Author’s Note:
“I wanted to write the teplizumab story because it captures the enormous challenges that face every scientist and every volunteer committed to curing and preventing T1D. It is hard. It takes passion. It takes purpose. It takes tireless and unselfish dedication. It takes perseverance. And it takes luck. But mostly, it takes hope and belief: hope that the work can make a difference and belief that it will.”
– Doug Lowenstein, Breakthrough T1D volunteer since the diagnosis of his daughter, Emma, in April 2001
Ninety-five percent of all drugs that start human clinical trials are never approved by the U.S. Food and Drug Administration (FDA). This is a story of how one drug beat those overwhelming odds. It is a story of success and failure, multiple near-death experiences, and luck. But mostly it is also a story of relentless and ceaseless perseverance by a small cadre of scientists who never wavered in their certainty about the drug’s transformative potential to save lives.
The drug’s generic name is teplizumab. Rather than just treating the symptoms of the burdensome and life-threatening autoimmune disease, type 1 diabetes (T1D), as every therapy since the discovery of insulin 100 years earlier had done, teplizumab is the first therapy that treats the underlying causes of the disease. After teplizumab finally crossed the finish line in November 2022, more than 30 years after its creation, one participant called it “the little molecule that could.” Another long-time observer simply said, “it went through curves, bumps, hills, and brick walls. It’s amazing given all this that it finally broke through.”
This is how it happened.
Chapter 1: The Scientists
Jeff Bluestone was born in 1953 and grew up in New Jersey, the son of a dry cleaner. “I thought I was going to focus on virology, but my dad’s friend had a cancer scare, and I met one of his doctors at Memorial Sloan Kettering Cancer Center (MSK) in New York who was an immunologist,” he recalled. This chance encounter convinced Bluestone to make a career pivot from virology to immunology and move from Rutgers University to the Sloan Kettering division of Weill Cornell Graduate School of Medical Science for his Ph.D. It was the first step in a journey that would lead Bluestone to being recognized today as one of the world’s leading immunologists.
cancer scare, and I met one of his doctors at Memorial Sloan Kettering Cancer Center (MSK) in New York who was an immunologist,” he recalled. This chance encounter convinced Bluestone to make a career pivot from virology to immunology and move from Rutgers University to the Sloan Kettering division of Weill Cornell Graduate School of Medical Science for his Ph.D. It was the first step in a journey that would lead Bluestone to being recognized today as one of the world’s leading immunologists.
Bluestone started his career at the National Institutes of Health (NIH) in Bethesda, MD, where he worked on kidney transplants and, by 1987, he was at the University of Chicago’s Ben May Department for Cancer Research. It was there that he met Kevan Herold.
 Kevan Herold grew up in the suburbs of Philadelphia. “Science was always my thing. It is what I always loved. It was the easy part of school.” So, it was no surprise that in 1974 he enrolled in Penn State University’s accelerated medical degree program. But a few weeks after arriving in University Park, his life changed.
Kevan Herold grew up in the suburbs of Philadelphia. “Science was always my thing. It is what I always loved. It was the easy part of school.” So, it was no surprise that in 1974 he enrolled in Penn State University’s accelerated medical degree program. But a few weeks after arriving in University Park, his life changed.
“I went to a fraternity open house, and I started feeling really sick there. It wasn’t like I drank tons of beer or anything like that. I just really didn’t feel well. In the next couple of weeks, I would get up at night and drink and go to the bathroom and so on. And one day, I was riding the campus shuttle and thinking, ‘I just can’t do this anymore.’” He went to the Campus Health Service and, hours later, ended up in the Emergency Room.
Herold had T1D, a chronic disease where the body’s own immune cells mistakenly destroy the insulin-producing beta cells that play a critical role in sustaining life. The diagnosis came out of the blue. At the time Herold was diagnosed, T1D put patients on a path to blindness, kidney failure, nerve damage, and heart disease. Patients faced scores of daily decisions related to how much insulin to self-administer: too much or too little could have life-threatening consequences. Most of those diagnosed with T1D, including Herold, have never heard of the disease before being diagnosed. Even today, 85% of those diagnosed with T1D have no family history. “I didn’t know anything about this. Now, all of a sudden, it’s like you’re drinking out of the fire hose,” Herold recalled about those early weeks. But instead of overwhelming him, Herold said, “all of this stuff became, frankly, intellectually very interesting.” He knew then that he wanted to become a medical researcher.
Herold went on to complete his medical degree at Jefferson Medical College in Philadelphia and soon landed at the University of Chicago to work with Frank Fitch, a renowned immunologist. In the late 1980s, Fitch introduced Herold to Bluestone. Over the next 30 years, Bluestone and Herold would collaborate, commiserate, and ceaselessly pursue the goal of using a new class of drug, called monoclonal antibodies, to prevent T1D.
