
Type 1 diabetes (T1D) occurs when the body’s immune system attacks the insulin-producing beta cells in the pancreas. But this doesn’t happen overnight—instead, it’s a gradual process of beta cell loss over time.
As the immune system attacks and destroys more beta cells, insulin production decreases, leading to abnormal blood-sugar levels. Eventually, enough beta cells are destroyed, and insulin production drops so low, that symptoms start to appear. Most people are diagnosed when these symptoms occur.
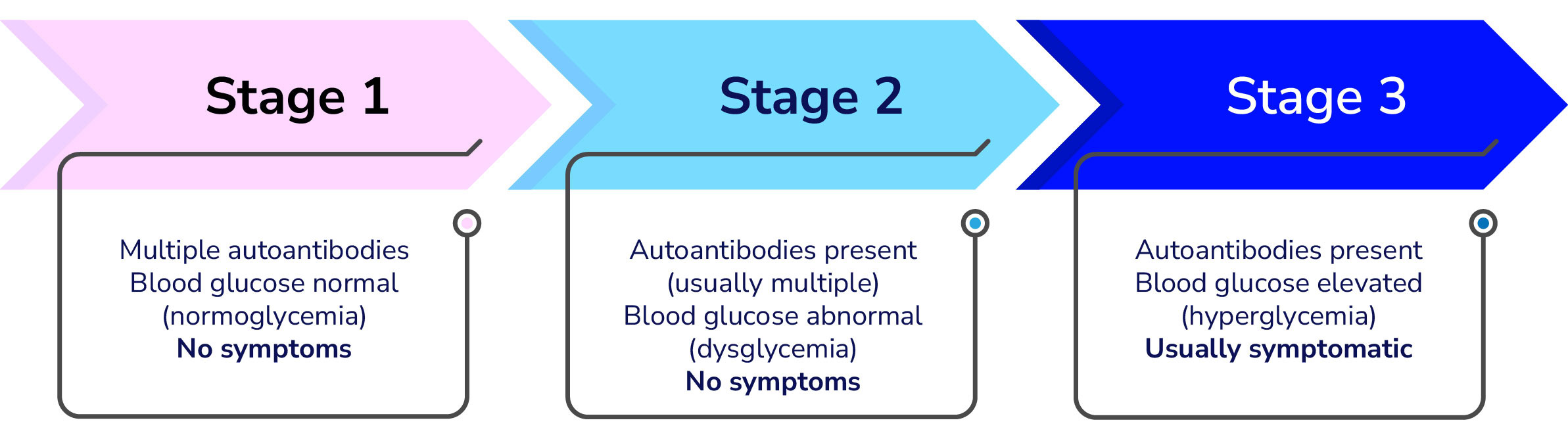
Insulin is an essential hormone that helps the body use the nutrients in our food as energy. Depending on how many beta cells are left in your body when your T1D is diagnosed, you may experience the honeymoon phase—which means you have a small number of beta cells that are still working and producing insulin.
Think of these remaining beta cells as a moderating force, helping you smooth out the peaks and valleys of managing T1D. That means you will likely need less insulin and may have an easier time reaching blood glucose targets.
Are some people more likely to experience the honeymoon phase?
The honeymoon phase is more common in (but not exclusive to) people who do not have diabetic ketoacidosis (DKA) at diagnosis, people with lower HbA1c levels at diagnosis, and children who are at least 5 years old.
How long does the honeymoon phase last?
The honeymoon phase is different for everyone! But it is always temporary. Generally, it lasts several months, but can be up to 1 to 2 years.
Is it possible to extend the honeymoon phase?
Currently, there are no FDA-approved therapies to extend the honeymoon phase. In the past decade, Breakthrough T1D has invested more than $273 million in research on disease-modifying therapies—which have the potential to slow, halt, prevent, or even reverse T1D onset—and several are in human clinical trials.
That includes baricitinib, a JAK inhibitor that interferes with signaling pathways critical to both immune cells and beta cells. In October 2025, Eli Lilly announced it was launching a new phase 3 clinical trial of baricitinib to examine whether the drug can preserve beta cell function in people with newly diagnosed T1D, which could potentially extend the honeymoon phase. Breakthrough T1D funded an earlier clinical trial investigating this therapy that helped make this next step possible.
What happens next?
Unfortunately, the honeymoon phase does not last forever. Even though this is a gradual process, eventually your immune system will destroy most or all of the beta cells in your pancreas. That means your daily insulin needs will gradually increase, and you may have more trouble managing your blood glucose levels.
It’s important to stay in contact with your T1D care team—they can help you make adjustments so you can reach your T1D management goals!
Autoimmune diseases occur when a person’s immune system wrongly attacks the body’s healthy cells, tissues, and organs. This response can result in a range of conditions, depending on which parts of the body are affected.
Autoimmune diseases are often called “invisible” because their symptoms are not always evident to the people around you. As a result, it may be difficult to get an accurate diagnosis, and many people facing autoimmune diseases feel a lack of support.
This Autoimmune Disease Awareness Month, we asked a few members of the T1D community to share their perspectives on living with an autoimmune disease.


Elizabeth
32 | New York
Jason
43 | Ohio
Jonah
21 | California
Yousra
26 | Washington, D.C.Q: What do you wish people knew about everyday life with autoimmune disease?
Arielle:
I wish people knew that T1D relates more to other autoimmune conditions than other types of diabetes. My immune system attacks my skin, pancreas, and thyroid every minute of every day.

Elizabeth:
That it’s not just the needles and insulin, the Dexcom, and Omnipod changes. There are so many mini decision-makings throughout the day that people don’t hear. It’s the first thing I think about when I wake up and the last thing before going to bed. So be patient with us, we have a lot on our minds!
Jason:
With T1D, you don’t get any days off. It’s there when you wake up in the morning, and when you go to bed at night, and all day in between. It’s constantly in the back of your mind. It goes beyond simply watching what you eat. There are so many things that can impact your blood sugar, like stress, medication, illness, exercise, and even lack of sleep!
Jonah:
No two days are the same. It’s possible the first time we meet, I’m having a great day where my blood sugar is in range, but the next time we meet, I’m in a terrible mood because I’ve had a week of out-of-control blood sugar and technology issues. The highs and lows of the disease impact everyday life more than people would expect.
Yousra:
Part of managing diabetes is trying to expect the unexpected; everything impacts T1D, from food to stress to exercise to sleep. Even if I do the exact same things every single day, my blood sugars would still be different, which doesn’t mean I am doing something wrong.
Q: Fill in the blank: “I wish others would _______ when I’m talking about my condition.”
Arielle:
Listen more.
Elizabeth:
Listen with the willingness to learn.
Jason:
Be inspired to help make a difference.
Jonah:
Ask more questions.
Yousra:
Ask questions.
