
What is the pipeline?

Every new medical device, therapy, treatment, and drug—including those for type 1 diabetes (T1D)—goes through the drug development pipeline. Getting a new therapy or device from the earliest stages of research eventually into the hands of people with T1D is a complicated process. Science takes time (from years to decades!), money (from hundreds of millions to billions!), and brainpower (lots and lots of brilliant scientists, doctors, researchers, and more)—and lots of it.

That’s what drives us and our work in the pipeline: people with T1D doing better.
This process is complex, to say the least. There are safety checks at every step of the way. Data are scrutinized, and preclinical and clinical testing must meet ethical standards. In fact, many new drugs and devices don’t make it very far in the pipeline—and those that do take a long time to get there.
Breakthrough T1D is unique in that we work across the entire pipeline—from start to finish—for every promising therapy or device that we invest in. Between Research, Advocacy, and Medical Affairs, we work at every single step to accelerate progress and get new treatments to people with T1D faster than ever.
That’s our value proposition. That’s what makes supporting us different than supporting an individual researcher, or a university, or a company.
Let’s dive a little deeper into the pipeline—and how we’re turbocharging it.
Time and money
Total money spent in the diabetes space in FY24:
$146 million
By Breakthrough T1D
$521 million
Total T1D research support, including by Breakthrough T1D
$160 million
By the Special Diabetes Program
$412.9 billion
In healthcare
Total investments in new drugs and devices:
12 to 15 years
Estimated time it takes a new drug to get to the clinic
$1 billion
Estimated total cost to bring a new drug from discovery research to the clinic
3 to 7 years
Estimated time it takes a new medical device to get to the clinic
$522 million
Estimated total cost to bring a new medical device from discovery research to the clinic
The pipeline in action: Automated insulin delivery systems

Automated insulin delivery systems vs. artificial pancreas systems
At the very beginning, these devices were called artificial pancreas (AP) systems. Today, they are called automated insulin delivery (AID) systems. We’ll be referring to them as AID systems going forward.
The process at a glance:
$171 million
The amount of money Breakthrough T1D spent on AID system-related research from 2005 to 2024
12 years
The amount of time it took for next-generation AID systems to go from discovery research to clinical adoption after Breakthrough T1D became involved
Hundreds of millions of dollars
Total investment in AID systems from all stakeholders
60+ years
The amount of time it took for AID systems to become a reality, starting with the first experimental AP system in 1964
A deeper dive into the process:

Breakthrough T1D x AID systems
Breakthrough T1D has played a significant role in the evolution of AID systems. Doug Lowenstein, a long-time Breakthrough T1D volunteer and supporter, detailed the history of AID systems from the very beginning, and how we accelerated progress at every step of the way.
Discovery research
It’s the early 2000s. I’m a new AID system, but I don’t exist yet—I’m just an idea. I live in the minds of some scientists and researchers who think that I have the potential to one day become a reality. But, turning an idea into reality can be hard. It’ll take a lot of people, time, and effort for me to exist—but it’s not impossible. The question is: how? Answer: it starts with funding.
2005: Breakthrough T1D launches the Artificial Pancreas Project
Breakthrough T1D dedicates funds to scientists and researchers who have compelling ideas to turn AID systems into a reality, marking the start of the decades-long Artificial Pancreas Project (APP). These investments were key in jump-starting research into all the components needed to make an AID system work.
“[The goal was] to keep people alive and healthy until we find a cure. We were losing people…overnight of low blood sugars. If we could automatically dose insulin and have everybody go to sleep and all wake up, that was an incredible victory.” -John Brady, member of Breakthrough T1D Executive Committee in the early 2000s, former Chair of Breakthrough T1D’s International Board of Directors, and father of a son with T1D
Translational research
It’s 2008. Scientists are making progress on the three key parts that need to work in harmony for me to become a reality: a continuous glucose monitor (CGM), an insulin pump, and an algorithm that lets them talk to each other. But, like most new ideas, before I’m allowed to be tested on humans, I need to be tested in animal models first. Lucky for me, I skipped this step because someone made a tool that could simulate how I would act if I was attached to a human being.
