
Project ACT series
This article is part of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) will shape the future of cell therapies for type 1 diabetes (T1D). The next article in the series will discuss the progress of cell therapies in clinical trials.
We’ve made major progress in the development of cell replacement therapies for type 1 diabetes (T1D) over the past couple of decades. We know that manufactured islets, such as Vertex’s VX-880 (now Zimislecel), can restore insulin therapy independence and glucose control when implanted into people with T1D.
However, there is more work to do.
We need to make sure the cells survive and function, ideally without immunosuppression, and ensure that these therapies are accessible to everyone with T1D. Read on to learn more about where there’s room for improvement and what we’re doing about it.
Optimizing manufactured islets
Priority #1: Cell source
Current external cell sources that are not manufactured in the lab, such as those from deceased donor pancreases or a person’s own cells, are in extremely limited supply. The only FDA-approved cell therapy for T1D, Lantidra®, requires donor cells, and it can take up to three pancreases to get enough islets for one transplant. This is unsustainable and limits the number of people who can get this therapy.
We can consistently generate an unlimited source of islet cells in the lab. This way, we can make enough insulin-producing cells for everyone with T1D and have a single cell source to test and compare multiple strategies to protect them.
Breakthrough T1D is funding an initiative at the Advanced Regenerative Manufacturing Institute (ARMI) to scale up Dr. Jeffrey Millman’s protocol to generate unlimited manufactured islets in a reliable, automated, and reproducible way. Breakthrough T1D is also building a partnership with the Cedars-Sinai Biomanufacturing Center to accomplish this goal.
Priority #2: Cell survival
Implanted islets that move through the bloodstream can cause an inflammatory reaction, resulting in cell death. Alternatively, those that are implanted in devices that are cut off from the blood and immune system are unable to get nutrients and oxygen, again leading to cell death.
It doesn’t have to be the pancreas, but islets need access to nutrients and oxygen so that they can survive for long periods of time and produce insulin.
Breakthrough T1D is funding research to develop scaffolds, which are specialized biomaterials that islet cells can stick to and get nutrients and oxygen to help them survive. Similarly, islets implanted in encapsulation devices, such as Sernova’s Cell Pouch™, can access nutrients and oxygen while having the added benefit of being protected from the immune system. There are also various Breakthrough T1D-funded clinical studies that are investigating different places in the body for manufactured islets, including the omentum and abdominal wall.

Omentum
The omentum is a fatty tissue layer that surrounds and protects the organs in your abdomen. Researchers are testing it as a new implantation site for manufactured islets.
Priority #3: Cell protection
Like organ transplants, manufactured islet therapies from an external source are recognized by the immune system as “non-self,” leading to immune rejection. Currently available cell therapies require broad immunosuppressants that may come with unwelcome side effects, including increased risk of infection and malignancy and toxicity to kidneys, nerves, and islet cells themselves.
By swapping standard immunosuppressives with options that have less complications, more people with T1D will be able to access these therapies.
Research and clinical studies funded by Breakthrough T1D approach immune protection from many different angles. This includes genetically engineered islets that can evade immune detection, encapsulation devices, and immunomodulatory therapeutics that can dampen the immune response.
The future of manufactured islets and T1D cures
There’s a lot of promising solutions in the pipeline. What’s next? Unlocking access.
“We recognize that approval of cures is not a life-changing breakthrough if people do not have access to the therapy itself,” said Aaron Turner-Phifer, Senior Director of Health Policy at Breakthrough T1D. “Building on the experience gained from past breakthroughs, we are working now, across our Mission teams, to identify and remove any potential barriers to people accessing cell therapies.”

“While our work is just beginning, we’ve already conducted market analysis to identify clinical and payer barriers to give us clarity on where to start. We are directly engaging policymakers and health plans to educate them on T1D cell therapies. We’ve also launched data projects to begin to generate the types of data required to positively inform future policy and coverage decisions.”
Aaron Turner-Phifer
In the coming years, it’s likely that first-generation manufactured islet therapies will be available to people with T1D with severe hypoglycemia unawareness and will require broad immunosuppression.
Later, advancements in cell survival and immune protection—combined with the advent of more tolerable immune suppression approaches—will open the doors for more people with T1D to access these life-changing therapies.
Project ACT
Scientific progress takes time, money, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Without continuous support from the T1D community and its supporters, we would never have gotten this far. Breakthrough T1D looks forward to a future where manufactured islet therapies are a reality for everyone with T1D, and we will not stop until we get there.

