
Breakthrough T1D is back from Barcelona! The final two days of ATTD were jam-packed with more incredible updates across all areas of our mission. Breakthrough T1D staff chaired and participated in many sessions—including Breakthrough T1D Senior Vice President of Research, Esther Latres, Ph.D.; Vice President of Medical Affairs Anastasia Albanese O’Neill, Ph.D., APRN, CDCES; Senior Director of Research, Jonathan Rosen, Ph.D.; and more.
Jump down to:
Screening
Screening is important! From preventing life-threatening diabetic ketoacidosis (DKA) at diagnosis, to giving people the opportunity to intervene with Tzield or disease-modifying therapies in clinical trials, to having more time to plan and prepare for type 1 diabetes (T1D) onset—screening and early detection are essential to our mission. And it must happen at a large scale if we’re going to achieve cures—which is why it was so heavily represented at ATTD 2026.
Vice President of Medical Affairs Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, chaired yet another panel where screening was again discussed. To repeat from our earlier recap, T1D Screening and Early Detection is a key mission priority for Breakthrough T1D. Multiple presentations illustrated how important it is, and why we need to screen more people to achieve our vision of a world without T1D.
One theme, and why it came up over and over again, is that we need more public education about screening, the benefits of early detection, and how mission-critical it is. It’s not an accident that there were many sessions that all hammered this home.
What can you do? Tell your community about screening!
Disease-modifying therapies
What are disease-modifying therapies?
Disease-modifying therapies are drugs that change the course of T1D. That means slowing or halting the progression of the disease, preventing it from ever occurring, and reversing it entirely.
There were several exciting presentations from some key Breakthrough T1D collaborators. This includes Jay Skyler M.D., MACP, FRCP, who outlined the challenges and obstacles that must be overcome at every stage in T1D in order to fulfill his personal mission to eradicate T1D before he retires. This includes disease-modifying therapies at different stages of T1D.
Another noteworthy lecture came from Chantal Mathieu, M.D., Ph.D. (who happens to be on the advisory committee for the Breakthrough T1D Clinical and Research Congress!). She focused on C-peptide, which is natural biomarker of insulin production.
Breakthrough T1D believes C-peptide should be a validated clinical trial endpoint. Dr. Mathieu agrees! Currently, metrics like HbA1c are used to evaluate the efficacy of therapies for early-stage T1D. In Dr. Mathieu’s opinion, HbA1c as a clinical trial endpoint will mean, simply, that it will be a very long time before another DMT joins Tzield as FDA an approved therapy for this disease.
And, most importantly, there are real benefits to maintaining beta cell mass and, therefore, having increased C-peptide function! We know this! And it’s up to Breakthrough T1D, academia, and others to persuade regulators that it’s time for it to be accepted as a clinical trial endpoint so there can be more options for people with T1D.
Cell therapies
Cell therapies, which place insulin-producing cells inside people with T1D, are a huge Breakthrough T1D priority, as exemplified by Project ACT.
Senior Vice President of Research, Esther Latres, Ph.D., chaired a session titled “Islet Cell Therapy: Cure for Type 1 Diabetes – How Far Are We?” This session highlighted Breakthrough T1D’s approach to cell therapies in pursuit of our ultimate goal: manufactured islets, produced at scale, transplanted without the need for chronic immunosuppression.

Cell therapies: Where are things now?
Islet transplants have been around for decades, but they require chronic immunosuppression. Additionally, deceased donor islets are in limited supply, meaning there are not enough islet cells for everyone with T1D who may want them.
Update on Eledon’s tegoprubart
Let’s start with the topic everyone is talking about: the use of Eledon’s tegoprubart with deceased donor islet cell transplants.
Breakthrough T1D is funding a trial at the University of Chicago evaluating the use of the targeted immunosuppressive therapy tegoprubart, instead of standard, broad immunosuppressants like tacrolimus, in islet cell transplants. The data so far has been very encouraging—and Principal Investigator, Piotr Witkowski, M.D., Ph.D., was at ATTD to share more.
What is Breakthrough T1D’s relationship with Eledon?
