Since 1994, Marshalls has been a committed supporter of Breakthrough T1D.
As Breakthrough T1D’s largest corporate partner, Marshalls raises more than $3 million each year through in-store fundraisers at checkout, donations from the TJX Foundation, and store support of local Breakthrough T1D chapters across the country. These combined efforts have resulted in more than $47 million in donations to-date for Breakthrough T1D, helping to advance our mission of accelerating life-changing breakthroughs to cure, prevent, and treat type 1 diabetes (T1D) and its complications.
Marshalls’ 2025 in-store fundraising campaign for Breakthrough T1D runs from Sunday, August 31, through Saturday, September 27. Find your local Marshalls here.
Creating a better future

Digital creator Kris Leeper’s type 1 diabetes journey began with his diagnosis in 2013. It was, in his own words, “terrifying.”
“My wife and I were bombarded with conflicting information,” Kris said. “We were navigating unchartered territory as a family, just two years into our marriage. I felt lost and uncertain about the future.”
T1D technology changed the game for Kris. “The introduction of a continuous glucose monitor (CGM) and pump revolutionized my outlook on living with T1D. I immediately felt better,” he said. “Access to and understanding of diabetes technology has profoundly improved my daily life.”
Kris appreciates Marshalls for bringing awareness to Breakthrough T1D’s efforts to make life-changing technologies a reality for the more than 1.5 million Americans facing T1D.
“Companies like Marshalls play a pivotal role in bringing Breakthrough T1D’s mission to the mainstream,” he said. “Marshalls’ support for advocacy, research, and fundraising demonstrates the power of a major brand in creating a better future for people living with or caring for those with T1D.”
Driving breakthroughs forward
1 in every 31 families in the U.S. are impacted by type 1 diabetes—it places a tremendous financial, emotional, and mental burden on those who live with it and their loved ones. With support from partners like Marshalls, Breakthrough T1D is making everyday life with T1D safer and easier through technology—like the CGM Kris uses, and automated insulin delivery systems.
Every donation collected through the Marshalls partnership has also helped drive critical research in cures for type 1 diabetes. From the launch of the Special Diabetes Program in 1997, to the first manufactured islets making insulin in 2014, and the FDA approval of Tzield, the first disease-modifying therapy for type 1 diabetes, Breakthrough T1D is driving innovation forward to make the condition a thing of the past.
“The future of type 1 diabetes is bright,” Kris said. “We are on the cusp of living longer, experiencing fewer complications, and enjoying a life without limits.”
Breakthrough T1D thanks Marshalls for its incredible partnership in improving and changing life with type 1 diabetes.
Hy-Vee’s dedication to Breakthrough T1D’s mission is personal.
Type 1 diabetes (T1D) first struck the Hy-Vee family in 1921, when company co-founder Charles Hyde’s oldest son, Paul, died from the disease when he was 8 years old.
Since 1998, Hy-Vee has been a trusted partner of Breakthrough T1D, raising nearly $20 million through a variety of corporate and store events, including Walks in the Midwestern and Southern U.S. Hy-Vee also participates in Rides across the country; in 2025, they were recognized as a top fundraising team, bringing in over $230,000 for T1D research.
Hy-Vee’s 2025 in-store fundraising campaign for Breakthrough T1D runs from September 1 through 30. Find your local Hy-Vee here.
Working together for cures

Sam and Lauren Raiche are loyal Hy-Vee customers. They’re also dedicated fundraisers, advocates, and volunteers for Breakthrough T1D. Their 8-year-old son, Alexander, was diagnosed with T1D at age 3.
They appreciate Hy-Vee’s commitment to Breakthrough T1D’s mission. “It means so much to know that a company we already trust and shop with is also supporting a mission so close to our hearts,” Lauren said. “Living with type 1 diabetes is a daily challenge for our family, and seeing Hy-Vee stand behind research and programs that directly impact families like ours makes us feel grateful and supported every time we walk through their doors.”
Alexander represented Kansas at the 2025 Breakthrough T1D Children’s Congress. As a Delegate, he met with representatives to advocate for renewal of the Special Diabetes Program, which funds critical type 1 diabetes research. “Advocacy has become a cornerstone of our journey,” Lauren said. “We firmly believe that raising awareness and engaging with the community are essential steps toward making meaningful progress and, ultimately, achieving cures.”
Accelerating cell therapy breakthroughs
Support from partners like Hy-Vee helps fuel that progress toward cures for type 1 diabetes. Breakthrough T1D’s cures portfolio includes cell therapies, which replace destroyed beta cells with protected functional cells to restore insulin therapy independence and glucose control, ideally without immunosuppression.
Over the past decade, Breakthrough T1D has funded more than $156 million in cell therapies research, including partnerships with organizations like Vertex Pharmaceuticals and Sana Biotechnology.
In 2024, Vertex launched a pivotal clinical trial for zimislecel (formerly VX-880), which uses manufactured islets to restore the body’s ability to produce insulin. The therapy, however, requires the use of immunosuppression to protect the transplanted cells from rejection. The islets used in zimislecel are derived from the Breakthrough T1D-funded work of Doug Melton, Ph.D., who first turned precursor cells into insulin-producing cells in 2014.
A 2025 study from Sana Biotechnology showed that hypoimmune (HIP) donor-derived islets are making insulin and avoiding immune detection in the first person treated. The T1D Fund: A Breakthrough T1D Venture invested in Sana to help advance their HIP technology platform.
Breakthrough T1D continues to drive innovation to develop cell replacement therapies and eliminate the need for immunosuppression. With the generosity and support of partners like Hy-Vee and families like the Raiches, we will change the lives of everyone facing type 1 diabetes.
Breakthrough T1D is Wawa’s longest-standing partnership. Since 1994, the popular convenience store chain has raised millions of dollars for type 1 diabetes (T1D) research through in-store fundraising campaigns at more than 1,060 locations across the Eastern U.S.
Wawa’s partnership with Breakthrough T1D is further enhanced by donations from The Wawa Foundation, which is committed to helping Breakthrough T1D create a world without T1D and building strong communities by supporting causes related to health, hunger, and everyday heroes.
Wawa’s 2025 fundraising campaign for Breakthrough T1D runs from July 31 to August 20. Customers can donate $1, $3, or $5 or round up at checkout to support life-changing breakthroughs for people living with T1D.
Wawa lives out the values it speaks to
Wawa super fan, Nate Keeney, has lived with T1D for 27 years. He is proud to support a company that lives out the values it speaks to.
“Long before working at Breakthrough T1D, I loved Wawa for their larger selection of food options and snacks,” Nate said. “I always knew they partnered with the organization, but seeing how their efforts impact everyone living with T1D has given me a renewed passion to continue supporting Wawa.”
Wawa walks to cure T1D
Breakthrough T1D Walk is another way Wawa helps support the T1D community and fund critical T1D research. The Wawa Foundation matches employee Walk fundraising efforts up to $100.
Wawa associates also volunteer at Walk events to distribute in-kind food and beverages to participants.
“I see Wawa everywhere at our local Breakthrough T1D Walk in Philadelphia, with their classic soft pretzels and iced teas (and diet iced teas!) to fuel us as we walk to support T1D research and community,” Nate said. “The Goose also leads the T1D parade before the Walk begins.”
Wawa helps fuel life-changing research
Wawa helps Breakthrough T1D advance its mission to accelerate life-changing breakthroughs to cure, prevent, and treat type 1 diabetes and its complications.
That includes technology like continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems, which have been life-changing for Nate.
“My insulin pump and CGM have been a wonderful addition to my diabetes routine—they’ve helped me get my A1c into a great range and identify trends,” he said. “Also, the ability to manage my T1D from my phone has made life on the go easier, whether it’s traveling for work or traveling for fun to different states or countries.”
Wawa’s support also fuels research in cures for type 1 diabetes. Cell therapies that insert healthy insulin-producing cells into people with T1D with minimal or no immunosuppression is a breakthrough Nate is hopeful for.
“So much effort, intensity, and care are going into funding research, particularly with cell therapies,” he said. “It makes me hopeful for cures within my lifetime.”
Together, Wawa and Breakthrough T1D are championing life-changing research and strengthening the T1D community.