2008: Breakthrough T1D-funded scientists create simulator tool to bypass animal studies
The simulator tool, developed from the initial Breakthrough T1D grants, allowed scientists to model how an AID system would respond to real-life scenarios, like eating a certain amount of carbs or exercising, and how this would translate to blood sugar outcomes like time in range or hypoglycemic events. The FDA accepting the use of this tool was a major win—without animal studies, AID system development was accelerated by years.
“The simulator saved at least five years of animal studies because we didn’t require an algorithm to be tested in an animal model to be deemed safe and effective before going into human studies. That entire chunk was eliminated.” -Sanjoy Dutta, Ph.D., Breakthrough T1D Chief Scientific Officer
Clinical trials
It’s 2012. Scientists have come up with different versions of my components that are ready to be tested in humans. For the next four years, I’ll be attached to people with T1D around the world who courageously volunteered themselves to test if I actually work. Turns out, I do a pretty good job at managing blood sugar—and I’m safe!
2012 to 2016: AID systems make headway in clinical trials
While clinical trials for AID systems started in earnest in 2008, they kicked into high gear in 2012, when investigators started conducting trials using different AID components and algorithms in real-life settings. Progress moved quickly, and results from numerous studies supported the idea that AID systems are both safe and effective. Breakthrough T1D funding—along with the Special Diabetes Program—helped move these trials forward.
“Getting involved in AID system trials to me was my chance to pay it forward for somebody else. I have lived 45 years with T1D…I think there’s something comforting in knowing that my body was used for something that not only had the potential to make me healthier, but really was for other people. I do think there’s something to be said for doing good, feeling good, and this is what it felt like being part of trials.” – Alecia Wesner
Regulatory approval
Flashback to 2009. While scientists are busy figuring me out, people at Breakthrough T1D are already thinking about and planning for my future—and how to work with the decision-makers at the FDA who will ultimately decide my fate. The FDA has also given their two cents about the best ways researchers can test me in clinical trials to get the data needed for me to get approved. Fast forward to 2016, it finally happens: I get FDA approval!
2009 to 2016: From APP roadmap to FDA approval
In 2009, Breakthrough T1D published the AP Roadmap, detailing what the future of AID systems will look like—and how we plan to get there. Two years later, we worked with the FDA on clinical trial design for AID systems so that there was a clear path to approval. After human clinical trials, the FDA approved Medtronic’s hybrid closed-loop MiniMed 670G—officially marking the first AID system to be available in the U.S.
“I do give [Breakthrough T1D] credit for pushing, for saying there’s a real need for this.” -Jeff Shuren, M.D., Head of the Center for Devices and Radiological Health at the FDA at the time
Access
Flashback to 2008. At this time, people were still unsure if one of my main components—a CGM—was a reliable way to measure blood sugar. This all changed when a first-of-its-kind clinical trial showed that CGMs are better than finger pricks and glucose meters, and they were covered by insurance shortly after. This decision paved the way for my future: after my approval in 2016, insurers began offering to cover me.
2008 to 2017: Insurance coverage evolves
The landmark Breakthrough T1D-funded clinical trial in 2008 provided the data to convince private insurers to cover CGMs. Nearly a decade later, Breakthrough T1D launched its “Coverage2Control” campaign to advocate for insurance coverage of T1D treatments, therapies, and devices—including AID systems—ultimately resulting in all major private insurers offering coverage of AID systems by the end of 2017, followed by Medicare shortly after.
“Seeing the artificial pancreas go from concept to reality…is what makes Breakthrough T1D and all of the advocacy volunteers—who sent an email, made a call, signed an action alert, or met with their Member of Congress—very proud of this historic achievement and the impact that these will have on the individual lives of those with type 1 diabetes.” -Cynthia Rice, former Chief Mission Strategy Officer at Breakthrough T1D
Adoption
It’s present day. There are tons of iterations of me, and even more coming. People get to choose which version of me they like best. I’m covered by most health insurance. I’ve come a long way since I was just another thought in the minds of a few scientists who had a vision… and now I’ve come to life! Even so, not everyone has chosen to use me yet—and I will continue to evolve and grow until I can make the lives easier of as many people with T1D as possible.