Breakthrough T1D has had a relationship with Eledon for several years, since Breakthrough T1D brought Eledon into the T1D space. Starting in 2023, Breakthrough T1D began funding a clinical trial that uses their drug, tegoprubart, with islet transplants in T1D, with initial additional support from The Cure Alliance and the Diabetes Research Institute. Additionally, the T1D Fund: a Breakthrough T1D Venture invested in Eledon in 2023.
All 12 recipients are off external insulin or on target to stop external insulin.
To date, 12 people with T1D have received deceased donor islet transplants and are receiving tegoprubart infusions every three weeks. All of the individuals are doing exceptionally.
Here’s some stats to back it up:
- All 12 subjects immediately experienced improved blood sugar levels and control.
- As of today, 10 of the 12 individuals are entirely off insulin therapy. The other two are on that trajectory.
- There have been zero unexpected adverse events, severe hypoglycemic episodes, thrombotic events, opportunistic infections, signs of rejection, or gastrointestinal symptoms.
- Crucially, there have been no signs of kidney and neurotoxicity, which is something that is observed in individuals who receive islet transplants and use tacrolimus.
The next step for this therapy is a study testing deceased donor islet cells in people with T1D and chronic kidney disease, funded by Breakthrough T1D.
The main takeaway? This remains a very promising alternative to current immunosuppression.
Sana’s gene-edited islets continue to make insulin
Per-Ola Carlsson, M.D., Ph.D., presented early results from a novel cell therapy approach designed to help transplanted islet cells evade immune attack while continuing to produce insulin.
The strategy uses gene editing to create hypoimmune islet cells—cells engineered to avoid detection by the immune system while maintaining their insulin-producing function. This approach is one of several next-generation cell therapy strategies being prioritized by Breakthrough T1D to help overcome one of the biggest barriers to cell replacement therapies: immune rejection.
Prior to this study, the concept had been demonstrated in non-human primates. Carlsson and his team have now taken the next step by testing the approach in a person living with T1D.
In this first-in-human study, deceased donor islets were genetically modified to become immune-evasive and then transplanted into the forearm. Because this was a phase 1 trial, the primary goal was to assess safety, while also monitoring islet function through C-peptide, a marker of the body’s own insulin production.
Importantly, the transplant included only about 5% of the number of cells typically needed to fully restore insulin production, reflecting the exploratory nature of the study.
The results so far are encouraging. After 60 weeks, there have been no severe or unexpected adverse events, meeting the trial’s primary safety endpoint. At 14 months after transplantation, the participant continued to produce detectable C-peptide, indicating that the transplanted cells remained alive and functional.
Researchers also observed that C-peptide levels temporarily declined after about one year, likely due to beta cell exhaustion, but subsequently recovered. This is an encouraging signal that the transplanted cells may be capable of regaining function.
While still very early, these findings provide important proof of concept that gene-edited, immune-evasive islet cells can survive and function in a person with T1D. If confirmed in larger studies, this approach could help move the field closer to cell therapies that work without long-term immune suppression—a major goal for the future of T1D cures.
Addressing challenges in cell therapies
Holger Russ, Ph.D. explored a key problem that must be overcome: manufacturing islets at a large scale is hard to do! He shared the processes that his team at the University of Florida uses and noted a secondary problem: we make all these cells, and a significant portion are lost post-transplantation. To solve that, he presented his work on different gene-editing strategies to keep these cells alive.
Katy Digovich, CEO of Minutia, discussed a different problem that needs to be solved: how do we monitor the transplanted islets? Minutia’s approach is to integrate gold (yes, gold) nanoparticles into the islet clusters, which allows them to safely monitor the cells and track graft survival and rejection.
All of this work is supported by Breakthrough T1D and/or the T1D Fund.
More to come soon for Vertex’s manufactured islets
Data from Vertex Pharmaceutical’s phase 1/2/3 clinical trial for zimislecel was presented by Trevor Reichman, M.D. This data is not new, but it reinforces the reasons for optimism around this manufactured islet cell therapy.
This therapy consists of fully differentiated, manufactured islet cells, infused into the portal vein using an approach similar to traditional donor islet transplantation. Participants are on immunosuppression, and the safety profile is consistent with the use of immunosuppressive medications.
Unfortunately for the T1D community, we’re not going to get new data for Vertex for some time because the data currently being collected will be used for their upcoming FDA submission. However, stay tuned for a Q&A with Vertex in the near future!