The short version
Breakthrough T1D’s newest publication outlines what the future of beta cell replacement therapies looks like—and how we can make these therapies a reality for everyone with type 1 diabetes (T1D) who wants them through innovative clinical trial design and expanding the pool of eligible trial participants.
Breakthrough T1D, in collaboration with other leading experts in the field, recently published an article titled “Future Directions and Clinical Trial Considerations for Novel Islet β-Cell Replacement Therapies for Type 1 Diabetes” in the journal Diabetes. Breakthrough T1D Research and Advocacy staff who contributed to the publication include Sanjoy Dutta, Ph.D., Chief Scientific Officer, Esther Latres, Ph.D., Vice President of Research, and Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs.
Beta cell replacement therapies have the potential to cure type 1 diabetes (T1D) by removing the need for external insulin. While donor islet transplantation can result in insulin independence and results from early trials with manufactured islets are encouraging, people with T1D need better tools and therapies. This publication serves as a roadmap for the entire field—including researchers, product developers, industry, regulators, clinicians, and people with T1D—to address these needs and ensure that beta cell replacement therapies can get to people with T1D as quickly and safely as possible.
Read on to learn more about what the future of beta cell replacement therapies looks like.
Key Takeaways
- Currently available beta cell replacement therapies are limited to people with T1D with unstable blood sugar management, typically measured by HbA1c levels, in addition to dangerous lows (hypoglycemic events) that require immediate assistance. These therapies also require lifelong immunosuppression.
- New and improved beta cell replacement therapies are on the way, and we need to make sure that they are available for people with T1D beyond those with unstable blood sugar management and severe hypoglycemic events.
- Clinical trials for these therapies must be strategically designed to include more people with T1D given the potential for insulin independence.
- Shared decision-making between people with T1D and their care team will be critical for evaluating the risks versus benefits of a beta cell replacement therapy, especially as more options become available.
Where we are now
People living with T1D depend on external insulin to manage their condition throughout their lives. Despite advancements in diabetes technology, such as continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems, most people with T1D are unable to achieve blood sugar targets, rendering them at a higher risk for complications, reduced quality of life, and lower life expectancy.
It’s simple: we need to do more for the T1D community.
One promising avenue to meet these needs is through cell replacement therapy. Lantidra®, the first FDA-approved donor-derived islet replacement therapy for T1D, has proven to be safe and effective in eliminating severe hypoglycemia, providing insulin independence, and improving quality of life. However, this therapy is limited to people with severe hypoglycemia and requires immunosuppression to prevent rejection of the transplanted cells, which can have side effects. Even more, these donor-derived islets are in limited supply.
Alternative beta cell replacement therapies are emerging—and we can no longer limit their development to people experiencing severe hypoglycemia.
As stated in the publication:
Given the proven benefits of islet transplantation extending far beyond the amelioration of severe hypoglycemia that has been documented and the understanding of the risk profile gained over the past 20 years, consideration must be given to broadening the application for islet beta cell replacement.
Developing cell therapy strategies to meet unmet needs of the T1D population
Breakthrough T1D’s vision for the T1D community includes beta cell replacement therapies with no immunosuppression that are available to everyone who wants them. We are committed to making this a reality through our Project ACT (Accelerate Cell Therapies) initiative, which will accelerate the development of these therapies to achieve our vision as quickly as possible.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
We are entering an exciting era of beta cell replacement. Emerging therapies are addressing challenges such as cell source and scalability, resulting in the development of islets derived from sources other than donors, including manufactured islets and porcine islets. Up-and-coming therapies are also testing different transplantation sites, methods of delivery, and cell protection strategies to prevent immune rejection with the fewest side effects possible (and ideally no immunosuppression). Learn more about what scientists are doing to optimize beta cell replacement therapies.
Breakthrough T1D’s continuous support of many of these therapies, such as Vertex’s manufactured islet therapy zimislecel, has been critical to accelerating them through the clinical pipeline. Explore emerging beta cell replacement therapies in clinical trials now—and see how Breakthrough T1D’s commitment to these therapies helped make this possible.
When successful, the advent of new, safe, scalable, and effective beta cell replacement therapies will provide the T1D community with options. As these therapies are moving their way through the pipeline, we need to ensure they are being studied in a broader T1D population who stand to benefit.
So, how do we make this happen?
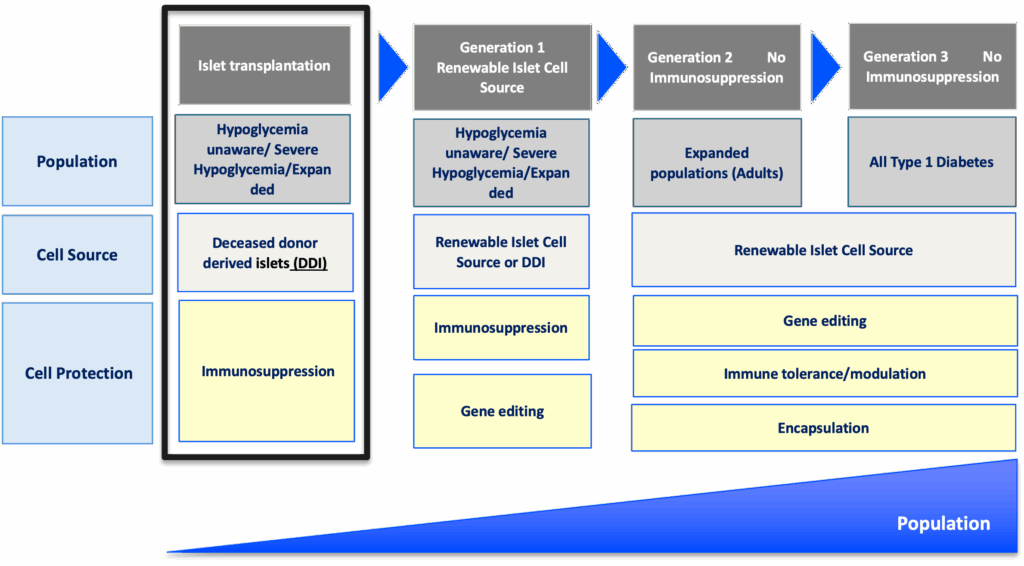
The roadmap for emerging beta cell replacement therapies
The goal
Accelerate availability of emerging next-generation beta cell replacement therapies for every person with T1D who wants them by designing clinical trials that speed their development, regulatory approval, access, and adoption.
Expanding the T1D population eligible for beta cell transplantation
Current trials testing beta cell therapies necessitating immunosuppression require participants to have elevated HbA1c levels and recurrent severe hypoglycemic events. This limits the pool of participants to people who meet the requirements and are most likely to benefit, given the side effects associated with chronic, broad immunosuppressants.
Clinical trials for emerging beta cell replacement therapies should broaden eligibility criteria so more people with T1D can participate—and experience the potential benefits. When designing new clinical trials, sponsors and regulators should consider including a broader range of HbA1c levels, clinically important or serious hypoglycemic events, and other complications.
Studies have found that the T1D community is generally open to beta cell replacement therapies as a potential solution to T1D, and people are willing to accept the associated risk versus benefit considerations for the possibility of becoming insulin independent. A Breakthrough T1D assessment also found that physicians are interested in recommending beta cell replacement therapies to people with T1D—especially if they don’t require immunosuppression.
“Clinical trials to support the development of islet cell replacement therapies need to evolve to include a broader representation of people living with T1D who could benefit from these novel therapies. This includes expanding the outcomes used to assess the benefits of cell replacement that reflect how people with T1D feel and function.”
Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs

Placing people with T1D at the center of clinical trial design
The outcomes used to assess the effectiveness of cell therapies currently in clinical trials, including those involving deceased donor islets, are acceptable for emerging beta cell replacement therapies. These include on-target HbA1c levels, absence of severe hypoglycemia, significant reduction or elimination of external insulin, and restoration of the body’s insulin production as measured by C-peptide.
Other endpoints that should be considered include CGM metric targets like time-in-range in addition to person-reported outcomes. Understanding how a beta cell therapy may affect a person’s health-related quality of life—such as diabetes distress, fear of hypoglycemia, or social and family dynamics—will be critically important for calculating the risk to benefit ratio of these therapies.
Read more about why person-reported outcomes are important for cell therapies.

