screen?

To make the case for general population screening, Breakthrough T1D convened a group of international experts to establish guidance for the clinical adoption of screening for early detection. This paper is expected to be published later this year and will be a clinical roadmap for general population screening—so more people can realize the benefits.
Breakthrough T1D Chief Scientific Officer, Sanjoy Dutta, Ph.D., also spoke to our organization’s vision for expanding general population screening.
Breakthrough T1D is funding a series of pilot screening programs across the country. These centers aim to develop and test diabetes center workflows for screening and monitoring for T1D. We are learning a lot from these, as presented by Carla Demeterco-Berggren, M.D., who runs a Breakthrough T1D-funded screening pilot in San Diego. Some key takeaways:

- With proper monitoring, DKA is almost completely preventable. Her center has had 0 people who had autoantibodies detected present with DKA at diagnosis.
- Successful T1D screening takes an integrated approach within the hospital setting, from hospital leadership to the nurses running the diabetes clinics.
- More importantly—it takes a team! Screening is just one step. Monitoring is where DKA prevention happens. And it takes a team to do that.
- The more people we screen, the more people can benefit!
Disease-modifying therapies
Disease-modifying therapies (DMTs) have the potential to change the course of T1D by slowing, halting, or reserving its progression. Teplizumab (called Tzield in the U.S.) was the first DMT approved by the FDA in November 2022 for individuals with early-stage T1D, delaying the onset of clinical, symptomatic T1D and the need for insulin therapy by a median of two years. Since its approval, and thanks to increased education and awareness around screening, hundreds of people have had the opportunity to benefit from teplizumab.
Breakthrough T1D and Tzield
Learn more about the first disease-modifying therapy for T1D and how Breakthrough T1D helped guide it through every stage of therapy development.
The changing landscape of available T1D therapies requires healthcare professionals (HCPs) to consider how to best work within existing care structures to support their patients who are using therapies like teplizumab. In an informative session led by Breakthrough T1D-funded researchers Prof. Colin Dayan, BSc, MBChB, Ph.D., FRCP from Cardiff University, Michael Haller, M.D. from the University of Florida, and Prof. Olga Kordonouri from Children’s Hospital Auf der Bult in Hannover, Germany, these superstar endocrinologists walk us through practical considerations for use of teplizumab in clinics and real-world experiences from the perspective of both teplizumab recipients and caregivers.

- People considering teplizumab should work with their diabetes care team to ensure they meet pre-assessment, vaccination, and other requirements and are able to tolerate intravenous access for the duration of the infusion.
- Teplizumab requires 14 consecutive infusion days, and people may have the opportunity to finish the infusion process at home with a home healthcare aide after the first five days in an infusion clinic. Everyone is consistently monitored for safety both during and after the infusion process.
- Multidisciplinary staff, diverse training, and specific infrastructure are required for successful teplizumab infusion, monitoring, and follow-up. This includes infusion nurses, on-call clinicians, pharmacy coordination, thorough documentation, after-hours care, support for travel and lodging for families coming from afar, and centers that have the capacity to deliver teplizumab for 14 days straight.
- In a survey of 47 people who received teplizumab in the U.S., 49% had a family history of T1D, 47% were initially misdiagnosed with type 2 diabetes, and 36% had other autoimmune conditions.
- In the same survey, 98% of people reported that their rationale for choosing teplizumab was to delay T1D onset. While most respondents were grateful to receive the therapy and would recommend it to others, people still check blood sugars often and worry about what to eat.
Cell therapies
While the current standard of care for T1D has taken immense strides in the recent past, one thing is abundantly clear: people with T1D are not doing well enough. Per data presented by Breakthrough T1D-funded researchers Jason Gaglia, M.D., Trevor Reichman, M.D., Jennifer Sherr, M.D., and Jay Skyler, M.D. at Vertex Pharmaceuticals’ Industry Session, globally, about 25% of people with T1D are hitting their ideal HbA1c target of under 7%. Automated insulin delivery (AID) systems—which have been transformative for T1D care—are improving outcomes, but the majority of people are not achieving their ideal goals. This means that most people with T1D are more at risk for the most common complications of the condition, including heart disease, kidney disease, and diabetic eye disease.
Among the individuals who are achieving ideal HbA1c goals, an acute complication begins to emerge: severe hypoglycemic events.
This paints a pretty clear picture: we need better therapies to help people do better, and cell therapies have the potential to fill the gaps between how people are currently doing and where they need to be to prevent long-term and acute complications.
Currently, deceased-donor islet transplants are an option, but they are not an option for the majority of people with T1D due to a scarcity of deceased-donor islet cells (it can take 4 donor pancreases to get enough cells for one person) and the drawbacks of chronic immunosuppression.