2017 and on: More and more AID systems come to life
After the first hybrid closed-loop AID system was approved, the flood gates were opened. More and more systems are coming to market each year, and they keep getting better. They’re smaller, easier to use, and better at managing blood sugar. They’re covered by both government and private insurance plans. They’re an integral part of routine discussions between people with T1D and their healthcare providers, and people have options to choose which system is best for them. This is a future that was difficult to imagine two decades ago—and now it’s a reality. Even so, the work continues until AID systems are a reality for more and more people with T1D.
“What we brought to bear is resulting in a safer and easier life for hundreds of thousands, and soon millions, of people with T1D, including my son, that is going to keep them safe until something like a cure comes along,” -Jeffrey Brewer, one of the APP founders
The final stage: Cures and improved lives
It took tons of time, money, people, and effort to get us where we are today, but we’re not at the finish line yet. “The end game for AID systems,” says Breakthrough T1D CEO Aaron Kowalski, Ph.D., “is to have multiple compatible pumps, glucose sensors, and algorithms, so that patients can mix and match what they prefer.”
The end game for T1D as a whole, however, is cures. AID systems have greatly improved the lives of those with T1D—and will continue to do so now and in the future—while we continuously work toward cures that are one day available to everyone with the condition.
The path followed for AID systems is a roadmap for other therapies coming down the pipeline. Breakthrough T1D’s Project ACT is taking a page from the AP roadmap and applying it to cell therapies, so that functional cures can get to people with T1D who want them as quickly and safely as possible.

While I’m proud of my work as a scientist at Breakthrough T1D on AID systems (and my brother and I currently wear AID systems that are derived from Breakthrough T1D-supported work), more than anything else I want to take off my diabetes devices and achieve what our founders set out to do—find cures for T1D.”
Breakthrough T1D successfully took an idea and turned it into a reality—and we’ll do it again and again until T1D is a thing of the past.
Imagine a world where everyone understood what it means to live with type 1 diabetes (T1D). That’s our goal every November for Diabetes Awareness Month: to create a global movement that shines a light on T1D and the resilient people who face it.
Building awareness and understanding about T1D is more important than ever. When we see T1D, we move closer to life-changing breakthroughs in research, advocacy, and support—helping people with T1D live fuller, healthier lives as we drive toward cures.
Together, our stories change how people see T1D.












Share your story with us!
If you’re inspired by the stories above, share your story too. What do you wish people understood about life with T1D? How have T1D breakthroughs changed your life? Post on social media, tag @BreakthroughT1DHQ, and use the hashtag #SeeT1D.
There are a lot of commonly believed myths about type 1 diabetes (T1D). The misinformation and confusion it causes can be harmful to our T1D community—and people who may be at risk of developing this condition.
In addition to our pursuit of cures and breakthroughs, we work to spread awareness about T1D to dispel myths and keep people informed—whether they live with T1D or not.
So let’s play Two Truths and a Lie, a T1D edition. In each of the questions below, you see three statements about life with T1D. Only two are true. Your challenge is to separate fact from fiction and identify the lie. Are you ready?
Take the quiz
- T1D can develop suddenly in both children and adults at any age
- T1D can be caused by diets that are too rich or too high in sugar
- Around 85% of T1D diagnoses are in people without a known family history of the condition
- Rapid and unexplained weight gain
- Slow-healing cuts
- Experiencing extreme thirst or frequent urination
- Several collegiate and professional athletes currently live with T1D
- Depending on the person and the type of activity, exercise may cause a person to experience a drop OR a rise in blood-glucose levels
- It is not recommended that people living with T1D engage in physical activity without the direct supervision of a medical professional
Check your answers
Now it’s time to see how you did by checking your answers:
B. Diet does not cause T1D—and there are no dietary changes one can make to prevent its onset.
A. Unexplained weight loss—not weight gain—is a common T1D symptom. This can indicate that a person’s body isn’t able to get enough energy from the food they eat.
C. Exercise isn’t just recommended—it’s especially important for people living with T1D. Staying active is great for the body and the mind, but, just like most other parts of life with T1D, it requires some extra preparation before diving in.
We’re committed to ensuring that people affected by this disease always have the information and support they need to live healthy and safely—while we pursue life-changing cures and breakthroughs every day.
You can help!
With a donation, your support will accelerate our path toward a world without T1D.
Halloween is just around the corner, and kids are getting their costumes ready!