“There are still significant unmet needs in the T1D community. Breakthrough T1D’s roadmap is supported by the assessment of clinically meaningful outcomes and driving research toward solutions that address key factors such as cell sources and protections strategies that will broaden the people with T1D who could benefit from emerging cell replacement therapies.”
Esther Latres, Ph.D., Vice President of Research
Innovative trial designs to accelerate development of cell therapies
Clinical trials for beta cell replacement therapies are generally based on a single-arm, open-label design—meaning there is no placebo group and both participants and researchers know which therapy is being administered. While this design can work for emerging beta cell therapies, single trials with multiple arms testing alternative transplant sites, immune protection strategies, or other methods have the potential to speed up the pace of development.
Similarly, adaptive trial designs use mid-trial interim analyses of study data to inform the remainder of the trial. This helps researchers learn what’s working (or not working) and adjust the design accordingly, with guidance from regulatory agencies, so the rest of the trial is a focused and efficient use of time and resources. Potential interim changes to trial design include reducing the number of participants required, eliminating doses, recruiting people who are most likely to benefit, or stopping the trial outright due to clear success or failure.
By applying guidance in therapeutic development and innovative trial designs to emerging beta cell replacement therapies, we can move early-stage trials along faster, thereby allowing regulators to make decisions sooner. To support quicker trials and reduce the possibility for delays, researchers, developers, and regulators around the world need to work together to achieve convergence on trial populations, endpoints, and innovative designs that will meet regional requirements.
Learning from past successes
People with T1D continue to live with unmet needs with still significant risk for long-term complications, and they need more therapeutic options. Right now, most clinical trials for beta cell replacement therapies requiring immunosuppression are limited to a small portion of the T1D population. This needs to change—especially given the potential for insulin independence. The T1D community must be put first when making decisions about beta cell replacement therapies, and Breakthrough T1D is making sure that this happens.
Adjusting how we approach clinical trials for emerging beta cell replacement therapies will be critical for ensuring we accelerate the research, development, regulatory approval, access and adoption of these novel therapies. Breakthrough T1D successfully accomplished this for AID systems—and we are confident that following a similar roadmap for cell therapies will get us to the finish line, faster.
The journey of AID systems
Learn more about the critical role of Breakthrough T1D in driving AID systems forward, recounted by Breakthrough T1D volunteer Doug Lowenstein.
Curative therapies for T1D are in reach. This roadmap, in conjunction with our Project ACT initiative, is key to bringing beta cell replacement therapies to every person with T1D who wants them.
To make this a reality, everyone needs to work together. As stated in the publication, “This requires a comprehensive strategy and a coordinated collaboration across stakeholders in every field relevant to islet cell replacement.” This roadmap is a guide for moving toward our common goal of a cure for T1D as soon as possible.
Breakthrough T1D will continue to lead the way until the T1D community can choose the beta cell replacement therapy that works best for them, regardless of blood glucose management or hypoglycemia status. Everyone deserves the chance to benefit.
Breakthrough T1D, in partnership with Friends of Mewar and UNICEF, participated in the Udaipur Type 1 Diabetes Summit: Advancing Access, Equity, and Action, in Udaipur, Rajasthan, India. The event gathered government leaders, health experts, community advocates, and international partners to develop a collaborative roadmap for strengthening type 1 diabetes (T1D) care across India.
Breaking down barriers and stigma
Hosted by Friends of Mewar’s Founder and Breakthrough T1D Global Ambassador Princess Padmaja Kumari Parmar, who has lived with T1D for more than 40 years, the two-day summit was a critical milestone in building partnerships that drive sustainable, system-wide improvements in T1D care and equity.
“Timely healthcare access and accurate information are vital for managing type 1 diabetes and other non-communicable diseases,” said Princess Padmaja. “Collaboration is key to breaking down barriers and stigma, and ensuring that children and youth receive the support they deserve.”
Topics discussed included integrating T1D into national health frameworks, scaling digital and rural care solutions, promoting equity and stigma reduction, and aligning regional action across Southeast Asia. The summit also featured updates on the T1D Basic Care Pilot Program, a partnership between Breakthrough T1D and the William J. Clinton Foundation, showcasing early successes in improving insulin and glucose monitoring availability in public health settings.
The Udaipur Type 1 Diabetes Summit concluded Princess Padmaja’s strong call for sustained innovation, investment, and partnership to address the urgent need for support for youth living with T1D in India and around the world.
Advancing cures while improving lives
The summit aligned with Breakthrough T1D’s mission of advancing cures while improving lives. Affordable insulin, access to T1D technology, early diagnosis to prevent complications at diagnosis, and psychosocial support are all critical to improved health outcomes on the path to cures.
“In India and many other countries, children and young people face significant barriers to care,” said Lynn Starr, Chief Global Advocacy Officer at Breakthrough T1D. “Our commitment is that when cures arrive, they are accessible to everyone, everywhere, regardless of geography or income.”
Driving change for the T1D community

Born in Udaipur, Rajasthan, India, Princess Padmaja was diagnosed with T1D at age 5. At the time of her diagnosis, tools and insulin options for managing T1D were limited—checking blood glucose levels or dosing insulin based on carbohydrate quantities wasn’t even possible.
“Witnessing the progression of T1D breakthroughs over the years has been nothing short of remarkable,” said Princess Padmaja. “When I first started insulin injections, it was a cumbersome routine, requiring multiple injections a day. Today, thanks to advancements in insulin pump technology, managing T1D has become more streamlined and efficient, improving my quality of life.”
As a Breakthrough T1D Global Ambassador, Princess Padmaja is working to combat the stigma of T1D and address the challenges surrounding the condition. She hopes to inspire changes within the government’s policies that could support people with T1D and within India’s healthcare system.
“As a Global Ambassador, my aspirations do extend beyond borders,” she said. “The challenges surrounding T1D are not exclusive to India; they are present globally.”
“By rallying support and implementing these measures, I can help tackle these challenges, ultimately improving the quality of life for those with T1D.”
Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’ve reported on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding. See below for all our ADA 2025 coverage spanning cures, improving the lives of people with T1D, and clinical adoption of T1D therapies, treatments, and devices.
Improving Lives
Medical Affairs
ADA recap with Aaron Kowalski, Ph.D.
Looking for more ADA news? Tune in below to hear Breakthrough T1D Chief Executive Officer Dr. Aaron Kowalski cover the top advancements presented at ADA 2025.
Thanks for joining us throughout our coverage of the ADA 85th Scientific Sessions. We’ve made incredible progress across all our research priorities areas, including cures therapies, treatments, and devices. Breakthrough T1D staff and leadership were on-site hosting and participating in panel discussions, meeting with industry leaders, and engaging with researchers from around the world to accelerate T1D research progress. Breakthrough T1D-funded research was front-and-center, showcasing the reach of our impact driven by supporters like you. That’s a wrap on ADA 2025—we’re already looking forward to ADA 2026!