When Tzield was approved by the United States Food and Drug Administration (FDA), the type 1 diabetes (T1D) community had real cause to celebrate: The first disease-modifying therapy for T1D had cleared one of the last major hurdles to becoming available.

Disease-modifying therapies
Also "DMTs" for short, these therapies prevent, slow, halt, or reverse T1D progression.
But once Tzield was on the market and covered by health insurance companies and other payers, a new hurdle emerged: a majority of healthcare providers across the country were unaware of the drug, let alone how to administer it.
The clinical guideline for Tzield infusion did not become available until a year and a half after the FDA approved the drug. To date, 500 people in the U.S. with early stage T1D have received Tzield. Compare that to the annual incidence rate of T1D in the U.S. according to the T1D Index:
According to a 2023 study in the journal Diabetes Technology & Therapeutics, Tzield isn’t the only advanced T1D therapy with a surprisingly low adoption rate.
The FDA approved the first artificial pancreas (AP) system in 2016. Less than a decade later, there are now eight such approved systems on the market. These systems—also called automated insulin delivery (AID) systems—lead to better T1D management and health outcomes—yet only 16 percent of people with T1D in the United States use them.
Similarly, the FDA approved Lantidra, the first donor-derived cell therapy for T1D, in 2023. To date, one person has received it.
Increasing adoption to improve health
Closing the gap between access to and adoption of T1D therapies is a mission priority for Breakthrough T1D.
“It’s similar to the question: ‘If a tree falls in the forest and no one is there, does it make a sound?’” said Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, Associate Vice President of Breakthrough T1D’s Community Screening and Clinical Trial Education programs. “In this case, if you have a cutting-edge new therapy but most healthcare providers don’t know about it, don’t prescribe it, and don’t know how to administer it, does it have an impact?”
The organization recently announced the establishment of a Medical Affairs unit. The team will address the numerous challenges contributing to the slow adoption of groundbreaking T1D therapies, delaying their life-changing potential for many people living with the disease.
Challenges we are addressing:
HCPs have much greater knowledge of type 2 diabetes—or T2D—which is more prevalent.
HCPs need comprehensive guidelines to support new, approved treatment options.
There are too few clinical environments with the equipment and expertise to administer advanced T1D therapies and treatments, such as new T1D devices, therapies that require infusions like Tzield, and treatments that require implantation, such as cell therapies.
There are too few endocrinologists and certified diabetes care and education specialists with knowledge and competency in advanced T1D therapies.

With the establishment of our Medical Affairs team, we are reaffirming our organization’s commitment to creating a world where every individual with type 1 diabetes has access to life-changing therapies. By addressing systemic barriers and fostering clinical readiness, Breakthrough T1D will be pivotal in driving the timely adoption of emerging therapies and transforming care.”
As part of this organizational change, the Community Screening and Clinical Trial Education team, led by Albanese-O’Neill, will be integrated into Medical Affairs.
The team will focus on developing education materials for healthcare professionals in the U.S. and around the world; empowering people with T1D to participate in shared decision-making with their healthcare teams about emerging T1D therapies; helping to establish and socialize clinical care guidelines tailored to regional needs; and expanding clinical trial participation through community activation and HCP education.
“We have been doing a great deal of work to expand our HCP education, T1D community screening, and clinical trial education programs for more than three years now,” said Albanese-O’Neill, who has been with Breakthrough T1D as a fulltime staff member since 2022. “Given what we are seeing with adoption rates and with Dr. Danne joining us, we are now putting all of this work together in one department with a more strategic approach.”