Q: What are your tips for living a healthy and happy life while managing an autoimmune disease?

Arielle:
My main tip is to stay resilient and on top of your health. You will have to take extra steps in the day to equip your body for every activity and moment, but with your strength and preparation, you can do it!
Build a strong proactive support system, too. My husband, my son, my sister, my dad, my aunt, my close friends…they are all the reasons I can remain strong and the reasons I feel my best. They understand the prep work and are right alongside me for the ride. My son, Jace, often hands me gummies when my blood sugar dips low, and my CGM alarm goes off. He says, “One for mommy’s blood sugar and one for Jace’s tummy.”
Elizabeth:
Try not to become hyper-fixated on perfection—good blood sugars 100% of the time, eating healthy 100% of the time, being active 100% of the time…it’s not realistic!! If you want to go to the gym but enjoy a glass of wine and a burger after, that’s okay! If your blood sugars were a little high but you got a good night’s rest, that’s okay! Try shifting your focus to the good.
Jason:
First and foremost, don’t try to handle everything yourself. Make sure you have a great support team behind you. This includes finding the right physician who’s a good fit for you, but also making sure you have a family member, friend, neighbor, or someone close to you whom you can talk to when you’re struggling. And understanding there are days you’re going to struggle—it’s part of the disease. You can count carbs exactly, take the exact dose of insulin you should, and still not get the result you want. It’s part of the journey of managing the disease. Type 1 diabetes has an incredible community, from amazing organizations like Breakthrough T1D with so many support resources to activities like meet-ups, online communities, and so much more.

Jonah:
Everything in moderation. I’ve lived a happy life by doing everything in moderation. I eat pretty healthily, but if I want to eat something that could be seen as unhealthy, I don’t eat a ton of it. I exercise a lot, but I make sure not to overdo it and hurt myself. I try not to beat myself up if I’m having a rough week. After 15 years, I know there will be blips and tough spots, but it won’t impact my overall health in any severe ways.
Yousra:
You are always your own biggest advocate, which is part of managing the nuances of your condition. If you’re concerned about something big or small, take the time to learn more, ask questions, and find options that feel like the right fit for you. Even if treatment plans are similar, there is no “one-size-fits-all” policy for managing your day-to-day.
Q: What do you wish you could tell your younger self?
Arielle:
My first autoimmune condition was T1D, and I was diagnosed at 4 years old. I wish I could tell younger me that there might be more conditions to come in your future, but you have the strength to deal with it. You will become independent faster than most your age, you will have challenges in your adolescence and years to come, but you will be resilient through it all. You will grow up to work at the top organization for type 1 diabetes research. Be proud of who you are!
Elizabeth:
You go, girl!

Jason:
Don’t beat yourself up over your numbers. I used to go to my doctor’s appointments feeling like I was getting the results of an exam in school, and if my numbers weren’t where I wanted them to be, I’d feel like a failure. Some months are going to be harder than others, and that’s okay—that’s life. With advancements, it’s going to become easier to manage the disease, and it’s truly going to take some of the stress away.
Jonah:
There are a lot of fantastic people you will meet with this disease that come from all different places, and you will be able to bond with them on a level most friendships never get to.
Yousra:
You have made it through 100% of your bad days, so don’t be too hard on yourself when it comes to an unexpected test result or a day with unexplainable blood sugars. Give yourself a moment to be proud of progress as you work for more.
Q: What is your hope for the future?
Arielle:
My hope for the future is that the work we are doing at Breakthrough T1D will help other autoimmune conditions as well. They are all linked, and we are getting closer to cures!
Elizabeth:
I, of course, hope one day there is a cure for type 1 diabetes! But until then, I hope people just take the time to educate themselves on things they may not understand and always lead with kindness. You never know what someone else is going through!
Jason:
My hope is in a cure! Thinking about the day I get to take off my insulin pump doesn’t feel like a dream—it feels inevitable. Until there’s a cure, I’m hopeful about the incredible technological advances that have been made. I think back to when I was first diagnosed 25 years ago, and relying on only a few finger pricks throughout the day to see how I was doing. I would go to bed at night fearful that I would have low blood sugar in the middle of the night and not realize it.
Now, I can look at my phone and see my readings every 5 minutes for the entire day, and my insulin pump automatically adjusts insulin throughout the day. I get alerted any time my blood sugar is low, and my wife can look at her phone and see my blood sugar—even if I’m thousands of miles away! It’s truly amazing how far we’ve come. I feel like our progress isn’t linear either; it’s exponential, and advancements are moving so quickly now.
Jonah:
A cure that is accessible to all people living with this disease, regardless of socioeconomic status or citizenship.

Yousra:
I hope that shame and stigma are taken out of the equation when it comes to talking about T1D, especially since it’s not something any of us could have avoided. I would love for people to know what life is like with T1D to get a better sense of all the hard work it takes to try to stay healthy.
Share Your Story
Living with T1D looks different for everyone. We want to hear your story: the highs, the lows, the challenges you’ve overcome, and the people who have supported you on your journey.
March is Autoimmune Disease Awareness Month. Breakthrough T1D is taking this opportunity to provide education and shed light on these often “invisible” conditions.
What is Autoimmune Disease?
Autoimmune diseases occur when a person’s immune system wrongly attacks the body’s healthy cells, tissues, and organs. This response can result in a range of conditions, depending on which parts of the body are affected. In type 1 diabetes (T1D), the immune system targets the beta cells in the pancreas that produce insulin, a hormone that helps the body convert sugar to fuel and regulates blood-sugar levels.
There are over 100 known autoimmune diseases, including:
- Type 1 diabetes
- Celiac disease
- Lupus
- Hashimoto’s thyroiditis
- Rheumatoid arthritis
- Addison’s disease
- Multiple sclerosis (MS)
- Psoriasis
It’s not uncommon for people to have more than one autoimmune disease. This is because many autoimmune diseases share common genetic backgrounds. Also, once the immune system becomes dysregulated because of one autoimmune disease, it can trigger additional immune malfunction and start attacking other parts of the body.
Autoimmune diseases are chronic and can significantly impact daily life. They can affect anyone of any race, age, or gender, though women are considerably more likely to develop them. This increased risk is likely due to hormonal and genetic factors.
What Causes Autoimmune Diseases?
Family history and environmental elements, such as hygiene, viruses, and toxins, may contribute to the development of autoimmune diseases. However, the exact cause is still unclear, and there are currently no cures for any autoimmune diseases. Breakthrough T1D wants to change that.
The Potential for Cures
Our funded research to cure, prevent, and treat T1D and its complications also has the potential to impact other autoimmune diseases. This is because many of the same breakdowns in the regulation of the immune system that lead to the attack on beta cells may also be responsible for the attack on other tissues and cells. Therefore, an immune-based therapy that shows promise in one autoimmune disease has a good chance of working in others.