The future Vertex and the field at large is driving toward is one where manufactured islets are mass produced at scale, which solves the scarcity problem, and are used with novel techniques to keep them safe from the immune system—including gene-edited cells and alternatives to modern immunosuppression (like Eledon’s tegoprubart).
One other note: someone in the audience inquired as to why more people in the U.S. do not receive islet transplants. The panelists were in agreement that it is due to the current regulatory environment for deceased-donor islet cells, which classifies them as biologics, not organs. This reinforces Breakthrough T1D’s efforts to urge the Department of Health and Human Services to reclassify deceased-donor islet cells as organs—which it has the legal authority to do now.
Improving Lives
All the major T1D tech companies were present and accounted for at ATTD. Omnipod, Abbott, Tandem, Minimed, Dexcom, and more highlighted their current devices and how they can improve diabetes management.
We’re not going to cover the specifics of these devices, but one theme was consistent throughout the presentations: people utilizing AID systems and continuous glucose monitors (CGM) have better outcomes.
But there were some exciting developments around adjunctive therapies, the use of artificial intelligence, and a new approach: gene therapy.
The use of Artificial intelligence (AI) is a hot topic at ATTD. This topic was presented by longtime Breakthrough T1D collaborator Boris Kovatchev, Ph.D., in the opening lecture and Giacomo Cappon, M.D., Marc Breton, Ph.D., and others in a plenary session chaired by Breakthrough T1D CEO Aaron Kowalski, Ph.D.