Empowering clinicians with education
The team recently launched comprehensive, expertly redesigned HCP education and training resources.
These resources—which are accredited, free-of-charge, and live or on-demand—offer a significant focus on early detection for the earliest stages of T1D, monitoring guidance for positive test results, clinical trial opportunities, and the latest on cutting edge T1D therapy research and development, including disease-modifying therapies and islet cell therapies.
While designed specifically for healthcare professionals who can earn 4.5 credit hours of continuing medical education, the resources are available to the public. The on-demand feature means busy healthcare professionals with schedules that include all kinds of shifts imaginable can access this turn-key resource on their own time.
For a deeper dive, Breakthrough T1D’s resources will also offer live sessions, allowing time to interact with and learn from leading experts in the T1D field, including Albanese-O’Neill and Danne, in addition to those affiliated with different clinical facilities and institutions across the nation.

Our goal is to make this education as accessible as possible.”
Detecting T1D before symptoms present
A simple blood test can detect T1D in the earlier stages—before obvious symptoms develop. The biggest challenge is educating clinicians and the general population about it.
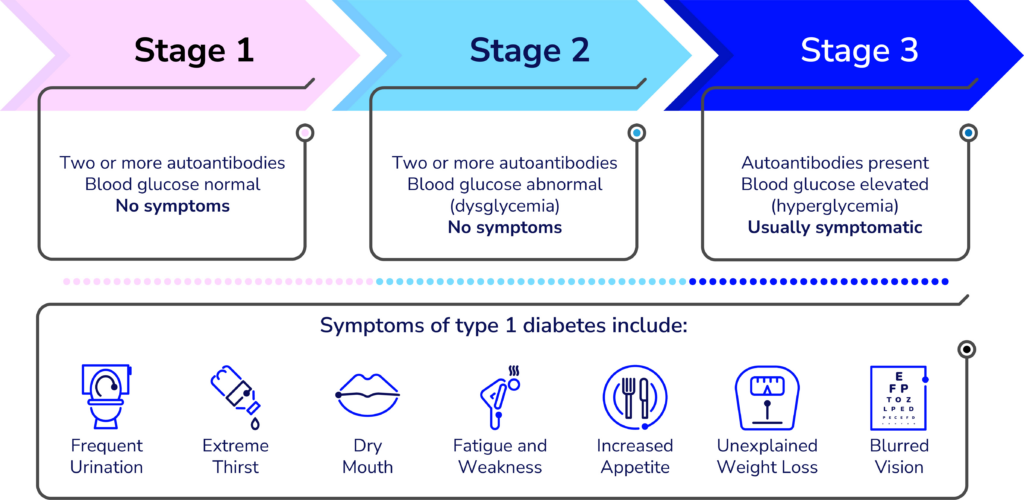
“Endocrinologists, Pediatricians, and some other specialty physicians learn about T1D screening and monitoring during their residencies, but it’s not a part of the general curriculum of the first four years of medical school,” said Lally, who built the learning management system for the new resources and is organizing the virtual offerings. “We’re working to advance that knowledge to yes, doctors, but also other clinicians whose patients could benefit.”
Many clinicians hesitate to order unfamiliar tests—especially if they are unsure what to do with the results. Most people who see any kind of healthcare provider could benefit from screening for T1D—according to a paper published in the journal US Endocrinology, roughly 85 percent of people diagnosed with type 1 do not have a blood relative with the autoimmune disease.
“Clinicians need to learn about the stages of type 1 and the specific autoantibody tests that identify type 1 versus type 2 and identifying type 1 in individuals at risk before they need insulin,” said Colleen Buggs-Saxton, M.D. Ph.D.
Buggs-Saxton, a Pediatric Endocrinologist at Wayne Pediatrics in Michigan, is the clinical leader of a Breakthrough T1D Early Detection pilot clinic. Using the new resources, she and Albanese-O’Neill are going to lead a grand rounds about T1D early detection at her institution, which is affiliated with the Wayne State University School of Medicine and healthcare system.