ADA Recap Series
This article is the last of our three-part ADA Recap Series. Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’re here to report on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding.
Medical Affairs (+Research and Advocacy)
Breakthrough T1D’s Medical Affairs program is bridging the gap between access to and adoption of therapies, treatments, drugs, and devices for T1D. To accomplish this goal, the Medical Affairs team is working on healthcare provider (HCP) education, development of clinical care guidelines, and clinical trial education and awareness.
Breakthrough T1D’s Research and Advocacy teams work closely with Medical Affairs to accomplish our shared goal of bringing the most promising therapies and treatments to the T1D community as fast as possible. Below, you’ll find Breakthrough T1D staff and leadership at ADA working together across each of our organization’s priority areas to share knowledge, spark discussions, drive innovation, and ultimately move us closer to achieving our mission.
Guidelines for T1D management
Guidelines for cardiovascular and kidney disease in T1D
- Presenter: Camila Manrique-Acevedo, M.D.; University of Missouri
- Cardiovascular disease (CVD) risk has decreased in people with T1D over time, but it is still the leading cause of death in people with T1D, representing a critical unmet need.
- Treatment recommendations and guidelines have largely relied on evidence from studies in T2D.
- Dr. Manrique-Acevedo recommends lipid-lowering medication (like statins such as Lipitor®) to reduce LDL to target levels. Based on an observational study, this class of drugs can reduce CVD risk by 22% in people with T1D.
- She also recommends lifestyle modifications, blood pressure management, weight management, and early and proactive screening for CKD.
- Development of clinical guidelines is a priority of Medical Affairs. The guidelines presented here are not published and do not necessarily represent the consensus of experts in the field. However, Dr. Manrique-Acevedo started the conversation to standardize CVD clinical care for people with T1D, which currently remains an unmet need.

Continuous ketone monitoring (CKM): Why does it matter?
CKM technology can have transformative benefits for intervening early when ketones are rising to prevent diabetic ketoacidosis (DKA), among other uses. Breakthrough T1D has been supporting the development of CKM technology, but it’s not quite a reality yet. However, we’re working hard to bring CKM to the T1D community, and we believe we’ll be there soon.
Consensus guidelines highlight: Breakthrough T1D staff making an impact
Clinical adoption of CKM
Development and clinical adoption of CKM will be a crucial step toward preventing DKA in people with T1D. Thomas Danne, M.D., Ph.D., Chief Medical Officer, Global, convened an International Breakthrough T1D Consensus Group to report on the application and utility of CKM for people with T1D. The report was submitted for publication just before ADA 2025. During the Symposium titled “Continuous Ketone Monitoring – Innovations and Clinical Applications,” five members of the Breakthrough T1D Consensus Group chaired and presented on how the initiative will impact the clinical adoption of CKM. The presentations covered the basics of ketones, self-monitoring of ketone levels with CKM, new data emphasizing the potential of CKM impact, and the importance of shared decision-making between HCPs and people with T1D—all of which were outlined in the consensus document. Development of CKM consensus guidelines is a critical first step toward clinical adoption of CKM so the T1D community can benefit.


Key takeaways
Guidelines for T1D management, especially clinical consensus guidelines that have been built by international experts, are key to advancing clinical adoption of the newest therapies, devices, and treatments for T1D and its complications. CKM consensus guidelines are coming soon!
Education and awareness


Precision medicine
Precision medicine refers to therapies and treatments that are targeted to a specific population of people based on genetics or other factors. This subpopulation is likely to respond more favorably to the precision medicine intervention compared to the general population.
Education and awareness highlight: Breakthrough T1D staff making an impact
Supporting the role of women in the T1D clinical care team
Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, Vice President of Medical Affairs, participated in a panel discussion about women in diabetes professions. She gave an overview of her personal career journey, providing valuable insight into the benefits of mentorship, training initiatives designed for career growth, fostering leadership for women, and developing supportive networks. The panel highlighted disparities such as lower numbers of women attending conferences and occupying leadership roles in diabetes professions. By understanding barriers and amplifying the achievements of women in the diabetes workforce, we can foster a culture of inclusion, provide women with opportunities for visibility and leadership, and ultimately create a stronger care team for people with diabetes.
Precision medicine for T1D
Joshua Vieth, Ph.D., Senior Director of Research, co-hosted a panel discussion with Ulf Hannelius, Ph.D., MBA, Chief Executive Officer of Diamyd Medical, titled “Precision in Diagnosis, Power in Treatment: The Future of Type 1 Diabetes.” Stephen Karpen, Pharm.D., Senior Director of Regulatory Affairs, was a panelist. The panel also included Laura Jacobson, M.D. (University of Florida), Emily Sims, M.D. (Indiana University), Jason Gaglia, M.D. (Harvard University), and Alecia Wesner (person with T1D).
The panelists discussed a range of topics, including how precision medicine can change the treatment landscape for disease-modifying therapies and practical challenges that people with T1D will face, such as coverage of screening to determine eligibility for precision medicine-based therapies.
A significant focus of the panel discussion was clinical trial education and awareness. Recruiting for trials that may only work for a portion of people with T1D may prove difficult—and it’s critical that HCPs communicate why a certain therapy may or may not be a great fit for an individual. Additionally, the panelists suggested that clinical trial education should be built into training for diabetes specialists and endocrinologists so they can appropriately talk with patients and share the most important information. As Dr. Sims put it, we need a “precision approach to talking about precision medicine.”
Speaking on behalf of the perspective of people with T1D, Ms. Wesner stressed the importance of educating not only HCPs but the T1D population about clinical trials, including those based on precision medicine. She discussed the impact of communicating the emotional side of clinical trial participation to people with T1D and how their decision to volunteer could benefit the T1D community as a whole.
See below for a recording of the panel discussion.
DiabetesMine #DData ExChange event
This event brought together researchers, industry partners, clinicians, and regulatory specialists to discuss a range of diabetes topics. Esther Latres, Ph.D., Vice President of Research, hosted a panel discussion with industry titled “Headway and Hurdles: How can Diabetes Cell and Gene Therapy Succeed?” The panelists discussed the promise of cell therapies and role of Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) in making these therapies a reality for people with T1D, especially those that do not require immunosuppression. Getting the word out about our work toward curative cell therapies—and discussing solutions to the challenges we’ll face bringing them from labs and into clinics—is a cornerstone of Breakthrough T1D’s Medical Affairs strategy.

Breakthrough T1D Advocacy shout-out
Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs, participated in a panel titled “Influencing Health Policy in 2025: A New World Order?” The discussion focused on the policy and regulatory environment surrounding funding for diabetes research, including the critical importance of continued renewal of the Special Diabetes Program. The panel also discussed the need to emphasize the voices of people with T1D in developing cures.

Key takeaways
Education and awareness—from HCPs, to people with T1D, to researchers—is essential for clinical adoption of new therapies that can benefit the T1D community. At multiple events that Breakthrough T1D staff led or participated in, diverse audiences learned about precision medicine, health policy, cell therapies, and supporting women on T1D care teams.
From T2D to T1D: Shortening the clinical trial timeline for repurposed drugs

Biomarker
A biomarker is a measurable change that is indicative of disease or response to a therapeutic intervention.