For families impacted by type 1 diabetes (T1D), a holiday centered around glucose-spiking treats can be a little scary. Breakthrough T1D is here to give you some tips to ensure your Halloween is fun, not frightful!
Focus on more than candy
Halloween is about dressing up and having fun! Participating is important—it’s a chance for kids to get imaginative and express their creativity with their friends.
Make it a learning experience
Use Halloween to teach your child how to enjoy treats responsibly. This can help them learn to estimate carbohydrates and calculate insulin doses. Record insulin doses and blood sugar levels after you child eats candy to learn what works best.
Our Halloween Guide has more helpful tips to make your holiday scare-free, including carb counts for candy!
Find a balance
Restricting sweets can lead to guilt and an unhealthy relationship with food. Encourage balancing healthy meals and exercise alongside occasional treats.
Let go of perfection
Look beyond the day’s blood sugar levels. Reinforce positive self-talk when things don’t go perfectly. If their blood sugar is high, help them understand how to handle that number without self-blame. Take a correction dose and move on.
And don’t forget: Halloween candy comes in handy for treating low blood sugar!
Happy Halloween!
Cold and flu season is right around the corner. Being sick is never fun, but for people living with type 1 diabetes (T1D), it poses extra concerns. When a person gets sick, their body is under stress. To combat that stress, the body naturally releases hormones that help fight disease. Those hormones, however, can cause blood-glucose levels in the body to rise and disrupt the way insulin works in the body.
When living with T1D, having a sick day plan of action is crucial.
Sick day checklist at a glance
- Check your blood sugar regularly
- Stay hydrated
- Don’t stop eating
- Check for ketones
- Have glucagon on hand for emergencies
- Have anti-nausea medication on hand
- Develop a sick day plan with your healthcare provider
Closely monitor blood-glucose levels and ketones
When you’re under the weather, you need to monitor your blood glucose and ketones more often than usual. Check your continuous glucose monitor (CGM) if you have one, or frequently use finger sticks. It may be necessary to administer extra insulin to bring down higher blood glucose levels.
Elevated ketone levels often occur when blood sugar readings are high. This can signify that the body is using fat and muscle for energy, instead of sugar. Be on the lookout for ketones, as very high levels could lead to diabetic ketoacidosis (DKA), a dangerous condition that demands immediate medical attention and can be fatal if left untreated.
Be aware of how medications affect blood sugar
Many over the counter (OTC) medications can affect blood glucose levels. Sugar-free cough syrups or cough suppressant pills taken orally are good choices for people with T1D. Other drugs, like decongestants, also raise blood glucose. On the other hand, aspirin in large doses can lower blood glucose levels.
People with T1D must also be aware of the effects of pain and fever reducers. Acetaminophen, which helps counteract minor aches and fevers associated with the flu or a cold, can cause false or no readings in CGMs and can be toxic to the liver and kidneys. Anyone with kidney complications should speak to their healthcare provider before using these drugs. Ibuprofen should also be administered with care, as it can enhance insulin release and increase the risk of hypoglycemia, or low blood sugar.
Tips for eating and drinking while sick
Because it can be difficult to stick to normal eating habits when feeling unwell, it’s good to always have products on hand that will help you maintain a healthy carbohydrate intake. Foods like crackers, unsweetened applesauce, or yogurt are relatively easy on the stomach. Have anti-nausea/antiemetic medication like ondansetron (Zofran) available for sick days so that you can continue to eat and drink, even in small amounts. If you have difficulty swallowing because of a sore throat, liquids like smoothies or soup can be a soothing, short-term solution.
It’s important to stay well hydrated, especially when ketones are present, to flush them out. Water, broth, and sugar-free gelatin, popsicles, or soft drinks are great ways to stay hydrated when experiencing a cold or the flu with type 1 diabetes.
Prepare for emergencies
When living with type 1 diabetes, you must be ready for the worst, even if you never experience it. Work with your diabetes care team to develop a plan for managing your T1D while sick. Always have emergency glucagon on hand to treat severe lows and make sure you know how to use it.
Have your doctor’s phone numbers (including how to reach them at night and on weekends or holidays) ready in case you encounter these symptoms:
- Vomiting or diarrhea for more than six hours
- Having trouble breathing
- A fever that won’t go away after a couple of days
- Large levels of ketones
- Unable to keep food or liquid down and are experiencing hypoglycemia
If you do need to make an emergency room visit, it is very important that you mention that you have T1D.