Improving Lives
Breakthrough T1D Director of Research, Jonathan Rosen, Ph.D., chaired the session “Bridging the Gap Between Academia and Industry in T1D.” This is an important priority area for us as we see ourselves as “gap fillers.” There is a pipeline from academia to industry to people with T1D, and Breakthrough T1D’s strategy is to identify these gaps and ensure that the pathway is as smooth—and complete—as possible.
This session featured several leaders from biotech illustrating how, ideally, everyone can work together to move products along. Some key points:

- Academia, startups, and big pharma companies all have strengths and weaknesses. All roles are important and it’s crucial to understand the limitations and capabilities of each player.
- Other key players: governments and non-profit organizations. Thanks to the Breakthrough T1D Advocacy team, there has been billions of funding to T1D research over the past few decades. All these products must undergo regulatory review. All play a part!
- What Breakthrough T1D can do is identify the shared objectives. Industry and Academia are not opposing forces, and it’s up to Breakthrough T1D to ensure everyone is bringing their strengths to the table and row in the same direction.
The use of adjunctive therapies, including GLP-1s, is a priority of the Breakthrough T1D’s Improving Lives Portfolio. The use of these drugs, which is often off-label and includes semaglutide and tirzepatide, is increasing in the T1D community.
The data backs this up. Satish Garg, M.D., presented research showing these medications may support weight loss, lower insulin requirements, and improved blood glucose, specifically including better time in range. We also heard again from Viral Shah, M.D., who gave a recap of the Breakthrough T1D-funded ADJUST-T1D study, which showed that the GLP-1 receptor agonist semaglutide (Ozempic) can improve weight and glycemic outcomes in people with T1D.
It’s true: GLP-1s are increasingly being used off-label in the T1D community, as demonstrated by the Breakthrough T1D-funded work of Francisco Pasquel, M.D., MPH. Using large-scale patient datasets from electronic health records, Dr. Pasquel found that rates of GLP-1 use are highest in people living with T1D and obesity in addition to those with cardiovascular disease (CVD) or kidney disease. While this class of drugs has potential to improve weight and blood sugar management, we need larger clinical trials to confirm safety, identify less-common side effects, and get a better idea of the non-glycemic benefits that GLP-1s may have for the T1D community.
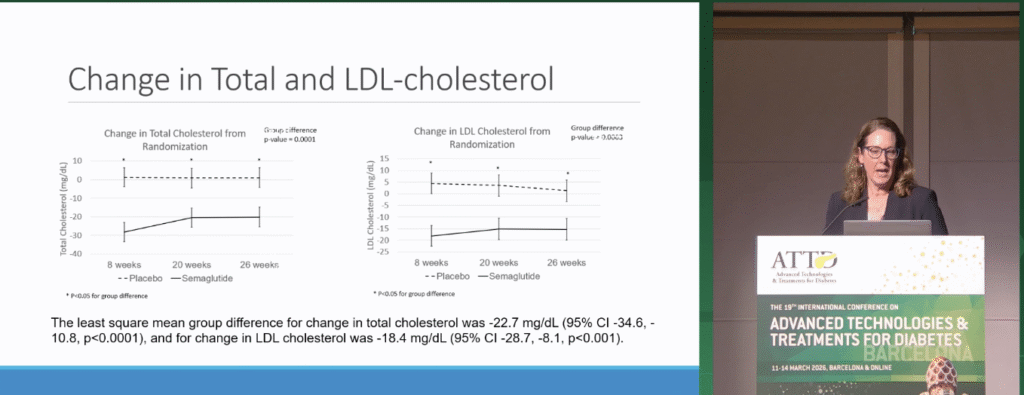
To make some headway on the benefits of GLP-1s beyond glucose management, Janet Snell-Bergeon, Ph.D., MPH walked us through cardiovascular outcomes that were measured as a part of the ADJUST-T1D study. People treated with semaglutide had significant decreases in cholesterol and blood pressure. After plugging participant data into a cardiovascular risk prediction tool, Dr. Snell-Bergeon reported that people with T1D treated with semaglutide have a 20% reduced CVD event risk after five and ten years. We need more therapies to address CVD in T1D—while large-scale clinical trials are needed, this data demonstrates the potential for GLP-1s to improve cardiovascular outcomes in people with T1D.