Bridging biomarker
A bridging biomarker can be used to extrapolate the effectiveness of a treatment from one indication or population to another. In this case, a bridging biomarker that was effective in measuring therapeutic benefit in type 2 diabetes (T2D) can be used to measure the same benefit in T1D.
Finerenone for T1D
- Presenter: Hiddo Heerspink, Ph.D.; University Medical Center Groningen
- Finerenone has proven effective in preventing progression of chronic kidney disease (CKD) in T2D.
- The phase 3 FINE-ONE clinical trial, which is testing whether finerenone can prevent progression of CKD in people with T1D, is nearly complete. The treatment period is only six months.
- The primary endpoint is change in urinary albumin-to-creatinine ratio (UACR), an indicator of kidney damage. UACR is being used as a bridging biomarker for this trial based on demonstrated effectiveness of predicting finerenone-driven kidney benefits in T2D.
- This has reduced the treatment time—and therefore clinical trial length—by a significant amount. Shorter clinical trials mean people may be more inclined to participate, and HCPs need to effectively communicate this to interested volunteers.
- This is also an important lesson in clinical trial design and the benefits of repurposing drugs for T1D that have already shown benefits in T2D.
Key takeaways
Shortening clinical trial timelines will be critical to getting groundbreaking therapies from clinical testing to people with T1D faster. We can learn lessons from treatments that work in T2D and repurpose them for T1D to make this possible. Encouraging HCPs to discuss shorter clinical trial options with interested trial participants will be essential to recruitment, which in turns moves trials along even faster.
The ADA 85th Scientific Sessions have officially come to a close—and we were inspired and energized by the incredible advancements we saw first-hand across cures therapies, treatments, devices, and Medical Affairs initiatives. Thanks to all the researchers, scientists, clinicians—including many who were funded by Breakthrough T1D—who joined the discussion and shared the exciting T1D research progress from labs and clinics around the world. See you next year!

ADA Recap Series
This article is the second of our three-part ADA Recap Series. Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’re here to report on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding. Look out for tomorrow’s article for updates on Medical Affairs.
Cures
Breakthrough T1D’s Cures program focuses on early detection, disease-modifying therapies, and cell therapies with the goal of working toward effective cures for T1D.
Cell therapies were front-and-center at ADA 2025. We have some exciting clinical trial updates and new ideas for optimizing islet transplantation.
Cell therapies

Autologous cell transplantation
Autologous cells are those removed from an individual and implanted back into the same individual. These cells can be modified in a laboratory before implantation. Autologous cells are still susceptible to autoimmunity in T1D, so cell protection strategies (gene-editing, encapsulation, immune modulation, etc.) are expected to be required.

Allogenic cell transplantation
Allogenic cells are those that are derived from a source other than the recipient, such as deceased donors or precursor-derived manufactured cells. Allogenic cell transplants require immunosuppression because they stimulate an immune response. Breakthrough T1D’s Cell Therapies program is focused on allogenic cells—specifically manufactured cells—because they can be generated at large scale.
One-year updates on Vertex’s manufactured cell therapy, zimislecel
- Presenter: Michael Rickels, M.D. (University of Pennsylvania)
- Zimislecel (VX-880) is a manufactured islet therapy that requires immunosuppression, infused into a vein in the liver in people with T1D who have impaired hypoglycemic awareness and severe hypoglycemic events.
- The phase 1/2 clinical trial, which is part of the pivotal phase 1/2/3 FORWARD-101 trial, is complete. Twelve participants received a single infusion of a full dose of cells and were followed for at least one year.
- All 12 participants achieved the primary endpoint, which was elimination of severe hypoglycemic events and HbA1c levels less than 7%. 10/12 (83%) participants are insulin independent.
- All 12 participants demonstrated sustained insulin production as measured by C-peptide, reduced external insulin therapy use, and achieved greater than 70% time in range.
- There were no serious adverse events. Mild to moderate adverse events were consistent with the immunosuppression regimen, infusion procedure, and complications from T1D.
- These data were published in the New England Journal of Medicine and represent further evidence of the curative potential of manufactured islet transplantation for T1D.
- Breakthrough T1D’s support for Doug Melton, Ph.D.—whose proprietary lab-created beta cells are now being advanced by Vertex—goes back decades, both via research grants and an investment from the T1D Fund: A Breakthrough T1D Venture.
6-month update on Sana Biotechnology’s immune-evasive islets
- Presenter: Per-Ola Carlsson, M.D., Ph.D. (Uppsala University)
- Sana’s donor-derived islet therapy engineered with Hypoimmune (HIP) technology can evade the immune system without immunosuppression.
- These cells were implanted intramuscularly in a first-in-human study into a person with T1D with no measurable insulin production.
- Six months post-transplant, this person is consistently making their own insulin, as measured by C-peptide levels. Yet, they still require external insulin therapy because they received a smaller dose of cells than the dose that would be required to achieve insulin independence. They did not experience any serious side effects, so the cells and procedure are safe and well-tolerated.
- A Mixed Meal Tolerance Test (MMTT) confirmed that these cells are not only surviving but also responding to changes in blood glucose levels.
- This is a promising first step toward a functional cure for T1D that does not require immunosuppression. Sana Biotechnology is planning on applying this technology to manufactured islets.
- Sana has received support from the T1D Fund to advance their HIP technology in islets, and Breakthrough T1D continues to work closely with them.
A new transplantation site for autologous manufactured islets
- Presenter: Hongkui Deng, M.D. (Peking University)
- Cells derived from adipose tissue (fat) can be removed from a person and chemically induced in the laboratory to become islet cells.
- Implantation of autologous manufactured islets into the sub-anterior rectus sheath in preclinical models of T1D improves glycemic control.
- In humans, this implantation site is easily accessible by an ultrasound-guided needle.
- In a first-in-human study, autologous manufactured islets were implanted into this site in a person with T1D. This person no longer needs external insulin therapy and has greatly improved blood glucose control. This person had also received a liver transplant and was taking immunosuppressants.
A new encapsulation device for immune protection of transplanted islets
- Presenter: Nicolas Laurent, Ph.D. (Adocia)
- Adoshell® is a novel islet cell encapsulation device that can shield islets from the immune system, meaning that immunosuppressants are not needed.
- The hydrogel-based device is non-degradable, easily retrievable, and allows the exchange of glucose and insulin from the vasculature surrounding the device while excluding immune cells from encapsulated islets based on pore size.
- This device showed promise in animal models, and human clinical testing is next.