Clinicians should consider autoantibody testing for adults who have been diagnosed with type 2 but don’t have typical clinical features and require insulin to manage their blood sugars.”
“This is a novel way these resources can be used—as the basis of a locally and or virtually-provided grand rounds,” said Albanese-O’Neill.
While much of the emphasis of T1D early detection programs has been on children and teens, its applications are much broader—anyone can develop T1D at any age and unfortunately, misdiagnoses happen. According to an article published in the journal The Lancet, Regional Health: Europe, it is estimated that nearly 40 percent of adults older than age 30 with T1D may have been misdiagnosed with T2D.
“Most clinicians are very comfortable ordering an HbA1c test to classify people with type 2 diabetes, but they do not know what tests to order to classify people with type 1,” added Buggs-Saxton.

Grand rounds
Grand rounds are educational meetings and presentations for clinical teams at a given institution or healthcare facility to provide a summary of updates to the standards of care.

What to do with positive test results
The screening test is just the first part of T1D early detection. Clinicians also need to know what to do with positive results once they come in. Breakthrough T1D’s HCP resources offer extensive education on the topic.
Less than one year ago, Breakthrough T1D and other leading diabetes organizations developed monitoring guidance to help clinicians support people who test positive for stage 1 or stage 2 T1D. The guidelines have been endorsed by leading medical journals and organizations around the world.
”This monitoring guidance can help any clinician feel confident in providing adequate care in the early stages of type 1 and know when to refer to a specialist,” said Albanese-O’Neill.

I think of research, advocacy, and medical affairs as three legs of a stool—in terms of clinical adoption, each helps answer a different question. Research: Does it work? Advocacy: Will the regulators approve it and will insurance companies cover it? Medical Affairs: Do clinical teams have the competency and readiness to prescribe the treatment and educate and support people with T1D?”
It’s also helpful for the people who test positive for early stage T1D. Using the monitoring guidance, people can work with their healthcare team to monitor blood glucose levels to identify when insulin therapy may be needed; consider participating in clinical trials of disease-modifying therapies in development; and consider when and whether Tzield might be appropriate.
“There is currently one FDA-approved disease-modifying therapy for early-stage type 1 diabetes and additional therapies are being studied in clinical trials,” said Lally. “Identifying type 1 early gives the individual and their family time to learn more about type 1 and their options before reaching stage 3 T1D, which requires daily insulin therapy.”

Clinical trials: Increasing patient referrals
Clinical trials are a vital step for any treatment, drug, or device to make it into the hands of people with T1D. Currently, more than 300 clinical trials focused on T1D are actively recruiting participants.
Moreover, clinical trials can offer people the chance of receiving a cutting-edge treatment they may not otherwise be able to access.
Through its HCP resources and existing clinical trial resources, Breakthrough T1D is stressing the significance of investigational T1D therapies—while also clarifying common misconceptions about clinical trials.
“The clinical trial education portion of the program explains current trial opportunities and the critical need to increase diversity in diabetes research,” said Lally.
Despite the importance of clinical trials, many are delayed due to slow enrollment, adding cost and prolonging the results. A 2020 Tufts University study found that nearly 90 percent of clinicians surveyed felt comfortable talking about clinical trials.
“Unfortunately, the survey also revealed that annually, fewer than one percent of patients are referred to clinical trials,” said Lally.
So, why aren’t more clinicians referring their patients to clinical trials? Time and resources.

The most challenging part is helping patients understand what a clinical trial is, what it involves, and how previous scientific advances were only possible because of clinical trials. Healthcare providers often don’t have enough time in their busy clinics to discuss this with patients and families.”
Jacobsen, who is with University of Florida Health (UF Health), is one of the faculty members for Breakthrough T1D’s new HCP resources—specifically, the offering related to currently recruiting clinical trials for T1D disease-modifying therapies.
Jacobsen stresses that families and individuals with T1D also need specific education on the potential of receiving a placebo during a clinical trial—and why it’s an impactful part of any clinical trial.
Clinicians also may not know how to quickly get and stay up to date on current trial opportunities and how to get individuals who test positive for T1D autoantibodies involved.
“We provide a streamlined presentation about how to talk to families of people with T1D or people at risk for T1D about clinical trials,” explains Jacobsen, “We can direct families to one of several websites for more detailed information, like Breakthrough T1D’s Clinical Trial Finder.”