“The strategies we are testing to rebalance the immune system are applicable to many autoimmune diseases,” said Joshua Vieth, Senior Director of Research and head of the disease-modifying therapies program. “We work closely with other autoimmune organizations such as the Lupus Research Alliance and the National MS Society to coordinate on research strategy, maximize the resources each of us are putting into the field, and accelerate therapeutic advances that can benefit those living with autoimmune diseases such as T1D and beyond.”
Autoimmune Disease by the Numbers
100+
Number of known autoimmune diseases
50 million
Approximate number of Americans living with autoimmune disease
25%
Percentage of Americans who have more than one autoimmune disease
80%
Percentage of Americans with an autoimmune disease who are women
The Invisibility of Autoimmune Diseases
Autoimmune diseases are often called “invisible” because their symptoms are not always evident to the people around you. Bodily responses to autoimmune diseases, such as joint pain, digestive issues, or fatigue, can be severe, yet they are not always outwardly visible. As a result, it may be difficult to get an accurate diagnosis, and many people facing autoimmune diseases feel a lack of support.
When you are living with an autoimmune disease, it’s important to advocate for yourself and educate those around you so that you can get the help you need.
Breakthrough T1D’s commitment to supporting the entire T1D community includes providing you with information and resources to help you navigate life with type 1 diabetes (T1D).
The data is conclusive: Continuous glucose monitors (CGMs) help millions of people better manage their T1D. However, we know that, at times, people in our community experience issues with their CGMs. This can include inaccurate readings, faulty sensors, customer service issues, lost time, and unexpected or high costs.
Breakthrough T1D created this guide to help you with sensor issues, from what to do if your symptoms and CGM reading do not match (check with a glucometer!) to how to get a sensor replacement as quickly as possible.
On This Page:
The most important thing to do if you are having an issue with your CGM, or if your symptoms do not match the reading on your CGM, is to always follow the manufacturer’s guidelines and check your blood sugar with a glucometer.
Getting a replacement sensor
If you are having CGM-related issues, please see each company’s customer support contact information and replacement policies below:
Abbott
February 6. 2026 Statement by Abbott
“Abbott initiated a medical device correction on Nov. 24, 2025, for certain FreeStyle Libre 3 and FreeStyle Libre 3 Plus sensors in the U.S. after internal testing determined that some of these sensors may provide incorrect low glucose readings. We immediately contacted customers to make them aware and provide support.
The U.S. Food and Drug Administration issued an early alert on Dec. 2 and classified this as a Class I recall on Jan. 14, 2026.
People can visit www.FreeStyleCheck.com to check if their sensors are potentially affected and request a replacement. No other Libre products are impacted.
Abbott has identified and resolved the cause of the issue, which relates to one production line among several that make Libre 3 and Libre 3 Plus sensors. The company continues to produce Libre 3 and Libre 3 Plus sensors to fulfil replacement and new orders and does not expect significant supply disruptions.”
Sensor replacement and troubleshooting information from Abbott:
“Abbott offers a generous, no-charge warranty replacement program for its FreeStyle Libre CGMs, which can be mailed directly to you or picked up at a retail pharmacy. Abbott’s U.S. customer care team is available by chat 24/7 or to speak over the phone 7 days a week from 8 a.m. to 8 p.m., excluding holidays. For the quickest response, you can also use the online sensor support form.”
Ways to get a FreeStyle Libre sensor replaced:
- Call (Toll-Free): 1 (855) 632-8658
- Available 7 days a week from 8 a.m. to 8 p.m. ET, excluding holidays
- Chat online
- Available 24/7, excluding holidays
- Use online sensor support form
- Available 24/7
- Any calls made to Abbott’s pump partners are supported by Abbott agents 24/7, every day including holidays
Support resources can also be accessed directly through the latest Libre App.1
- Click on the Profile icon on the bottom navigation bar
- Click “Help”
- Scroll to the Support section and click “Customer Service”
- Click “Start a sensor support request”
- If your sensor is covered under Abbott’s warranty replacement policy, you can choose to receive:
- Free shipment to your location, or
- An electronic voucher to redeem at your pharmacy for a free replacement (only available if you use a retail pharmacy to pick up your sensors)2
Additional Resources
- Access answers to frequently asked questions and Libre support guides here.
1. The FreeStyle Libre systems apps are only compatible with certain mobile devices and operating systems. Please check the Support section of Abbott’s website for more information about device compatibility before using the apps. Use of the FreeStyle Libre systems apps may require registration with LibreView.
2. Participating pharmacies are subject to change without notice. Product availability may vary by retailer.
Dexcom
Statement by Dexcom:
“Dexcom provides sensor replacements for confirmed product failures. These replacements are shipped directly from Dexcom and can be expedited if customers need the product urgently. For customers who obtain their Dexcom supplies from a pharmacy, a voucher may be issued for same day replacement pickup at their local pharmacy. Dexcom customer support is here: https://www.dexcom.com/support.
If you need immediate assistance, call 1 (844) 607-8398 to speak with a tech support agent. They are available 24/7.”
Additional links:
- Phone: 1 (888) 738-3646
- Request a replacement sensor
- Virtual education: Dexcom CARE – Get started with your Dexcom CGM | Dexcom
- Patient assistance program: Dexcom PAP – Welcome to the Dexcom Patient Assistance Program
Medtronic
- Phone: 1 (800) 646-4633, menu option 1
- Customer support
- Request a replacement sensor
Senseonics
- Phone: 1(844) 736-7348
- Customer support
What to do if you run out of sensors
Replacement sensors:
- Contact the manufacturer for a replacement and in an emergency, your diabetes care team may have samples on hand that they can share with you.
Have a backup plan:
- You should always have a blood glucose meter and non-expired blood glucose test strips on hand as a backup.
- If your symptoms don’t match the reading on the sensor, double-check using a blood glucose meter, as is consistent with all manufacturer’s instructions.
- Contact your diabetes care team or the manufacturer’s customer support team for advice if meter and CGM values differ by more than 20%.
- When using a blood glucose meter, be sure to wash your hands first and use non-expired test strips.
Cost
- Explore the Diabetes Technology Affordability Tool from ADCES.
- Children in the state of Florida can contact Home – Help A Diabetic Child for emergency assistance with supplies.
Clinic-based education
- You should receive education on how to use and troubleshoot your CGM from your diabetes care team. Often, this education can be provided during your clinic visit or as a separate education visit.
Strategies to improve accuracy
- Hydrate. Sensor values may be off when you are dehydrated.
- For Dexcom only, calibrate within the first 24 hours.
- Note: Libre devices cannot be calibrated.
- Be vigilant! Remember, if symptoms do not match the number on the sensor, use a blood glucose meter.
- If the sensor and meter continue to report values with a difference of more than 20%, change the sensor and request a replacement.