Prevention is key
Preventing illness is always the best outcome. Getting a flu shot every year is strongly advised. Make sleep and hydration a priority, and wash your hands often during cold and flu season.
Check out our Sick Day Guide for more information on managing cold and flu with type 1 diabetes.
Analysis compiled by Avalere Health and supported by Breakthrough T1D (formerly JDRF) finds that research funded by the Special Diabetes Program (SDP) has yielded more than $50 billion in federal healthcare savings.
Created by Congress in 1997 and administered by the National Institutes of Health (NIH), the SDP provides federal funding to support research to prevent, treat, and ultimately cure type 1 diabetes (T1D).

The Special Diabetes Program (SDP) is set to expire soon! Ask your Members of Congress to renew the SDP!
Working closely with the SDP’s champions in Congress—Senators Susan Collins (R-ME) and Jeanne Shaheen (D-NH) and Diana DeGette (D-CO) and Gus Bilirakis (R-FL)—Breakthrough T1D leads advocacy efforts supporting continued funding for the SDP.
The SDP has directly supported many of the most important innovations that have changed how T1D is managed—including continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems.
Today, these technologies are the standard of care and are used more broadly, including by people with type 2 diabetes (T2D) contributing to improved health outcomes for people with T1D and T2D.
Understanding the SDP’s Economic Impact
To better understand the program’s economic impact, Avalere Health conducted an analysis of estimated federal cost savings associated with SDP-supported technologies.
The analysis focused on direct medical expenditures and modeled the savings resulting from the use of CGM and AID systems including savings to the tax-payer supported public insurance programs like including Medicare, Medicaid, and the Department of Veteran’s Affairs.
Avalere Health’s findings indicate that CGMs and AID systems alone have generated at least $50 billion in federal healthcare savings through improved glucose management and reduced diabetes-related complications.
The actual federal savings and total economic impact of these technologies is likely higher as the analysis does not account for indirect cost savings—such as improved productivity (such as, not having to take time off work to treat a low, etc.), reduced disability, or long-term prevention of complications.
CGM and AID 2024 utilization and savings, total federal savings from start of SDP
| Technology | Population Using Technology (2024) | Annual Savings Per Patient (2024) | Federal Savings By Diabetes Type (1998-2024) | Total Federal Savings (1998-2024) |
|---|---|---|---|---|
| Continuous Glucose Monitors (CGMs) Without AID | T1D: 749,700 T2D: 1,738,600 | $5,502 | T1D: $10.9 Billion T2D: $21.2 Billion | $32.1 Billion |
| Automated Insulin Delivery System (AID) | T1D: 780,300 T2D: 1,856,400 | $5,306 | T1D: $5.4 Billion T2D: $12.9 Billion | $18.3 Billion |
T1D therapies on that are on the market or currently in development have benefited from SDP-supported research in numerous ways, including:
- Clinical trials and technology validation for CGMs and AID systems
- Development of disease-modifying therapies, including Tzield, the first therapy approved by the FDA to delay the onset of insulin therapy dependence in T1D
- Development of advanced therapies for diabetic eye disease
- Early-stage beta-cell replacement research, including Lantidra, the first therapy approved by the FDA to place insulin-producing cells back in the body and ultimately reduce or eliminate the need for insulin therapy
The SDP’s Future
This study and existing literature confirm that the SDP has demonstrated a strong return on investment—both clinically and economically. The SDP has also made life with T1D more manageable. AID systems are now standard of care and they are leading to better outcomes with less disease burden.
Ongoing support for diabetes research and treatments will be essential to realizing the full potential of emerging innovations for individuals with diabetes
Disruptions in SDP funding may hinder clinical research infrastructure, delay scientific advancement, and slow patient access to therapies that reduce complications and lower overall costs.
As policymakers evaluate the future of the SDP in the debate over government funding stakeholders should consider how funding decisions will maintain or interrupt momentum in diabetes research, bringing advanced therapies to market, and how that will impact continued cost savings from improved health outcomes for people with diabetes.
Read the Insights & Analysis report and the full white paper on the Avalere Health website.
