
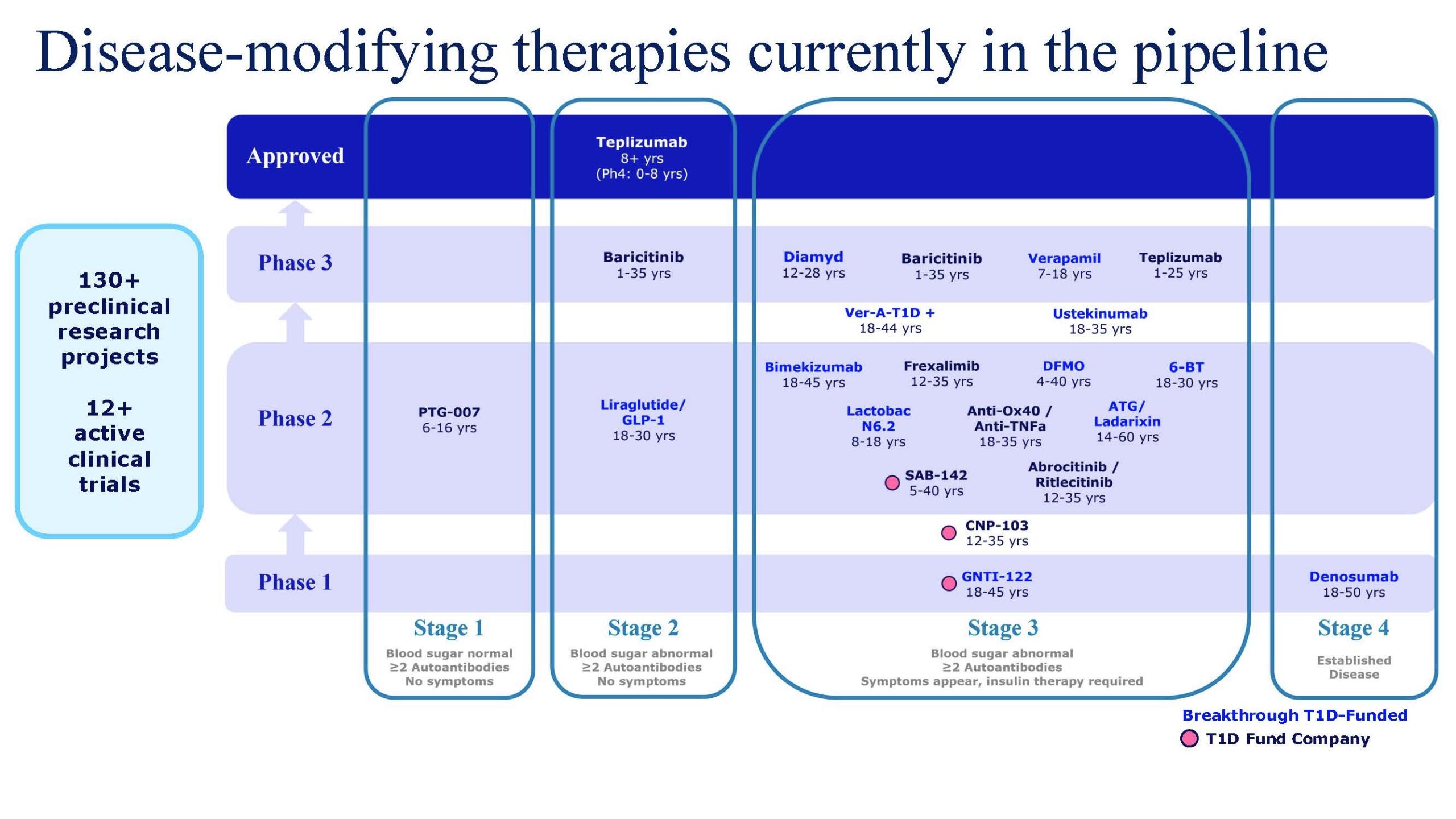
Breakthrough T1D is focused on developing drugs, or disease-modifying therapies (DMTs), that change the course of T1D for everyone affected by it—and those yet to be. That means slowing or halting the progression of the disease, preventing it from ever occurring, and reversing it entirely through immune therapies and beta cell therapies.
Note: To enlarge the image below, left click on it.
Update on Diamyd’s DIAGNODE-3 Clinical Trial
On March 27, Diamyd Medical announced results from an interim analysis of the Breakthrough T1D-funded Phase 3 DIAGNODE-3 trial, which assessed the ability of of Diamyd (retogatein or rhGAD65) to preserve insulin production (C-peptide) in the first 174 out of 321 participants in the trial.
Phase 3 Clinical Trials pave the way for discussions with regulatory agencies toward approval.
Surprisingly, and contradictory to positive data from prior phases of this clinical trial, the interim analysis showed that there was no observable preservation of insulin production (C-peptide). These results underscore the complexity of immune therapies in T1D and confirm Breakthrough T1D’s continued support of the clinical development of emerging disease-modifying therapies.
Breakthrough T1D’s Perspective on the DIAGNODE-3 Results
While this outcome was not expected, Phase 3 trials provide significant learnings for the development of future therapies and the field overall. Critical insight can be gained through the analysis of the data, even from clinical trials that do not meet their primary endpoint. These moments sharpen our understanding and accelerate better-designed approaches and more targeted application of therapies that are in development.
“Progress in type 1 diabetes research and treatment has always required persistence and learning from every study,” said Josh Vieth, Ph.D., Breakthrough T1D Senior Director of Research. “These results reinforce that immune interventions in type 1 diabetes are complex and highly context dependent. It’s imperative that we take these results seriously, understand what they mean, and use them to make strategic decisions about what’s next for retogatein and other disease-modifying therapies. There remains an unmet need for safe, effective therapies that rebalance the immune system in a targeted way, and Diamyd Medical is a pioneering approach to precision medicine for type 1 diabetes in clinical applications.”
What’s Next
Breakthrough T1D is in contact with the Diamyd team about next steps, which will include careful, collaborative analysis of the data from the trial to understand why these results differ from previous results. Diamyd plans to continue core operations, including manufacturing activities, during this period of analysis before determining the appropriate course of action. This includes the possibility of testing this therapy earlier in the progression of T1D (Stage 1 or 2), for which they have previously received “Fast Track” designation, an FDA program which would expedite this process.
Our Multi-part Strategy for Disease Modifying Therapy Development
Breakthrough T1D has been funding DMT research for decades. Following the 2022 approval of Tzield, the first DMT for T1D, we remain focused on our strategy for the development of disease-modifying therapies that can slow, halt, or reverse the course of T1D.
This strategy aims to both rebalance the immune attack as well as support a person’s own beta cells. Tolerization, or re-training the immune system to stop attacking beta cells without broad immunosuppression is an important part of that strategy. We are currently funding disease-modifying trials with companies like GentiBio to test their engineered Tregs, and trials aimed at repurposing existing drugs including DFMO, Bimekizumab, Denosumab, and ATG for use in T1D. To learn more, consult our previous article about this work.