- Putting a sensor on is relatively easy, but using it effectively requires education. Obtain education from your diabetes care team.
Register issues
- These sensors are all regulated by the U.S. Food and Drug Administration (FDA). If you have product concerns that are unable to be resolved through each company’s customer service line, you can report those concerns to the FDA at https://www.fda.gov/safety/report-problem-fda.
Find your chapter
- You can always contact your local Breakthrough T1D chapter for local support and guidance. Find your chapter.
Nearly 3 years after Breakthrough T1D joined forces with Civica, a non-profit pharmaceutical company, its first insulin is now available for purchase at pharmacies across the country.
Check out our story from October that highlights the years of partnership between Civica and Breakthrough T1D that made today a reality.
We worked with our partners at Civica to answer some of the most common questions you may have about this insulin and where to get it.
What is it called?
Civica insulin glargine-yfgn. It will have a “CalRx” label in California.
What insulin is for sale?
Civica insulin glargine-yfgn is a long-acting insulin interchangeable with Lantus.
Where can I get it?
Broad availability is expected at U.S. pharmacies as production increases. Ask your pharmacist.
How do I get it?
If you have health insurance:
- Check your plan’s formulary to ensure Civica insulin glargine-yfgn is listed. If so, just ask your pharmacy to order it when you take in (or refill) your prescription.
If you are paying cash:
- Cash-paying patients can buy Civica insulin glargine-yfgn at the manufacturer’s recommended price, without any coupons or patient assistance program needed. Just ask your pharmacy to order it when you take in (or refill) your prescription.
For Californians:
- Civica’s insulin glargine-yfgn is available under the CalRx brand in California pharmacies.
How much does it cost?
Max recommended price: $55 per box of five pens. Your cost may be lower depending on insurance. This is the lowest list price for long-acting insulin, without hidden rebates or markups.
Do I need a prescription?
Yes. If you already have a prescription for insulin glargine (e.g., Lantus), you do not need a new one because Civica insulin glargine-yfgn is interchangeable. Confirm with your provider and pharmacist.
Can I buy a single pen?
No. Pens are sold in boxes of five pre-filled pens.
Is it covered by insurance?
Yes, but you don’t need insurance to buy it. Out-of-pocket costs could be as low as $0 depending on your plan.
You can learn more about Civica insulin at CivicaInsulin.org.
Mason Miller is a relief pitcher for the San Diego Padres. He’s known for his fastball (often 100+ mph!) and cutter/slider, earning praise as one of baseball’s best relievers.
Breakthrough T1D sat down with Mason to learn more about his journey with type 1 diabetes (T1D) and the impact it has on him as a star athlete.
Q: What’s your T1D story?
A: I’ve now lived with type 1 diabetes for almost 8 years. I was diagnosed at the age of 20 during my sophomore year of college. I had lost about 25 pounds and was exhibiting all the tell tale signs, but thought no way it could be T1D. I was active, ate fairly well, why would it happen to me? Regardless, I was sent to Children’s Hospital in Pittsburgh, where I was diagnosed and educated on T1D. I really had no knowledge prior to my diagnosis.
I learned the ins and outs of T1D quickly, but I do feel that living and learning has given me more and more confidence as I tackle it with my daily endeavors. Getting healthy and seeing a huge weight gain, I was able to have a ton of success on the baseball field. When I was diagnosed, I was struggling as a D3 pitcher and now I’m pitching in the MLB for the Padres. A lot of hard work accompanied getting healthy and educated, but I do feel grateful for the technology that allows me to take such good care of myself.
Q: Why is T1D visibility important to you?

A: Visibility is important to me because of the platform I have as an athlete. I’m fortunate to have an opportunity to draw attention to things that matter to me. Type 1 diabetes is close to my heart as I live with it each day. I have a unique opportunity to connect with young kids and show them that T1D does not hold you back. You can still have and achieve dreams just like everyone else.
Q: What is one T1D misconception that you REALLY dislike?
A: That lifestyle, diet, or something I did caused my diagnosis.
Q: What is something you wish people knew about life with T1D?
A: The habits and routines that we develop mean a lot. With everyone, healthy habits are important but for people living with T1D, it’s crucial. Physical activity and diet go a lot further than physique or stamina. They help us create stable sugar levels in healthy ranges.
Q: Is there a routine, tool, or mindset that’s been especially helpful for you on game days?
A: My continuous glucose monitor (CGM) is a game changer. You learn so much about how different things affect you. It gives me the confidence to play without a second thought because I know my glucose and how it will react.

Q: How do you stay mentally strong while juggling a professional sports career and T1D?
A: Having a support system is incredibly important. My wife is, and will continue to be, my biggest supporter. But coaches, teammates, and others all can teach you something from their words or actions. I try to always be learning from those around me. I handle T1D like it’s normal for me. I still expect those around me to hold me to a high standard. It’s not a crutch. It’s an opportunity.
Q: What message do you hope your story sends to young athletes with T1D?
A: I always hope that my story is an inspiration to those living with T1D. Whether they have aspirations in sports or elsewhere, I hope my story gives them confidence to chase their dreams.
What’s happening?
Continuous ketone monitoring (CKM) is on its way. A new paper in The Lancet Diabetes & Endocrinology, titled “International expert recommendations on the application and utility of continuous ketone monitoring for people with diabetes,” was published by Breakthrough T1D and expert collaborators, spearheaded by our Chief Medical Officer, International, Thomas Danne, M.D., Ph.D. This publication summarizes the recommendations for effective use CKM use in people with type 1 diabetes (T1D) at risk of developing diabetic ketoacidosis. Read on to learn more about this technology, guidelines for its use, and what this means for the future.
Ketones and DKA: What you need to know
When there is not enough insulin in the body, glucose cannot be used for energy, and the body uses fat for fuel instead. Excessive breakdown of fat ultimately causes ketones to build up in the blood. Very high levels of ketones in the blood can lead to diabetic ketoacidosis (DKA)—a life-threatening condition that requires immediate medical treatment.
Too often, people are diagnosed with T1D only after they have experienced DKA. Yet, even for people who are aware that they have T1D, DKA can and does happen. There are many reasons that it can occur, including the onset of illnesses, like the flu. Currently, people with diabetes use blood and urine strips to test for ketones—but even so, surveys show that very few people measure their ketones regularly, and DKA rates remain unacceptably high.
Given the current options, it remains very difficult to predict when ketone levels may be rising, and people lose opportunities to intervene early to prevent DKA altogether. So, how can we fill this gap?
The answer: continuous ketone monitoring.
CKMs are coming—but they’re not here yet
CKMs are in the works, and we can expect them to become a reality sooner rather than later. Similar to continuous glucose monitors (CGMs), CKMs will continuously measure ketone levels in the body and can alert users if their ketone levels are rising. This ability to track ketone levels in real-time could have substantial benefits in preventing DKA—and more.