Cell therapies highlight: Breakthrough T1D-funded research
Tom Bollenbach, Ph.D. (Advanced Regenerative Manufacturing Institute; ARMI) presented on challenges and solutions for large-scale manufacturing of islet cells. The goal is to generate scalable, automated manufactured islets that can be used by labs around the world to accelerate research progress using a unique, validated, and reliable cell source. Some challenges include ensuring the manufactured cells can survive shipment from the facility to research labs and maintain their insulin-producing capacity when they are used in different labs. ARMI is working with the Beta Cell Replacement Consortium to address these challenges.
Antonio Citro, Ph.D. (San Raffaele Hospital) presented his work on ensuring that transplanted islets have enough oxygen and nutrients (“vascularization”) to survive and function. Dr. Citro described an approach tested in animal models referred to as “natural scaffolds” in which all cells are removed from a donor organ, such as a lung, leaving behind blood vessel structures and other structural components. Islet cells can be injected and grafted onto this scaffold to create a mini organ of islet cells, which can then be tested for functionality before and after implantation.
Andrew Pepper, Ph.D. (University of Alberta) also presented vascularization strategies to increase the survival and functionality of transplanted islets. The implantation of a biomaterial under the skin will trigger the immune system and initiate a foreign body reaction, which results in the formation of blood vessels and structural components around the foreign object. Removal of the object leaves a hollow pre-vascularized core suitable for islet transplantation. This process can be optimized by using a biodegradable material, so no removal is required, and by the addition of “accessory cells” that help maintain a vascularized environment for islets.
Key takeaways
Cell therapies are making significant headway in clinical trials, and people receiving manufactured cells are becoming insulin independent. Researchers are tackling the biggest challenges for optimizing islet transplantation, including large-scale manufacturing, ensuring cell survival, and preventing detection by the immune system.
Disease-modifying therapies
A major focus at ADA 2025 was addressing the underlying immune mechanisms of T1D—including alterations in immune cells that facilitate beta cell destruction and other factors that contribute to autoimmunity onset. Read on for some highlights.
The role of B cells in T1D autoimmunity
- Presenter: Mia Smith, Ph.D., DVM (University of Colorado)
- B cells are a type of immune cell that can activate destructive immune cells that facilitate autoimmunity in T1D.
- B cells can become wrongly activated against insulin-producing beta cells due to converging dysregulation of factors that regulate immunity.
- These cells represent another potential target for disease-modifying therapies in T1D.
Disease-modifying therapies highlight: Breakthrough T1D-funded research
Emrah Altindis, Ph.D. (Boston College) presented on the role of the gut microbiome in T1D. His studies found that people with T1D tend to have more inflammatory bacteria in their gut microbiome. Dr. Altindis and his team identified a particular bacterial population that can enhance T1D onset in animal models due to changes in immune cells that contribute to T1D autoimmunity. These studies provide insight into additional factors that can drive immune changes in T1D.
Laura Sanz Villanueva, MSc (St. Vincent’s Institute of Medical Research), who works in the lab of Breakthrough T1D-funded researcher Professor Thomas Kay, MBBS, Ph.D., presented on a mechanistic follow-up study to the BANDIT clinical trial. The Breakthrough T1D-funded phase 2 BANDIT study showed that baricitinib, a JAK1/2 inhibitor that prevents immune cell communication, can increase insulin production as measured by C-peptide in people with recently diagnosed T1D. The present study found that baricitinib can reduce the number of natural killer (NK) cells in the pancreas, which are involved in the autoimmune destruction of beta cells. These data provide valuable insight into the mechanism of baricitinib-mediated protection of beta cells.
Key takeaways
T1D is driven by dysregulation of the immune system, which results in an attack on insulin-producing beta cells. Researchers at ADA 2025 spoked about novel factors—including B cells, NK cells, and T1D autoimmunity triggers—that may contribute to immune cell dysfunction in T1D.
Early detection
A key focus at ADA 2025 was the growing recognition of the heterogeneity of T1D, including autoantibody-negative disease onset, genetic variation, and the frequent misdiagnosis of T1D in adults, underscoring the need for greater diversity and inclusion in research and care. The expanded role of continuous glucose monitoring (CGM) and continuous ketone monitoring (CKM) was also highlighted, not only for daily management but as essential tools for understanding disease progression.
Using genetics to predict T1D risk
- Presenters: Richard Oram, M.D., Ph.D. (University of Exeter), Leslie Lange, Ph.D. (University of Colorado), Aaron Deutsch, M.D. (Massachusetts General Hospital), Josep Mercader, Ph.D.(Massachusetts General Hospital) and Eimear Kenny, Ph.D. (Icahn School of Medicine at Mount Sinai)
- Polygenic risk scores (PRS) estimate the risk a person has for developing a disease like T1D based on variations in different genes.
- Ancestry is a major influence on PRS, particularly based on differences in genes that regulate whether the immune system can distinguish between “self” and “non-self.”
- Most PRS models have been developed using data from European populations and have a limited ability to accurately determine risk in other ethnic groups, such as individuals of African and East Asian descent.
- Potential applications of PRS include incorporation into screening to better understand T1D risk, ensure accuracy in diagnostic tests, and develop precision medicine-based therapeutic approaches.
Understanding how genetic diversity contributes to T1D
- Presenters: Suna Onengut-Gumuscu, Ph.D. (University of Virginia), Dominika A. Michalek, MS (University of Virginia), Aaron Deutsch, M.D. (Massachusetts General Hospital), and Stephen I Stone, M.D. (Washington University School of Medicine), among others.
- These talks highlighted several studies conducted in diverse populations to better understand the pathophysiology of T1D.
- Work presented from Consortia, such as RADIANT, focused on rare and atypical forms of diabetes.
Controversies in CGM and benefits for early detection
- Presenters: Peter Calhoun, Ph.D. (Jaeb Center for Health Research), Michael Kohn, M.D., MPP (University of California San Francisco), Nicole Ehrhardt, M.D. (University of Washington) and Tadej Battelino, M.D., Ph.D. (University of Ljubljana)
- CGM use holds value in identifying progression in early stages of T1D prior to symptomatic onset.
- There was a call to update the clinical guidelines so that the benefits of CGM can be maximized within the T1D community—including at early and later stages of T1D.
- Integrating newer measures of blood glucose, like the glucose management indicator (GMI) and time in tight range (TITR), will be essential.
Contributions of CKM to early detection
- Presenters: Ketan Dhatariya, MBBS, M.D., Ph.D. (Norfolk and Norwich University Hospitals), Lori Laffel, M.D., MPH (Harvard University), Jennifer Sherr, M.D., Ph.D. (Yale University), and Richard Bergenstal, M.D. (HealthPartners Institute).
- It will be critical to explore whether ketone monitoring could help reduce the incidence of diabetic ketoacidosis (DKA) at stage 3 clinical T1D onset.
- Early detection of rising ketones will be important for people with T1D to take action before DKA occurs.
Early detection highlight: Breakthrough T1D-funded research and awardees
Brigitte Frohnert, M.D., Ph.D. (Barbara Davis Center for Diabetes), co-investigator with Breakthrough T1D-funded researcher Andrea Steck, M.D., presented on the evolution of CGM patterns prior to stage 3 T1D. Their longitudinal approach, with CGM data collected at three-month intervals, successfully distinguished individuals who progressed to stage 3 T1D clinical onset from those who did not. This research suggests that CGM may be beneficial for predicting clinical progression of T1D.
Anette-Gabriele Ziegler, M.D. (Helmholtz Munich) was awarded the Harold Hamm Prize for Biomedical Research in Diabetes 2025, highlighting her important contributions on T1D screening in the pediatric population.
Barbara B Kahn, M.D. (Beth Israel Deaconess Medical Center and Harvard Medical School) received the 2025 Albert Renold Award, and her role model for women in science was highlighted at the Women’s Interprofessional Network of the ADA (WIN ADA).
Key takeaways
T1D arises in diverse ways across age groups and ancestries, complicating diagnosis and treatment. At ADA 2025, experts highlighted how genetic and clinical heterogeneity demands more inclusive strategies. Tools like CGM and CKM were highlighted for their potential to enhance clinical management in the early stages of T1D.

Breakthrough T1D’s Cures team making an impact
Sanjoy Dutta, Ph.D., Chief Scientific Officer, participated in a panel discussion titled “Encapsulation vs. Naked Cell Therapy—Immune Challenges and Beta-Cell Perspectives in Diabetes Treatment.” The panelists covered different approaches to preventing immune attack of transplanted islets, including gene-editing, in addition to pros and cons of different transplantation sites.
Esther Latres, Ph.D., Vice President of Research, and Jay Tinklepaugh, Ph.D., Senior Scientist, hosted a workshop immediately after ADA titled “Islet Cells in T1D.” Breakthrough T1D-funded researchers gathered from around the world to discuss cell therapy clinical trial updates, mechanistic insights into islet function, targeted delivery of therapeutics to beta cells, and optimization strategies for islet transplantation.
Look out for tomorrow’s article for an update on Medical Affairs presented at ADA 2025!

ADA Recap Series
This article is the first of our three-part ADA Recap Series. Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’re here to report on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding. Look out for tomorrow’s article for updates on Cures.
Improving Lives
Breakthrough T1D’s Improving Lives program focuses on devices, insulins, adjunctive therapies, treatments for complications, and psychosocial interventions to improve the health and quality of life of people living with T1D.
Adjunctive therapies and complications
There was significant focus on GLP-1 receptor agonists (GLP-1RAs) and SGLT inhibitors (SGLTi) in reducing long-term complications and improving glycemic control in people with T1D.

GLP-1 receptor agonists
Glucagon-like peptide 1 receptor agonists mimic the hormone GLP-1, which elevates insulin and regulates appetite. Examples include Ozempic® (semaglutide) and Mounjaro® (tirzepatide), which acts on both GLP-1 and a similar target, GIP.