Cell therapies: Cures within reach
Cell therapies are one of the most promising approaches to curing T1D and one of the cornerstones of Breakthrough T1D’s cures research portfolio.
Advances in cell therapies have ramped up in recent years: participants in clinical trials of these therapies have been able to stop taking insulin. To speed this progress even more, Breakthrough T1D recently launched Project ACT (Accelerate Cell Therapies).
Cell Therapies
Also called islet cell therapies, these therapies replace destroyed beta cells so that people with T1D can again produce their own insulin.
The organization has long invested in cell therapy research and has a track record of success in making life-changing T1D therapies a reality—the prime examples being artificial pancreas systems (AP systems) and Tzield.
The work of Breakthrough T1D’s Research, Advocacy, and Medical Affairs teams—in partnership with the organization’s venture philanthropy fund, the T1D Fund—will be integral to Project ACT’s success.
Like AP systems, Tzield, and all other FDA-approved drugs, treatments, and medical devices on the market today, islet cell therapies will only become available after meeting all the required benchmarks—including clinical trials.
Clinicians who are in-the-know about clinical trials and how to help their patients enroll are but one of the numerous ways Breakthrough T1D’s Project ACT will make islet cell therapies a reality, faster.
“Clinicians are generally the most trusted source for this information, but most are not making those referrals, so the gap never closes,” said Lally. “We aim to change that.”
“We want every member of the diverse T1D community to be aware of clinical trials, how to participate, and where to find information,” added Albanese-O’Neill. “The next generation of breakthroughs depends on it.”
Editor’s note: This story co-written by Ginger Vieira, special contributor to Breakthrough T1D.
Type 1 diabetes (T1D) is caused by the autoimmune destruction of insulin-producing beta cells in the pancreas. People with T1D are dependent on insulin therapy to control their blood sugar levels, which is critical to prevent complications that occur when blood sugar is too high (hyperglycemia) or too low (hypoglycemia).
The best bet for T1D cures depends on cell therapies, which replace destroyed beta cells with protected, functional cells to restore insulin therapy independence and glucose control, ideally without chronic immunosuppression. Breakthrough T1D’s Cures Program has been instrumental in the incredible progress we’ve made in cell therapies research. The secret to success? Stem cells.
What are stem cells?
Stem cells are uniquely suited for use in cell therapies because of their biological characteristics. First, they are essentially a blank slate—we can biologically engineer them to become any cell we want, including beta cells. Second, stem cells can make copies of themselves while remaining a blank slate, meaning we can generate an unlimited source of transformable cells.
Since scientists first isolated human stem cells in 1998, there has been major progress in therapies that can replace or renew damaged and diseased tissue. For example, people with blood cancer often undergo chemotherapy and radiation to destroy cancerous blood cells. Stem cells are delivered directly into the bloodstream and travel to the bone marrow, where they transform into new, non-cancerous blood cells, replacing the important cells that were lost with healthy ones. The exciting progress in stem cell research extends to T1D—researchers have harnessed the power of stem cells to generate functional beta cells and islets, rapidly accelerating the drive toward T1D cures. Breakthrough T1D has been at the forefront of stem cell research in T1D for decades.
Stem cells are the foundation of our Cell Therapies Program
Breakthrough T1D’s Cell Therapies Program focuses solely on making and improving stem cell-derived beta cells. For stem cell-based therapies to become a reality for everyone with T1D, there are three primary goals that we are striving to achieve:
- First, we need to generate a renewable, scalable source of beta cells so that there are enough for everyone who needs them.
- Second, we need to find a habitable site to implant beta cells so that they remain functional and healthy for very long periods of time.
- Third, we must find a way to protect implanted beta cells from immune attack so that people won’t need to take chronic anti-rejection immunosuppressants, which can come with intolerable side effects.
Breakthrough T1D has put tremendous effort into achieving these goals; we’ve seen incredible progress in recent years, pushing us closer than ever to cures for T1D.
Breakthrough T1D has driven more than $156 million to cell therapies research in the past decade and has spearheaded several initiatives to drive this effort forward. In 2013, the Beta Cell Replacement Consortium was launched, bringing together 50+ of the brightest scientists and key industry players to integrate expertise in bioengineering, animal models, transplant medicine, and other key research areas.