Editor’s note: Content for this story drawn from Avalere Health’s Insights & Analysis report.
In December 2023, Mattel approached Breakthrough T1D with a partnership opportunity for the first Barbie with type 1 diabetes (T1D).
Less than two years later, in July 2025, Barbie with T1D was launched.
The collaboration reflects our commitment to ensuring that the millions of people living with T1D are seen, heard, and empowered.
Barbie with T1D was thoughtfully created with input from Breakthrough T1D employees who live with T1D, including Emily Mazreku, Director of Marketing Strategy, and members of our T1D community, including volunteers Natalie and Nadia Stanback. Here, we talk to them about their experience.
Q: What was it like being part of the creation of Barbie with T1D?
Natalie: “As a mom, it was a full-circle moment. I’m raising a daughter who is changing the narrative on what a T1D diagnosis means, not just for our family, but for the entire T1D community. And, as a sister to a brother who lost his battle with T1D, it’s a bittersweet moment. I wish, with all my heart, that he could have had examples of what it looks like to thrive with T1D instead of feeling the need to hide, and sometimes ignore, it.”
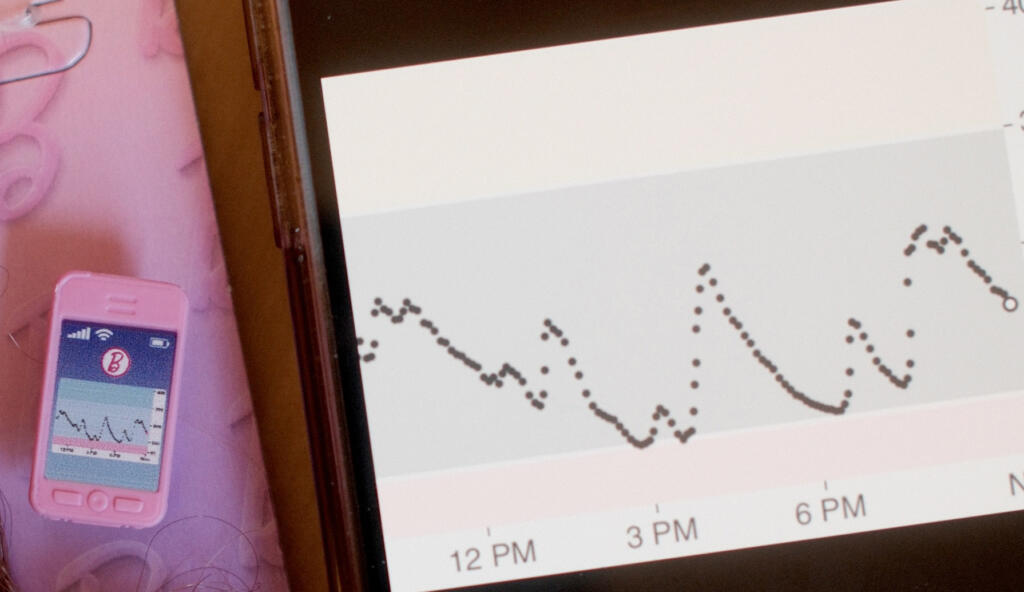
Barbie’s CGM reading, inspired by Emily Mazreku’s actual CGM reading
Nadia: “When I worked with Mattel, I told them that there should be a CGM trend line that was up and down because that happens a lot to people with T1D. I also thought she should have a fun overpatch for her CGM. I thought it was really cool that they listened to me!”
Emily: “As an adult living with T1D, being part of creating Barbie with T1D with Mattel, our team at Breakthrough T1D, and the T1D community was a dream come true. When Mattel asked what a day with T1D could look like, I shared a photo of my CGM graph, and now that same photo is on Barbie’s phone. Seeing my own (very real!) CGM reading on Barbie’s phone is something that will always feel surreal.”
“Barbie with T1D show[s] that all types of people can be cool and powerful.”
Nadia Stanback
Q: What does a Barbie with T1D mean to you?
Emily: “Barbie with T1D means seeing T1D—the highs, the lows, and everything in between—and most of all, celebrating the resilient people who face it. It means showing my 3-year-old daughter, Ellie, that Barbie confidently wears her diabetes devices for everyone to see, and as she says, ‘just like mommy.'”