Examples of people with T1D who may benefit from CKM
- Insulin pump users, including automated insulin delivery (AID) users, who are at higher risk for DKA due to possible infusion-set or infusion-site failures
- People with T1D and complications, such as chronic kidney disease or cardiovascular disease, who have an increased risk of DKA
- Pregnant women with pregestational or gestational diabetes, who may have increased resistance to insulin and therefore higher rates of DKA
As of today, there are academic groups and early-stage companies working on CKM. Abbott also has a glucose-ketone sensor (CGM-CKM) in development. Other companies, including Tandem, Beta Bionics, Sequel Med Tech, and Ypsomed, are planning to integrate Abbott’s ketone-glucose sensing device into their AID systems once it is available. We’re also keeping an eye a company called SIBIONICS, which currently offers a wellness-focused CKM that has demonstrated strong ketone-tracking data, although it is not available as a medical device yet.
Breakthrough T1D strongly supports the development of CKM to help protect people with T1D from the dangers of DKA. We previously funded five projects—two of which are co-funded by the Helmsley Charitable Trust—to support the development of CGM-CKM. Now, we’re getting ahead of the curve for CKM so we can learn how to use this technology to answer clinical questions about T1D.
We are currently looking for and investing in research projects to understand how ketone monitoring may improve outcomes for people living with T1D from multiple angles, including DKA prevention, understanding disease progression and pathophysiology, learning from clinically meaningful ketone patterns, and more. The goal is to accelerate the generation of evidence surrounding the use of CKM while these devices are on the horizon. This way, we can get a better idea of the kinds of benefits this technology can bring to the T1D community.
Snapshot of CKM recommendations for people with T1D
We feel optimistic that CKMs will become a reality soon. The next question is: how should they be integrated into T1D care? To answer this question, Dr. Danne and an international Breakthrough T1D consensus group published a report about how these novel technologies can be effectively used to help the T1D community.
The experts agreed on the following recommendations:
- CKMs should include trend arrows, akin to those on a CGM, and reflect rates of change of around 0.4 mmol/L per hour.
- Due to the existence of alarm fatigue, alarms should sound if ketone concentrations rise above the threshold of ≥3.0 mmol/L.
- The terminology for ketone levels should be: Normal, Elevated, High, Urgent High.
- All CKM users should be provided with a blood ketone meter to use if they are experiencing the symptoms of High or Urgent High ketones that do not match the CKM reading.
- All individuals who wear a CKM should receive education on what elevated ketones mean and what actions to take.
These are just a few of the highlights. Check out the full publication for a more in-depth look at what the experts recommend.
Why CKM guidelines are important
CKMs have the potential to transform T1D care by stopping dangerous, life-threatening DKA before it ever occurs. It is critically important that healthcare professionals (HCPs) and people living with T1D know how to most effectively use this technology to maximize its benefits.
This publication does just that. It’s a set of established guidelines, put together by experts from around the globe, to help standardize the integration of CKM into clinics once they become available. Both HCPs and the T1D community can refer to these recommendations as this technology is rolled out—and potentially becomes the standard of care—to start realizing the benefits of CKM as soon as possible.
Even more, this document can be used as a resource in future conversations surrounding regulatory decisions and payer coverage. Ultimately, we are preparing all stakeholders for a future that we know will be here soon—one where CKM is a valuable addition to daily T1D management.

“Experts from 12 countries agree: continuous ketone monitoring can be a game-changer. By detecting risk earlier and guiding faster action, CKM has the potential to reduce DKA, strengthen confidence for people with T1D, and transform how we deliver safe, proactive diabetes care.” – Thomas Danne, M.D., Ph.D., Chief Medical Officer, International
How this fits into our Medical Affairs goals
CKMs are coming—how can we ensure they make their way into clinics around the globe? By preparing the T1D community in advance of their arrival, we are setting the stage for CKMs to come. That way, once they are available, HCPs and people with T1D will already know what to do.
In essence, this is clinical adoption: the cornerstone of our Medical Affairs unit. We are preparing the right people at the right time by providing resources, education, and guidelines for a new technology. We are getting our ducks in a row before the new technology is here—so that we are 100% ready when it is.
Thanks to Dr. Danne and the expert consensus panel, we can be ready-as-ever for CKMs to materialize. We look forward to a future in which DKA is easily prevented and the T1D community can fully experience the benefits of continuous ketone monitoring.
Breakthrough T1D’s commitment to supporting the entire T1D community includes providing you with information and resources to help you navigate life with type 1 diabetes.
Continuous glucose monitors (CGMs) have helped millions of people better manage their diabetes. But we are aware that, at times, people in our community may experience issues with their CGMs. This can include inaccurate readings, faulty sensors, customer service issues, and more. If you are having an issue with your CGM or if your symptoms do not match the reading on your CGM, you should always follow the manufacturer’s guidelines and check your blood sugar with a glucometer.
Our community relies on these devices, and it is critical that they work as intended. If you are having issues, please see the contact information for each company’s customer service line where you can receive technical support, request a replacement sensor, and more.
Abbott
- Phone: 1 (833) 815-4273
- Customer support
- Request a replacement sensor
Dexcom
- Phone: 1 (888) 738-3646
- Customer support
- Request a replacement sensor
Medtronic
- Phone: 1(800) 646-4633, menu option 1
- Customer support
- Request a replacement sensor
Senseonics
- Phone: 1 (844) 736-7348
- Customer support
These sensors are all regulated by the U.S. Food and Drug Administration (FDA). If you have product concerns that are unable to be resolved through each company’s customer service line, you can report those concerns to the FDA here.
How quickly a year goes by—and a lot has happened for type 1 diabetes (T1D) in 2025! In these last few weeks leading up to the new year, Breakthrough T1D would like to take a moment to reflect on how far we’ve come in the past 365 days. Read on for the greatest T1D accomplishments in 2025—and the exciting things we’re looking forward to in 2026.
A special note for our donors
Breakthrough T1D would like to give a special thank you to our donors. Each and every one of us —staff, volunteers, T1D community members, researchers, advocates, clinicians, and more—are able to accomplish what we can because of you. Our shared commitment to a world without T1D is driven by your generosity, and truly no words can adequately express our appreciation for your continued support of our mission. Thank you for all that you do!
Check out our Top Advances 2025 video featuring Breakthrough T1D CEO Aaron Kowalski, Ph.D., and read on for more details!
Cell therapy milestones
First things first: cell therapies. One of the biggest breakthroughs that had the T1D community buzzing in the last year was Sana Biotechnology’s donor-derived islets that are genetically engineered to be invisible to the immune system. These cells are still making insulin 6+ months after the first person was treated—without immunosuppression! Sana—a portfolio company of the T1D Fund: A Breakthrough T1D Venture—published this accomplishment in The New England Journal of Medicine.