SGLT inhibitors
Sodium-glucose cotransporter inhibitors target kidney cells to prevent them from reabsorbing glucose into the blood so it gets excreted as waste. Examples include Farxiga® and Zynquista®.
While SGLTi and GLP1-1RAs have proven effective for heart and kidney disease in type 2 diabetes (T2D) and in people without diabetes, people with T1D have often been excluded from critical trials. Thanks to years of advocacy and support from Breakthrough T1D, T1D trials are ongoing—and real-world evidence suggests that GLP-1RAs and SGLTi could be impactful in the T1D community as well.
Real-world evidence for GLP-1RA use in T1D
- Presenter: Ildiko Lingvay, M.D.; University of Texas Southwestern
- People with T1D have self-reported that they decided to try GLP-1RAs for weight loss and improved glycemic control.
- Real-world evidence suggests that GLP-1RAs have a clinically meaningful impact on weight and reduced insulin dose.
- While GLP-1RAs are generally safe, some people have stopped use because of gastrointestinal side effects. These side effects are also seen in people with T2D and people without diabetes.
A review of SGLTi and GLP-1RAs in reducing chronic kidney disease (CKD) in T1D
- Presenter: David Cherney, Ph.D.; University of Toronto
- In the EMPA-KIDNEY trial that included non-diabetes participants and people with T1D or T2D, empagliflozin (SGLTi) improved kidney health in people with T1D.
- In the ATTEMPT trial, dapagliflozin (SGLTi) improved time in range (TIR), reduced HbA1c levels, and had positive effects on kidneys in youth with T1D.
- The Breakthrough T1D-funded enrolling phase 3 SUGARNSALT trial is testing whether sotagliflozin (SGLTi) can prevent progression of moderate to severe kidney disease in people with T1D, and it includes careful diabetic ketoacidosis (DKA) risk mitigation strategies.
- The SEMA-AP trial found that semaglutide (GLP-1RA) increases TIR in people with T1D when used alongside an AID system.
- The Breakthrough T1D-funded recruiting phase 2 REMODEL-T1D trial is testing if semaglutide (GLP-1RA) can improve kidney health in people with T1D.

Glucokinase
Glucokinase (GK) is an enzyme in liver cells that works in an insulin-dependent manner to regulate blood sugar. In people with T1D who have little insulin reaching the liver, GK can’t work as normal, contributing to higher blood sugar.
Use of a glucokinase activator for glycemic control
- Presenter: Klara Klein, M.D., Ph.D.; University of North Carolina
- In the phase 1/2 SimpliciT1 study, people with T1D who received the GK activator TTP399 showed improvements in blood glucose with fewer hypoglycemic events.
- A different study found that TTP399 does not increase the risk for DKA.
- These studies were done in collaboration with vTv Therapeutics, a company with funding and support from Breakthrough T1D and the T1D Fund: A Breakthrough T1D Venture. The phase 3 CATT1 study for TTP399 is testing whether it can reduce moderate to severe hypoglycemic events in people with T1D.
Adjunctive therapies and complications highlight: Breakthrough T1D-funded research
Halis Kaan Akturk, M.D. (University of Colorado), Janet Snell-Bergeon, Ph.D., MPH (University of Colorado), and Viral Shah, M.D. (Indiana University) presented findings from the Breakthrough T1D-funded ADJUST-T1D clinical trial, which was recently published in the New England Journal of Medicine Evidence. The trial tested whether semaglutide (GLP-1RA) can improve glycemic and weight outcomes in adults with T1D and obesity who are using an AID system. 36% of people treated with semaglutide met the primary endpoints of TIR greater than 70%, time below range less than 4%, and weight loss of 5% or more compared to the placebo, and the drug was well-tolerated and safe. This trial represents critical evidence for use of a GLP-1RA as a potential way to manage both glycemic control and weight in people with T1D.
Ye Je Choi, MPH (University of Washington) reported on the CROCODILE study, which examined metabolic alterations in kidneys of people with T1D. She observed early structural and metabolic changes in kidneys that occurred before the onset of clinical kidney disease and associated structural damage. Her work could contribute to the development of biomarkers that can help predict the onset of kidney disease in people with T1D before it occurs.
Jeremy Pettus, M.D. (University of California at San Diego) conducted a phase 2 clinical trial to address insulin resistance in people with T1D. External insulin therapy can increase levels of insulin in the blood relative to glucose, which reduces sensitivity and may contribute to cardiovascular disease (CVD). Treatment with the glucagon receptor antagonist volagidemab, which prevents the liver from releasing glucose into the blood, reduces insulin requirements by 15%, resulting in improved glycemic control and insulin sensitivity without changes in bodyweight.
Schafer Boeder, M.D. (University of California at San Diego) worked with Dr. Pettus on a phase 1/2 clinical trial that tested whether the addition of SGLTi to the glucagon receptor antagonist volagidemab can further improve glycemic control in people with T1D. The combination of therapies increased TIR up to 86% from 70% and reduced daily insulin use by 27%. Further research is needed to better understand the safety profile of this regimen.
Justin Gregory, M.D. (Vanderbilt University) worked with Dr. Pettus and Dr. Boeder on the above study. He also presented on the use of GLP-1RAs and dual GLP-1/GIP receptor agonists for reducing complications in T1D.
Key takeaways
Clinical trials with GLP-1RAs and SGLTi are providing encouraging evidence about these therapies’ potential to improve long-term health in people with T1D. Breakthrough T1D is working toward a future where these drugs are an option for people with T1D to better manage their blood sugar and reduce complications.
Devices
Real-world insights from Automated Insulin Delivery (AID) systems
- Presenter: David Maahs, M.D., Ph.D.; Stanford University
- Based on published real-world evidence for AID systems in people with T1D, TIR is increased by an average of eight to 15% from baseline in a range of studies across various systems.
- Youth with T1D have better glycemic control and reduced rates of DKA with AID systems. Those with lower TIR at the start of AID system use see the greatest improvements.
Real-world evidence: iLet Bionic Pancreas AID system
- Presenter: Steven Russell, M.D., Ph.D.; Beta Bionics
- The iLet Bionic Pancreas contains a continuously adapting algorithm that automatically determines insulin doses. No carbohydrate counting is required, and meals are only announced as breakfast, lunch, and dinner.
- Data was collected from 16,000 users over two years.
- Users achieved an average HbA1c level of 7.3%, down from 8.9%. This is accompanied by low rates of hypoglycemia and significantly improved self-reported quality of life.
Continuous ketone monitoring: Innovations and clinical applications
- Presenters: Ketan Dhatariya, MBBS, M.D., Ph.D. (Norfolk and Norwich University Hospitals), Lori Laffel, M.D., MPH (Harvard University), Jennifer Sherr, M.D., Ph.D. (Yale University), and Richard Bergenstal, M.D. (HealthPartners Institute).
- DKA rates are increasing in the U.S., but mortality rates from DKA are decreasing.
- The history of continuous glucose monitoring (CGM) offers a roadmap for continuous ketone monitoring (CKM) development, showing how early skepticism gave way to broad clinical impact.
- CKM could allow for earlier detection of rising ketones to prevent DKA. CKM also has the potential to identity infusion set failures, be a valuable addition to AID systems, help monitor early-stage T1D, and more.
- Five new studies funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) will develop CKM for safe and effective use of SGLTi in T1D.
- Tandem, Beta Bionics, Sequel MedTech, and Ypsomed announced plans to integrate Abbott’s dual glucose ketone sensor into their AID systems.
Making the case for time in tight range
- Presenter: Gregory Forlenza, M.D.; University of Colorado
- Dr. Forlenza presented on the benefits and challenges of time in tight range (TITR), also known as time in normal glycemia (TING), defined as blood glucose levels between 70-140 mg/dL.
- TITR will likely be more clinically beneficial than TIR as fluctuations outside of TITR may be better predictors of complications and offer a better therapeutic window for intervention.
- More research is needed to advance therapeutics that will allow people with T1D to achieve TITR before it can be integrated into clinical decisions.
Devices highlight: Breakthrough T1D-funded research
Erin Cobry, M.D. (University of Colorado) presented the results of a Breakthrough T1D-funded clinical trial evaluating an artificial intelligence-powered AID algorithm designed to not require meal announcements. She showed that this algorithm (used without meal announcements) improved overnight TIR, and provided equivalent daytime TIR, compared to participants’ usual care. A major goal for Breakthrough T1D is to advance AID systems that do not require meal announcements to improve both glucose outcomes and quality of life for people with T1D.
Key takeaways
Devices have transformed how this disease is managed. Systems are becoming easier to use with less user input—and, critically, people with T1D are doing better. This is the dream Breakthrough T1D had when we launched the Artificial Pancreas project 20 years ago. We will continue to drive toward our goal of developing systems that provide superior health outcomes with minimal user burden.
Insulins
Inhaled insulin treatment for youth with T1D
- Presenter: Michael Haller, M.D.; University of Florida
- Afrezza® is an inhaled, fast-acting insulin that has proven to be effective in adults.
- The phase 3 INHALE-1 study examined Afrezza® in youth with T1D. Users report greater treatment satisfaction and no increase in weight compared to injected rapid-acting insulin analogs.
- Afrezza® is safe for youth with T1D. The most common adverse events were pulmonary-related, such as coughing.