Natalie: “When Nadia was diagnosed with type 1 diabetes, I was focused on making sure she wouldn’t be defined by it. But she has shown me that she can—and will—do great things with T1D, and Barbie is a beautiful reflection of that; she is bold and powerful AND has T1D.”
Nadia: “I think that the different types of Barbies, including Barbie with T1D, show that all types of people can be cool and powerful.”

“Barbie with T1D is showing everyone with T1D that their experiences matter.”
Emily Mazreku
Director, Marketing Strategy, Breakthrough T1D
Q: Why does representation matter for conditions like T1D?
Emily: “T1D is a 24/7 condition that feels invisible and is often misunderstood, so when people with T1D see themselves, it’s powerful. Visibility not only helps people with T1D feel seen, it also builds confidence, starts a conversation, and raises awareness of what it really means to live with T1D. That’s what Barbie with T1D is doing—showing everyone with T1D that their experiences matter.”
Natalie: “Giving visibility to an often-invisible condition like T1D has opened dialogue. It’s a conversation that isn’t always comfortable, but when minds are open and voices are heard, there is so much learning to be had.”
“Nadia has shown me that she can—and will—do great things with T1D, and Barbie is a beautiful reflection of that.”
Natalie Stanback

Since 1994, Marshalls has been a committed supporter of Breakthrough T1D.
As Breakthrough T1D’s largest corporate partner, Marshalls raises more than $3 million each year through in-store fundraisers at checkout, donations from the TJX Foundation, and store support of local Breakthrough T1D chapters across the country. These combined efforts have resulted in more than $47 million in donations to-date for Breakthrough T1D, helping to advance our mission of accelerating life-changing breakthroughs to cure, prevent, and treat type 1 diabetes (T1D) and its complications.
Marshalls’ 2025 in-store fundraising campaign for Breakthrough T1D runs from Sunday, August 31, through Saturday, September 27. Find your local Marshalls here.
Creating a better future

Digital creator Kris Leeper’s type 1 diabetes journey began with his diagnosis in 2013. It was, in his own words, “terrifying.”
“My wife and I were bombarded with conflicting information,” Kris said. “We were navigating unchartered territory as a family, just two years into our marriage. I felt lost and uncertain about the future.”
T1D technology changed the game for Kris. “The introduction of a continuous glucose monitor (CGM) and pump revolutionized my outlook on living with T1D. I immediately felt better,” he said. “Access to and understanding of diabetes technology has profoundly improved my daily life.”
Kris appreciates Marshalls for bringing awareness to Breakthrough T1D’s efforts to make life-changing technologies a reality for the more than 1.5 million Americans facing T1D.
“Companies like Marshalls play a pivotal role in bringing Breakthrough T1D’s mission to the mainstream,” he said. “Marshalls’ support for advocacy, research, and fundraising demonstrates the power of a major brand in creating a better future for people living with or caring for those with T1D.”
Driving breakthroughs forward
1 in every 31 families in the U.S. are impacted by type 1 diabetes—it places a tremendous financial, emotional, and mental burden on those who live with it and their loved ones. With support from partners like Marshalls, Breakthrough T1D is making everyday life with T1D safer and easier through technology—like the CGM Kris uses, and automated insulin delivery systems.
Every donation collected through the Marshalls partnership has also helped drive critical research in cures for type 1 diabetes. From the launch of the Special Diabetes Program in 1997, to the first manufactured islets making insulin in 2014, and the FDA approval of Tzield, the first disease-modifying therapy for type 1 diabetes, Breakthrough T1D is driving innovation forward to make the condition a thing of the past.
“The future of type 1 diabetes is bright,” Kris said. “We are on the cusp of living longer, experiencing fewer complications, and enjoying a life without limits.”
Breakthrough T1D thanks Marshalls for its incredible partnership in improving and changing life with type 1 diabetes.
Nearly half of the states in the U.S. currently have a policy in place that restricts or bans student cell phone use in school.
For students with type 1 diabetes (T1D) who use technology to manage the condition, this can present challenges.
How do cell phones help with managing type 1 diabetes?
Technology can make managing T1D easier and safer. Cell phones have increasingly become part of the standard of care for type 1 diabetes.