One key thing to keep in mind when it comes to GLP-1s: safe implementation. Clinicians must work hard to avoid hypoglycemia while also preventing the onset of ketones and DKA. Breakthrough T1D has led this work for years, and is currently helping promote several clinical trials, including the SUPRASS-T1D study.
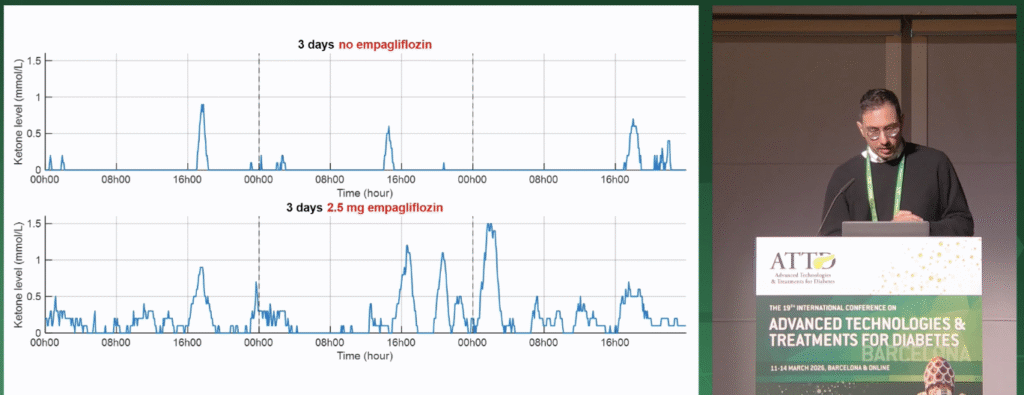
The risk of DKA also exists for a class of drugs known as SGLT inhibitors, which have proven to be effective for heart and kidney disease in type 2 diabetes (T2D) and people without diabetes, but are still being tested in T1D. Breakthrough T1D-funded researcher Ahmad Haidar, Ph.D., presented his work on changing ketone levels dependent on treatment with empagliflozin, an SGLT inhibitor, and lifestyle factors. Participants wore a continuous ketone monitor (CKM), and data showed that empagliflozin, in addition to lower-carbohydrate diets and greater physical activity, can increase ketone variability and the risk of ketosis. Dr. Haidar suggested that people with T1D may benefit from clinical time in range targets for ketones, and his studies demonstrate the potential of tracking ketone levels to predict and avoid DKA.
Thomas Danne wins an award

Breakthrough T1D Chief Medical Officer Thomas Danne, M.D., Ph.D., received the Children with Diabetes (CWD) President’s Award for Innovation. The award acknowledges a healthcare professional who exemplifies extraordinary leadership in using innovation and advancements in technology for devices that help people with diabetes thrive. An internationally recognized leader in endocrinology and pediatric diabetes, Dr. Danne has spent decades advancing research, clinical care, and global collaboration in the field.
“Breakthrough T1D is incredibly proud of Dr. Danne’s achievements and this well-deserved award for innovation and leadership in diabetes health care,” said Aaron Kowalski, Ph.D., Breakthrough T1D CEO. “Thomas’ many contributions to driving breakthroughs in type 1 diabetes therapy development and care have had a tremendous impact on the global T1D community and are instrumental in moving our mission forward, faster.”
Adios from Barcelona!
The next scientific conference we’ll be covering is the Annual American Diabetes Association (ADA) Scientific Session, which takes place from June 5-8 in New Orleans.
And, don’t forget, Breakthrough T1D will be hosting our first ever Clinical and Research Congress in Philadelphia in October! Over two and a half inspiring days, clinicians, researchers, scientists, healthcare professionals, industry leaders, and the T1D community will explore cutting-edge advances in T1D discovery science, immunotherapies, automated insulin delivery, islet cell therapies, psychosocial care, health equity, and more. Designed to foster collaboration and spark innovation, CRC 2026 will provide translational insights, interactive learning, and meaningful dialogue, all with one shared goal: improving lives and driving toward a world without T1D. Register today—and submit abstracts!
This article was written by Brian Herrick and Sandy Vogt, Ph.D.