On the manufactured cell therapies front, Vertex Pharmaceuticals continues to hit milestones with zimislecel (formerly VX-880)—a therapy that Breakthrough T1D has had a hand in for decades. Promising results from the phase 1/2/3 clinical trial were published in The New England Journal of Medicine, revealing that 10 of 12 participants (83%) are insulin therapy independent. The study is ongoing.
Tegoprubart, an immunomodulatory drug made by the T1D Fund portfolio company Eledon, is being investigated as an alternative to standard immunosuppressants to help prevent transplanted islets from rejection. In a Breakthrough T1D-funded trial, six individuals who have received a donor islet transplant and tegoprubart are now insulin therapy independent. Preliminary data suggest that tegoprubart is more tolerable and has milder side effects compared to standard immunosuppressants—paving the way for easier post-transplant management. The study is ongoing, and nine people total are enrolled. The promising results from this trial opened the doors to another Breakthrough T1D-funded study that will include people with T1D and chronic kidney disease (CKD)—a population that was previously excluded from islet transplants due to side effects associated with standard immunosuppressants.
Cell Pouch is an implantable device that provides a livable environment for transplanted islets to help them survive. The ongoing phase 1/2 trial investigating Cell Pouch with donor-derived islets is on track to meet its endpoints for safety and tolerability, and people are achieving insulin therapy independence. The final cohort of this trial, which is expected to initiate before the end of this year, will use manufactured islets provided by Sernova’s partner Evotec. Even more, this cohort will receive Eledon’s tegoprubart instead of standard immunosuppressants—accelerating progress on two fronts at once. Breakthrough T1D has been supporting the development of Cell Pouch over many years, and we look forward to seeing data from the final cohort.
Vertex and large-scale manufacturing
While zimislecel is in the works, Vertex has forged collaborations to manufacture these cells at a large scale once they are approved. They’ve partnered with Lonza to build a dedicated large-scale facility for eventual commercial production of zimislecel. They’ve also licensed technology from TreeFrog to scale up production capabilities. We call that planning ahead!
Civica’s low-cost, long-acting insulin available on January 1, 2026
Thanks to a huge win for insulin affordability, the first day of 2026 will kick off with a bang. Civica Rx’s insulin glargine-ygfn (which is interchangeable with Lantus®), will be available on January 1, 2026, for no more than $55 for five pens. Anyone with a prescription—regardless of insurance status—will be able to purchase it at a pharmacy. Breakthrough T1D has been working with Civica for nearly three years to make this possible.
Positive outlook for disease-modifying therapies
Building on results from a promising study funded by Breakthrough T1D, Eli Lilly launched two new pivotal clinical trials for baricitinib, a JAK inhibitor that is already approved for other autoimmune diseases. The trials will test whether baricitinib—which is taken as a once-daily oral pill—can delay the progression of stage 2 to stage 3 T1D (BARICADE-DELAY) or preserve beta cells in newly diagnosed stage 3 T1D (BARICADE-PRESERVE). Enrollment will open in 2026.
Another disease-modifying therapy is coming down the pike: ATG. Following from prior studies, the phase 2 MELD-ATG clinical trial found that low-dose ATG has the potential to preserve insulin-producing beta cells in children and young adults between five and 25 years old, and it was generally well-tolerated. In line with ATG’s potential, SAB Biotherapeutics—a company with funding support from the T1D Fund—is developing a next-generation ATG therapy.
Tzield, the first disease-modifying therapy approved to delay stage 3 T1D in people eight years and older in stage 2 T1D (before insulin therapy is required), has been accepted into the FDA Commissioner’s National Priority Voucher (CNPV) program for accelerated review. If approved, this could lead to expanded use of Tzield for people with newly diagnosed stage 3 T1D (when insulin therapy is required)—and this would be the first time people at this stage could have a therapy option besides insulin. We look forward to hearing more soon.
Right now, Tzield is only approved for use in delaying progression from stage 2 to stage 3 T1D for kids and adults eight years and older. The PETITE-T1D clinical trial is trying to change that. Interim results showed that Tzield is safe and well-tolerated in children under eight with stage 2 T1D, with no new safety issues reported. The study is ongoing and final results are expected in 2026.
Tzield in action: Breakthrough T1D’s Chris Dunn
Chris Dunn is a parent of four children, two of whom live with T1D. Knowing that immediate family connections are a risk factor for developing T1D, Chris and her children without T1D participated in screening. Much to her surprise, Chris learned that she was in stage 2. Chris knew that Tzield could potentially delay her need for insulin therapy, so she made the informed decision to try it.
A big win for kidney disease, plus new trials, devices, and more to improve the lives of people with T1D
Bayer shared data from the phase 3 FINE-ONE clinical trial, which investigated finerenone (Kerendia®)—a drug that is already approved for type 2 diabetes—for CKD associated with T1D. The results showed that finerenone significantly reduces a marker of kidney damage, marking the first treatment in nearly 30 years to achieve positive outcomes for CKD in people with T1D. This data will be submitted for regulatory review to expand finerenone’s indication to include T1D.
The Breakthrough T1D-funded phase 2 ADJUST-T1D trial investigated whether the GLP-1 receptor agonist semaglutide (Ozempic®) could benefit people living with T1D and obesity or overweight who are using an automated insulin delivery (AID) system. Thirty-six percent of people in the trial treated with semaglutide met the primary endpoints for glycemic and weight control compared to 0% in the placebo group, and the drug was well-tolerated and safe. These data—which were published in The New England Journal of Medicine Evidence—show that GLP-1 receptor agonists have the potential to help people with T1D manage both blood sugar and weight.
Building on the growing interest in GLP-1s for T1D, Eli Lilly launched two pivotal clinical trials for tirzepatide (Mounjaro® or Zepbound®), a dual GLP-1/GIP receptor agonist. These trials—which are enrolling now—will investigate whether tirzepatide can reduce blood sugar levels in people living with T1D and obesity or overweight over a period of 40 weeks (SURPASS-T1D-1) or 20 months (SURPASS-T1D-2). Industry investment in GLP-1s for T1D is a promising avenue toward a future where this class of drugs may be approved for use in members of the T1D community.
AID systems are getting better each year, and approximately 10 such systems are now approved for use in the United States. Here are a few advances in diabetes technology that stood out:
- Dexcom’s G7 15-day sensor—the longest-lasting FDA-approved wearable continuous glucose monitor (CGM) on the market—is now available for people in the U.S. 18 and over.
- Sequel Med Tech’s twiist AID system, which uses FDA-cleared Tidepool Loop technology, is now available in the U.S.
- Tandem’s SteadiSet 7-day infusion set has been cleared by the FDA and is expected to launch in the U.S. in 2026.