One of the more interesting areas discussed was the use of digital twins—virtual models of a person with T1D. These models allow researchers and clinicians to test potential changes in diabetes management in a simulated environment before talking through and implementing those adjustments in real life. The potential is big, but there are still risks. AI-generated “hallucinations,” e.g. 2 + 2 = 7, can occur, and any therapeutic recommendations need human review. Even so, the promise of these tools for T1D is significant.
Dr. Kovatchev and Dr. Breton also highlighted how AI is already being used in clinical trials. Their talks focused on AID As a Network (AIDANET), an AI-based AID system. This fully closed-loop setup—using a Dexcom G6, a Tandem t:slim insulin pump, and a smartphone—first learns from insulin pump and CGM data. Once it understands a person’s patterns, it takes over and fully automates insulin delivery. No mealtime announcements required.
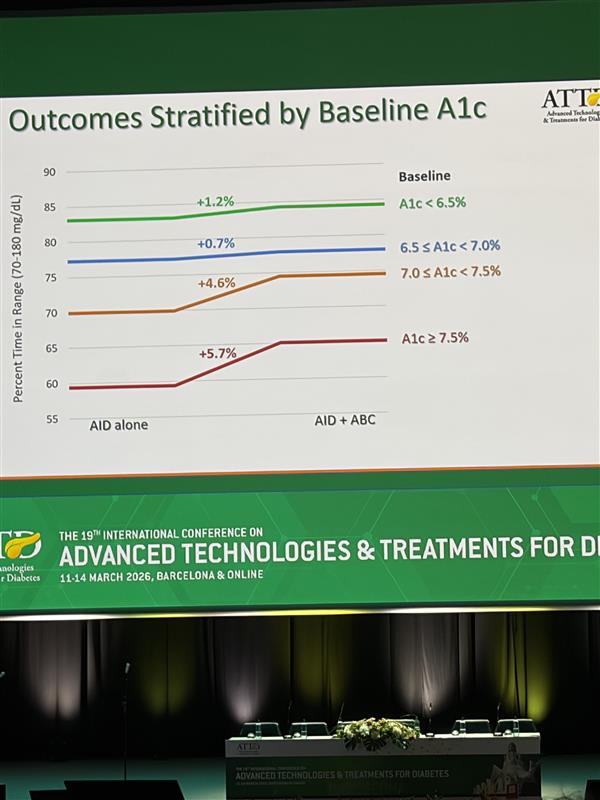
Dr. Breton shared data from a Breakthrough T1D–funded study using AIDANET, and the results were striking. People who already had lower HbA1c levels saw modest improvements, while those starting with higher HbA1cs experienced significant gains. In this fully closed-loop, no-bolus approach, AIDANET was better than standard use across nearly every outcome for the 34 people in the clinical trial—and overall, it delivered superior glycemic control.
How does it achieve this? Essentially, AIDANET performs about as well as usual care during the day and then outperforms it at night.
Breakthrough T1D continues to fund this work, including the AIDANET clinical trial in adolescents and young adults that Dr. Breton presented. The takeaway so far: AIDANET delivers equivalent control for people who are already well managed—and even better outcomes for those with higher HbA1c levels.
And here’s something to look out for: there is an ongoing clinical trial testing AIDANET at home in over 50 people with T1D. The data should come out soon—and should be another proof point in support of the use of AI in T1D.
Gene therapy in T1D
Longtime Breakthrough T1D collaborator Jeremy Pettus, M.D., was in attendance to share information from Kriya Therapeutics, a T1D Fund portfolio company. Kriya is pursuing a gene therapy for T1D, and it’s an exciting new approach to achieving glycemic control.
Gene therapies explained
Gene therapies are still fairly new—there are 7 approved today, and they primarily are used for rare diseases. They work by using a safe, non-pathogenic virus to deliver genetic information into the nucleus of a cell. This process does not alter the individual’s chromosomes or genetic makeup but can correct genetic abnormalities or change the behavior of cells to treat disease.
Applications in T1D
Kriya’s approach is to modify muscle cells to secrete insulin. This is very different from, for example, beta cell therapies, which place insulin producing beta cells into people with T1D. In Kriya’s therapy, there are no new beta cells or transplanted cells that require immunosuppression—it’s just enabling muscle cells to make insulin.
The administration of this therapy is fairly straightforward. For 2-3 months in advance of the procedure, the recipient takes immunomodulatory drugs. Then, they undergo a procedure where they receive intramuscular injections in both thigh muscles. Then, the person goes home. After 2-3 months, the cells are fully up to speed and making insulin, which has the potential to help control blood sugar levels and reduce insulin therapy requirements.
Clinical trials
In large animal models, the data is very encouraging—after receiving the therapy, blood glucose levels were normalized on the order of years. Based on this data, clinical trials are expected to begin in people this year.
This phase 1 clinical trial, called the Progress Study, will assess dosage and safety, and the phase 2 trial will expand to assess broader safety and efficacy.
This is a new and innovative approach supported by the T1D Fund. If you’re interested in participating in the clinical trial, stay tuned to our clinical trials page, which is regularly updated with opportunities like this.
Medical Affairs
Breakthrough T1D Chief Medical Office Thomas Danne, M.D., Ph.D. ,outlined the goals of our Medical Affairs team—which is to improve the world’s ability to accelerate implementation of current and future breakthroughs so that, ultimately, people with T1D can have better quality of life.

The current state of endocrinology globally is a growing concern. For example, we don’t have enough endocrinologists and diabetes educators to take on of the growing and aging population of people with T1D. Clinical adoption for teplizumab, the first approved DMT for T1D, has been slow. We must build the infrastructure today to ensure that in the future, there isn’t a gap between the breakthroughs in the lab becoming breakthroughs for people with T1D.
Dr. Danne outlined the vision for a training program that will entice a new generation of endocrinologists as the field evolves and new methods of treating T1D are available. Breakthrough T1D and ATTD are supporting a certificate program for an interventional diabetologist, which includes training in immunology, using immune therapies to treat individuals in stage 1 and stage 2, education on cell therapies for those in stage 3 and stage 4, and the other advancements in the pipeline. He also spoke about other work the Medical Affairs team is involved in including the Centers of Reference, an international collaboration creating a roadmap for implementing manufactured cell therapies into clinics once they become available.
More to come!
We have even more exciting updates to share with you. Keep an eye out for our recap of days 3 & 4 of ATTD, coming next week!
This article was written by Brian Herrick and Sandy Vogt, Ph.D.