Similarly, Breakthrough T1D established several Centers of Excellence, a collection of institutions driving exceptional advancements in immunology, stem cell biology, and gene editing, all of which are critically important to cell therapies research. The ultimate goal of these initiatives is to foster collaboration, exchange resources and data, and accelerate the development of stem cell-derived islet therapies.
Project ACT
To further support these efforts, Breakthrough T1D most recently launched Project Accelerate Cell Therapies (Project ACT) to simultaneously push research, development, regulatory policies, access, and adoption to increase the rate at which cell therapies without the need for broad immunosuppression will become available to people with T1D. This is important because, at this time, cell therapies require anti-rejection immunosuppressants, which can come with serious long-term side effects that may not be tolerable for everyone with T1D.
Looking forward
With Breakthrough T1D’s commitment to driving cell therapies forward, we have made significant headway in the development of life-changing therapies that can place healthy, insulin-producing beta cells back into people with T1D. “Even with today’s fantastic automated insulin delivery systems and advanced algorithms, those living with T1D still spend a significant amount of time interacting with their devices in order to maintain blood sugar control,” said Nicholas Mamrak, a scientist at Breakthrough T1D. “The prospect of cell therapies lies in the ability to take off these pumps and spend less time managing and worrying about their diabetes.”
The drive toward stem cell-based therapies becoming a reality for everyone with T1D continues—despite how far we’ve come, we still have more work to do.
This article is the first of a series exploring the different ways that Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) will shape the future of cell therapies for type 1 diabetes (T1D). Stay tuned for the next article, which will explore the present and future of cell therapies for T1D.
Today, Sana Biotechnology released significant clinical data: the first person with type 1 diabetes (T1D) who received deceased donor islets engineered to evade the immune system is producing insulin without immunosuppression.
The details
This is a big step for cell-based therapies for T1D. Sana’s first-in-human study consists of allogeneic islets, meaning they are derived from an external source, which in this case is the pancreases of deceased donors. These islets were engineered to avoid recognition by the immune system (hypoimmune) and were implanted intramuscularly into a person with T1D. After four weeks, circulating C-peptide increased, meaning that the beta cells are alive, healthy, and producing insulin—all without the need for immunosuppression and no safety issue. This is the first evidence of engineered islets successfully avoiding immune destruction.
What this means for the T1D community
While this is an incredibly promising step forward for the T1D community, currently available cell therapies that rely on deceased donor islets (Lantidra®) are only accessible to a small portion of the T1D population because there are very few donor cells available. They also require broad anti-rejection immunosuppressants, which can come with serious side effects that may not be manageable for everyone with T1D. Engineering cells to evade immune attack is a new path forward to protect the insulin-producing beta cells and avoid the use of immunosuppressants. Most importantly, this technology can now be applied to stem cell-based therapies, which is a scalable solution for many people with T1D.
What’s next: lots to look forward to
Breakthrough T1D believes that the best bet for type 1 diabetes cures lies in stem cell-based therapies since deceased donor islets are in short supply, while stem cell-derived islets can be produced at scale. We have now opened the doors to apply hypoimmune technology to stem cell-derived islets, moving us closer to the possibility of having enough immune-evading cells for everyone with T1D. While this will take significant time, effort, and money, every day we take another step toward a possible life-changing T1D cure.
Breakthrough T1D’s role
The primary objective of Breakthrough T1D’s beta cell replacement efforts is to place insulin-producing cells into people with T1D without the use of immunosuppressants. Breakthrough T1D strongly supports the development of stem cell-based therapies that do not require broad immunosuppression and recently launched an initiative to accelerate this faster than ever (Project ACT – Accelerate Cell Therapies). To contribute to the advancement of these game-changing therapies, the T1D Fund: A Breakthrough T1D Venture invested in Sana recognizing that their hypoimmune engineering technology held significant promise for type 1 diabetes cell therapies. We look forward to seeing how the trial progresses.