- Abbott’s Freestyle Libre 3 Plus sensor has integrated with twiist and T:slim X2 insulin pumps.
- Medtronic unveiled the largest real-world dataset pulled from users of the MiniMed 780G AID system.
To better understand the mental health needs of the T1D community, Breakthrough T1D and the Helmsley Charitable Trust co-hosted a Psychosocial Roundtable, convening mental and behavioral health experts across the T1D care spectrum. The discussion revolved around challenges and gaps in psychosocial care for people with T1D and how we can work together to address them. Thanks to the expert opinions of the attendees, Breakthrough T1D has a clear path forward for tackling these issues and supporting the mental and emotional wellbeing of the T1D community through research and awareness.
Making headway in rebalancing the immune system in T1D
Sanofi has entered into a licensing agreement with EVOQ Therapeutics, a company with support from Breakthrough T1D, to help develop and commercialize its NanoDisc technology. This technology has the potential to retrain the immune system to stop attacking beta cells. Industry partnerships like these are key to accelerating T1D research.
The Nobel Prize in Physiology or Medicine was awarded to three immunologists—Fred Ramsdell, Ph.D., Mary E. Brunkow, Ph.D., and Shimon Sakaguchim M.D., Ph.D.—for their discoveries in immune tolerance, meaning the immune system’s ability to distinguish between self and non-self. Building on these breakthroughs, Dr. Ramsdell co-founded Sonoma Biotherapeutics, a T1D Fund-backed company, to restore immune tolerance in autoimmune diseases like T1D. Another year, another researcher with ties to Breakthrough T1D winning the most prestigious award in science!
Launch of Breakthrough T1D’s Medical Affairs department
A noteworthy organizational change is marked by the launch of Breakthrough T1D’s Medical Affairs unit, which aims to bridge the gap between access to and adoption of T1D therapies, devices, and treatments. The team—which continues to expand—is tackling challenges associated with slow clinical adoption of T1D therapies. In less than a year, the team has already launched healthcare professional (HCP) education and resources and hosted two international workshops at Breakthrough T1D HQ focused on preparing the clinical workforce to bring next-generation cell replacement therapies to the T1D community. This is just the beginning, and we can’t wait to see what else our Medical Affairs unit will accomplish!
Continuous ketone monitoring consensus guidelines published
Continuous ketone monitoring (CKM) has substantial benefits for the T1D community, including preventing diabetic ketoacidosis. CKMs are coming—and to prepare, Breakthrough T1D’s Chief Medical Officer, International Thomas Danne, M.D., Ph.D., spearheaded an effort with expert collaborators to publish consensus guidelines for use of CKM in T1D. These guidelines will help HCPs integrate CKM into clinics and establish best practices for their use in the T1D community.
Tackling T1D on a global scale
Breakthrough T1D, in partnership with the Helmsley Charitable Trust and Roche and Sanofi’s Global Health Unit, launched ALIGN-T1D. This is a new global alliance uniting philanthropic, industry, and community leaders to strengthen government-led T1D care networks in low-middle income countries, integrating diagnosis, insulin access, monitoring, and education.
Breakthrough T1D helped organize an event at the European Parliament to highlight unmet needs for the T1D community and discuss how to accelerate cures—especially cell therapies—in the European Union. In a similar vein, an event co-hosted by Breakthrough T1D in Brussels, Belgium sparked conversation about overcoming challenges surrounding cardiovascular disease in T1D. Building long-term partnerships with EU Institutions and international diabetes organizations will allow us to work together towards global T1D breakthroughs.
Breakthrough T1D, in partnership with Friends of Mewar and UNICEF, participated in the Udaipur Type 1 Diabetes Summit: Advancing Access, Equity, and Action, in Udaipur, Rajasthan, India. The event gathered government leaders, health experts, community advocates, and international partners to develop a collaborative roadmap for strengthening T1D care across India.
Breakthrough T1D, Breakthrough T1D Canada, and the Stem Cell Network partnered to support four new projects led by Canadian researchers to drive high-impact research into manufactured cells and next-generation cell replacement therapies. This partnership is a novel and meaningful part of Breakthrough T1D’s global Project ACT effort to power high-impact cell therapies research.
The Australian government committed $50M over five years to the T1D Clinical Research Network (T1DCRN), a highly impactful research network driven by Breakthrough T1D Australia. This funding will support T1D research in prevention, precision medicine, and cures.
The Grand Challenge, a partnership between Breakthrough T1D UK, the Steve Morgan Foundation, and Diabetes UK, is funding 23 research projects to advance new treatments and cures for T1D. Their funding is supporting 189 researchers and collaborators across 49 institutions in eight countries. Recent breakthroughs from the past year include uncovering why T1D could be more aggressive in young children and developing a new insulin-glucagon molecule that could reduce dangerous hypoglycemic events.
Breakthrough T1D publications across the map
Breakthrough T1D Research and Advocacy staff, in collaboration with other leading experts in the field, authored a paper titled “Future Directions and Clinical Trial Considerations for Novel Islet Beta Cell Replacement Therapies for Type 1 Diabetes” in the journal Diabetes. This publication outlines what the future of beta cell replacement therapies looks like—and how we can make these therapies a reality for everyone with T1D who wants them through innovative clinical trial design and expanding the pool of eligible trial participants.
“The Urgent Need for Breakthrough Therapies and a World Without Type 1 Diabetes” was authored by Breakthrough T1D staff and leadership and published in Diabetes Therapy. This commentary exposes the many persistent challenges of living with T1D, despite incredible progress in recent years. The publication is a call to action for the entire T1D community—researchers, clinicians, advocates, people with T1D, regulators, and all other stakeholders—to work together to overcome barriers at all levels to make cures for T1D a reality sooner rather than later.
Breakthrough T1D Advocacy staff and collaborators published a paper titled “Perceptions of the Benefits and Risks of Novel Therapies for Type 1 Diabetes: A Qualitative Study” in the journal Diabetes Therapy. This Breakthrough T1D-funded study found that people with T1D and their caregivers are largely willing to try next-generation therapies, drawn by the promise of reduced reliance on insulin and freedom from constant disease management. These findings highlight the importance of ensuring that the perspectives of people with T1D and their caregivers are used to guide regulatory decisions around the benefits and risks of a new therapy.
Breakthrough T1D Advocacy staff and collaborators published an article called “We Are on the Verge of Breakthrough Cures for Type 1 Diabetes, but Who Are the 2 Million Americans Who Have It?” in The Journal of Health Economics and Outcomes Research. This study identifies key T1D demographics in the U.S. and predicts how these demographics may change over the next decade. These data will help inform elected officials and payers about how healthcare policies may ultimately affect the T1D community—ensuring that the policies in place are having the greatest impact they can.