Breakthrough T1D’s Improving Lives team making an impact
Courtney Ackeifi, Ph.D., Senior Scientist, hosted an Improving Lives Happy Hour with Breakthrough T1D-funded researcher Jeremy Pettus, M.D. The discussion included research priorities for adjunctive (non-insulin) therapies for people with T1D and their healthcare providers. They also discussed the importance of industry partnerships and the role of Breakthrough T1D in driving these relationships, which can accelerate new T1D therapies toward the clinic.
Dr. Ackeifi also spoke at the ADJUST-T1D trial update, contextualizing the use of adjunctive therapies like GLP-1RAs for superior glucometabolic control in people with T1D.
Look out for tomorrow’s article for an update on Cures research presented at ADA 2025!
It’s that time of year again: the American Diabetes Association’s (ADA) 85th Scientific Sessions is taking place from June 20-23, 2025. Scientists, healthcare professionals, and industry leaders will travel to Chicago, IL for the biggest annual diabetes conference in the world. Breakthrough T1D will be there to join the discussion about the latest-and-greatest advancements in type 1 diabetes (T1D) research, prevention, and care from the best and brightest in the field—including many researchers who have received funding from Breakthrough T1D.
Read on to learn more about what we’re looking forward to.
ADA’s 85th Scientific Sessions
This event is one of the largest diabetes conferences in the world, bringing together over 11,000 attendees to share and learn about cutting-edge research and advancements toward cures. Each year, Breakthrough T1D-funded researchers highlight their work, demonstrating the progress we’re making toward achieving our mission. Leading experts will present over 100 studies spanning cell therapies, disease-modifying therapies, diabetes technology, treatments, and more!
What we’re looking forward to
Cures
- Vertex Pharmaceutical’s phase 1/2/3 trial for zimislecel, a manufactured islet therapy that requires immunosuppression
- Sana Biotechnology’s phase 1 trial for UP421, a donor-derived islet therapy engineered to evade the immune system without immunosuppression
- Strategies for improving islet cell manufacturing, transplantation, survival, and immune protection
- Insights into immune mechanisms in T1D
Cures highlight: Breakthrough T1D-funded research
Several investigators from the Beta Cell Replacement Consortium will present updates on cell therapies research, and we’ll hear from Breakthrough T1D-funded researchers like Jeffrey Millman, Ph.D., on scaling up and optimizing manufactured islets.
Breakthrough T1D funded the phase 2 BANDIT study, which found that baricitinib, a JAK inhibitor, can preserve insulin production as measured by C-peptide. The team of Professor Thomas Kay, MBBS, Ph.D., will provide an update on what we learned from this trial.
Improving Lives
- Adjunctive therapies, like GLP-1 receptor agonists, to improve blood sugar and reduce cardiovascular and kidney complications
- Understanding risk factors, biomarkers, and underlying mechanisms of cardiovascular and kidney disease in T1D
- Innovations in continuous ketone monitoring
Improving Lives highlight: Breakthrough T1D-funded research
Excitingly, we’ll hear the outcomes of the Breakthrough T1D-funded phase 2 ADJUST-T1D study, which tested whether the GLP-1 receptor agonist semaglutide (Ozempic®) can improve blood glucose control in people with T1D and obesity.
Breakthrough T1D-funded researchers will have a lot more to say about adjunctive therapies for T1D—including GLP-1 receptor agonists, SGLT inhibitors, and others—and how they can improve glucometabolic control and reduce complications. We’ll also hear updates from Erin Cobry, M.D., about an artificial intelligence-based fully closed-loop (no meal announcements) automated insulin delivery (AID) system in adults and youths with T1D.
Did you know that Breakthrough T1D had a hand in AID system development from the very beginning?
Check out this story from Breakthrough T1D volunteer Doug Lowenstein, who recounts the historical journey of AID systems becoming a reality for people with T1D.
Breakthrough T1D is a leader in type 1 diabetes research
Each year, Breakthrough T1D has an increasingly important presence at ADA. Our leadership and staff organize panel discussions, chair symposia, present research, meet with industry leaders, and host gatherings to promote collaboration. Breakthrough T1D staff from each of our priority areas—Research, Advocacy, and Medical Affairs—will be in attendance.
As leaders in T1D research, we broaden our impact at ADA by shining the spotlight on Breakthrough T1D-funded scientists and clinicians. We are incredibly excited to see the advancements we are making toward cures and improving the quality of life of people with T1D—through our funded research and beyond.
Breakthrough T1D leadership and staff paving the way
- Sanjoy Dutta, Ph.D., Chief Scientific Officer, will co-chair a symposium with Mark Atkinson, Ph.D., titled “Cardiovascular Complications of Long-Term Diabetes: Type 1 vs. Type 2.”
- Dr. Dutta will also participate in a panel discussion titled “Encapsulation vs. Naked Cell Therapy—Immune Challenges and Beta-Cell Perspectives in Diabetes Treatment.”
- Esther Latres, Ph.D., Vice President of Research, will host a panel discussion with industry partners titled “Headway and Hurdles: How can Diabetes Cell and Gene Therapy Succeed?”
- Dr. Latres will also host a workshop titled “Islet Cells in T1D” after ADA, convening experts from around the world to dive deeper into cures research updates.
- Joshua Vieth, Ph.D., Senior Director of Research, will co-moderate a panel discussion with Diamyd Medical titled “Precision in Diagnosis, Power in Treatment: The Future of Type 1 Diabetes.” Stephen Karpen, Pharm.D., Senior Director of Regulatory Affairs, will be a panelist.
- Courtney Ackeifi, Ph.D., Senior Scientist, will speak at the ADJUST-T1D trial update.
- Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, Vice President of Medical Affairs, will speak on a panel about women in T1D.
Updates coming your way
Be on the lookout for important updates post-ADA in the News and Updates section of our website, including news stories dedicated to Cures, Improving Lives, and Medical Affairs.
Check out on-site coverage from ADA on our social channels featuring Breakthrough T1D leadership. Also, Breakthrough T1D CEO Aaron Kowalski, Ph.D., will host a live Facebook Happy Hour on Monday, June 30 at 6 PM Eastern Time. Details to come.
We can’t wait to share the exciting research updates we’ll hear at ADA with our T1D community. This is all made possible through your continued support—thank you!