Cell phones are often used in connection with a continuous glucose monitor (CGM) to track and manage blood sugar levels. CGMs linked to a cell phone also provide a way for parents to see their child’s blood-glucose in real-time so they have the ability to intervene remotely via text to their child or the school nurse.
In addition, some insulin pumps connect to cell phones to allow for mobile insulin dosing, including the Omnipod 5 and the Tandem tSlim: X2.
In the event of a T1D emergency, a cell phone can also provide the student with a way to contact their parents or school nurse if they need help.
“For students with type 1 diabetes, having access to a cell phone allows for maximum independence in their diabetes care, therefore, not missing class time,” says Shannon Dutton, Senior National Community Engagement Manager at Breakthrough T1D.
The importance of a 504 Plan
A 504 Plan is a legal, written document that specifies what reasonable modifications and accommodations the school must provide for a student with a disability, including type 1 diabetes. 504 Plans are a way for parents to hold the school accountable for meeting their child’s needs while in the school’s care.
“It’s important for students with T1D to have a 504 Plan in place to ensure their accommodations are clearly outlined for school staff,” Dutton says. “Having a cell phone listed as a medical device in a student’s 504 Plan allows them to have unlimited access to their phone throughout the day for glucose monitoring or insulin dosing.”
Keeping the lines of communication open
Parents and schools must work together to ensure students with T1D have access to the tools they need to succeed in the classroom.
Breakthrough T1D offers a variety of printable educational materials that parents can share with school staff for a better understanding of T1D management and technology:
- Using Technology to Manage Type 1 Diabetes
A detailed overview of how T1D technology works, including CGMs, automated insulin delivery systems, and smart pens. - What I Want My Teachers to Know About T1D
An explanation of type 1 diabetes and how it’s managed from the student’s perspective to the teacher. - What You Need to Know About Hypoglycemia (Low Blood Sugar)
An educational guide about the signs and causes of low blood sugar and how to treat it. - What You Need to Know About Hyperglycemia (High Blood Sugar)
An educational guide about the signs and causes of high blood sugar and how to treat it
A new study funded by Breakthrough T1D and published in the journal Diabetes Therapy shows that people with type 1 diabetes (T1D) and caregivers are largely willing to try next-generation therapies, drawn by the promise of reduced insulin reliance, greater normalcy, and freedom from constant disease management.
The findings highlight the importance of ensuring patient and caregiver perspectives guide regulatory decisions around the balance of a therapy’s risks and benefits.
The promise of next-gen therapies
Advances in T1D management, such as automated insulin delivery (AID) systems, mark major progress over the past decade. Despite these gains, insulin therapy remains physically, mentally, and emotionally demanding, and substantial unmet needs persist.
Next-generation therapies aim to move beyond insulin replacement through two approaches:
- Disease-modifying therapies (DMTs): Preserve insulin production by slowing or halting autoimmune destruction of beta cells.
- Cell Therapies: Restore insulin production by delivering new functional cells, with the potential for partial or complete insulin independence.
For the study, researchers interviewed people with T1D and caregivers to better understand their perspectives on the risks and benefits of these therapies.
What participants said
Participants expressed willingness to try new therapies, with 92% of adults, 70% of caregivers, and 100% of adolescents saying they would try a DMT at the time of T1D diagnosis. Every participant said they would try cell therapies.
Participants also shared their thoughts on top benefits of both DMTs and cell therapies.
- Top benefits: DMTs: Reduced insulin reliance and an extended period when a
person with T1D produces their own insulin; cell therapies: insulin independence,
more normalcy, and freedom from constant demands on diabetes.
Why this matters
To approve a product, regulators must judge whether the therapy’s benefits outweigh its
risks. This study shows that , people living with T1D and their caregivers have strong view
about which benefits matter and how much they matter. Because DMTs and cell therapies
bring significant benefits compared to insulin therapy, regulators should incorporate these
patient perspectives into regulatory decision-making.
About the study
The Breakthrough T1D Patient Preference Study is being conducted in two phases. In the first phase, researchers interviewed 26 individuals—12 adults, 4 adolescents, and 10 caregivers—about their views on DMTs and cell therapies. These insights are now shaping a large-scale survey that will measure how the T1D community prioritizes different risks and benefits. Results from that survey are expected later this year.