An analysis by Avalere Health, which was supported by Breakthrough T1D, found that research funded by the Special Diabetes Program (SDP) has yielded more than $50 billion in federal healthcare savings. This study supports the fact that the SDP has a strong return on investment, both clinically and economically, and demonstrates its ever-important role in bringing advanced therapies to the T1D community and improving health outcomes. Even more, SDP funding was recently extended through January 30, 2026, with retroactive funding back to October 1, 2025.
Consensus guidance on population screening for T1D coming soon
Breakthrough T1D’s Vice President of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, spearheaded an effort to establish a consensus on T1D screening guidance. These guidelines—which will be published soon—push for population-level T1D screening and provide guidance for HCPs to effectively integrate T1D screening into their clinics. The publication will outline the benefits, harms, and methods of T1D screening, who should be screened and how often, and how to effectively communicate screening results.
Breakthrough T1D announces first Clinical and Research Congress in October 2026
In a historic first, Breakthrough T1D is hosting our very own Clinical and Research Congress next fall! This meeting will bring together clinicians, researchers, scientists, HCPs, industry leaders, and the T1D community to explore cutting-edge advances in T1D research, clinical care, health equity, and more. The goal is to foster collaboration and spark innovation to drive our mission forward faster than ever.
Bonus advance: The first Barbie doll with T1D
In December 2023, Mattel approached Breakthrough T1D with a partnership opportunity for the first Barbie with T1D. Less than two years later, in July 2025, Barbie with type 1 diabetes was launched to a most enthusiastic reception from Delegates at Breakthrough T1D’s Children’s Congress. This groundbreaking global collaboration reflects a shared commitment to ensuring that the millions of people living with T1D are seen, heard, and empowered. It’s working—the dolls have been in high demand since launch!
One word: WOW! That’s a lot of exciting things that YOU helped make possible. Thank you for your support of Breakthrough T1D and everything you do to drive our mission forward. Each year, each month, each day, we get closer to a world without T1D. This wouldn’t be possible without our donors, community members, staff, leadership, and every single heart and mind around the globe affected by the work we do each day. Cheers to a successful 2025—and here’s to another year of accomplishments!
Management of type 1 diabetes (T1D) has gotten much easier as the technology for checking blood sugars and dosing insulin has improved. Continuous glucose monitors (CGMs) worn on the body check blood sugars every five minutes, so that people with T1D no longer need to poke their fingers numerous times a day. Similarly, insulin pumps allow for insulin dosing without an additional needlestick for each meal or snack. Wearable devices are not only more convenient for many people with T1D, but they also lead to better glucose control with lower HbA1C levels and more time in range.
Skin irritation from T1D tech is a common issue
Over the last decade, more people have been using wearable devices, which is good for the overall management of T1D. However, with devices stuck to the skin for days at a time, skin reactions are relatively common across many different brands of T1D tech.
Mary Burgess, who developed a reaction to her patch pump, explained, “My skin was getting so irritated and itchy that my pump would literally fall off in my sleep. My glucose numbers were high because I was scratching off my pod and the absorption was awful because my skin was so bad.” She continued, “It added one more layer of this disease I have to deal with already and that really started to tire me out.”
Caitlin Stauffer, a parent to a teen with type 1 diabetes who developed a reaction to the adhesive on his CGM, described her son’s experience in similar terms, “It looked like a burn and a whole layer of skin would come off. It was painful, itchy, and just one more thing that made this disease seem unbearable.”
Shamron Cook, another parent to two children with T1D who have had skin reactions, summed up the frustration: “I want pump infusion sets, CGM sensors, and overpatches that stay in place, but don’t destroy my kids’ skin!”
Dr. Jessica Ferris and Dr. Radhika Purushothaman, pediatric endocrinologists at Randall Children’s Diabetes and Endocrine Center in Portland, OR, agreed that skin complaints from type 1 diabetes technology happen frequently. Dr. Ferris estimated that about half of her patients have skin issues from their device. Both endocrinologists said that they mostly see mild reactions, characterized by redness, itching, and dry skin. Severe complications are much less common, but they do occur. Dr. Purushothaman explained, “Very occasionally, I see more serious reactions like breakdown of the skin, bleeding, or redness that lingers for days.”
Why do skin reactions from T1D technology occur?
Skin problems caused by type 1 diabetes technology can develop for different reasons:
- The adhesive patch can trap sweat and moisture close to the skin.
- Friction from the patch and damage to the skin from patch removal also can lead to irritation. This is called irritant contact dermatitis.
- People may develop allergic contact dermatitis from an allergy to the patch adhesive and/or a part of the CGM or pump housing. Allergic contact dermatitis can result in significant inflammation and even blistering or skin breakdown. Acrylates and colophonium (rosin), which help create a strong adhesive, are common culprits in allergic skin reactions.
- Products used to prep the skin also can trigger an allergic reaction.
Dr. Susan Massick, Associate Professor of Dermatology at the Ohio State University Wexner Medical Center, cautioned, “Anything that contacts your skin could cause an irritation or allergic reaction. With any new product, I recommend trying things on a small test area first. And, since these devices are worn for longer and longer periods of time, maintain your test area for at least a week to see if there is any delayed reaction.”
Tips on managing T1D tech skin irritation
People with type 1 diabetes rely on their diabetes technology, and healthcare providers want to do all they can to avoid device discontinuation. If you are noticing skin irritation from your device, there are interventions that your doctor may recommend:
- As a first step, doctors often try a spray or cream on the skin to calm the inflammation.
- If that is not effective, then your doctor might suggest using a skin barrier wipe or a thin film dressing to create a buffer between the device and your skin.
- Doctors may further advise patients to avoid harsh skin preps, like alcohol wipes or adhesive enhancers, if they are not needed.
In the event of severe skin reactions, seeing a dermatologist for patch testing can be helpful. Dr. Massick explained, “Patch testing done by a board-certified dermatologist can confirm allergic contact dermatitis when positive. Negative patch testing may point to more of an irritant contact cause.”
Additionally, she suggested that you try contacting the device manufacturer. “Some manufacturers have formal reporting systems where you can request information on adhesives and alternative adhesive options,” Dr. Massick said. Another measure to consider is switching to a different brand of device. Dr. Purushothaman reported that she sometimes recommends that patients switch brands, but only as a last resort when other interventions have not helped. While changing brands can be an adjustment, it might help you avoid reducing device use or discontinuing devices altogether.
As always, be kind to your skin. Use gentle cleansers and moisturizers to keep your skin healthy, and make sure your skin is fully dry before applying the device. If you need to remove hair, trimming rather than shaving the hair can lead to better results. With some simple measures, most people will be able to comfortably wear their pumps and CGMs while we work towards the day that the T1D community can remove their devices for good!
Written by Breakthrough T1D Guest Contributor Laura Tremblay