Chapter 2: Drinking the Kool-Aid
Monoclonal antibodies (mAbs) are highly precise tools that target specific molecules that are linked to specific diseases. In fact, many of today’s immune therapies that have slowed or cured various cancers and treated some autoimmune diseases are mAbs. When Herold and Bluestone connected in Chicago, the only FDA-approved mAb was called Orthoclone OKT3. It was used to prevent kidney transplant rejection. OKT3 was made by the Johnson & Johnson (J&J) subsidiary Ortho Pharmaceuticals. It was an anti-CD3 mAb. CD3 is a protein that is on the surface of a subset of immune cells called T cells. When a foreign organ is transplanted, CD3 T cells destroy the new organ. OKT3 was a groundbreaking drug. It blocks CD3, and the CD3 T cells disappear, preventing organ rejection.
There was just one problem: OKT3 caused serious side effects in patients and simply wasn’t a long-term solution to the problem. Bluestone saw that as a challenge. Working with a team of colleagues, he decided to try to “fix” OKT3. In 1986, the team successfully made a new anti-CD3 that worked in mice the same as OKT3 worked in humans but without the side effects. By 1992, Linda Jolliffe and Robert Zivin, scientists in the research arm of J&J, working with another company called Celltech, built on the mouse model Bluestone’s team created, and developed a modified (mutated) version of OKT3 that successfully suppressed organ rejection with minimal side effects. Its lab name was hOKT3gamma1 (Ala-Ala). Eventually, it would simply be known as teplizumab.
While this work was happening, Bluestone and Herold, as well as a French team, led by Lucienne Chatenoud and the late Jean-Francois Bach, were studying the use of anti-CD3s in autoimmune conditions. “Our lab had a big interest in T1D since the beginning of the 1980s,” Chatenoud said. “It was an interesting topic for immunologists because it had recently been discovered to be an autoimmune disease and it appeared that the T cells were the major effectors of beta cell destruction as they were in transplant rejection.”
Bluestone and Herold theorized that the same signaling as occurred in organ rejection occurred in T1D; only in T1D, the CD3 was mistakenly signaling T cells to destroy insulin-producing beta cells. They reasoned that if an anti-CD3 could block the progression of the disease before symptoms by neutralizing the attack, it could prevent T1D. In the early 1990s, they decided to test this bold and innovative theory.
In 1992, they published a study of mice with T1D that showed that modified anti-CD3 mAbs, which did not induce side effects, could prevent the disease. This exciting news was followed two years later with a report from a French research team headed by Lucienne Chatenoud and Jean-Francois Bach that extended the Bluestone-Herold findings using an even more robust mouse model showing the modified anti-CD3 could even reverse diabetes if given right at diagnosis.
The findings cemented both teams’ determination to use a human anti-CD3 mAb to treat T1D.
“It was in the nineties when we drank the Kool-Aid,” said Bluestone of anti-CD3’s potential to prevent T1D. “We were convinced because of the early human studies in kidney transplant and the mouse studies in T1D, that it could be the game changer.”
Unfortunately, as the scientists’ enthusiasm about anti-CD3s and teplizumab built, J&J’s wilted. The company decided it did not want to be in the mAb business, so it opted not to develop teplizumab and sold the modified OKT3 to a company called Centocor. But shortly thereafter, Centocor was acquired by J&J and once again, J&J killed teplizumab development.
Thus, within a few years, teplizumab had two near-death experiences, both executed at the hands of J&J.
But Bluestone and Herold were not turning back. Herold’s personal connection to T1D provided an obvious reason to persevere. For Bluestone, his passion evolved and grew as he met families and people living with T1D. “In a lot of situations with T1D, the first time you meet one of these parents with a kid with the disease you immediately appreciate that what you are doing is a lot more than research. These are real people that are suffering terribly and it’s often kids that are bearing the brunt of the challenges of managing the disease. And so, it was at that point in the early 1990s that I switched a big chunk of my research with teplizumab to T1D.”
The organization that facilitated many of Bluestone’s early interactions with the T1D community was a nonprofit patient advocacy group named the Juvenile Diabetes Research Foundation, now simply known as Breakthrough T1D.
Chapter 3: Breakthrough T1D
In 1970, a Philadelphia Mom named Lee Ducat was devastated when her nine-year-old son was diagnosed with T1D. According to a Breakthrough T1D history of its founding, Ducat said in the subsequent days she cried every night realizing her son would have to live with the disease the rest of his life. “I told myself, ‘You better do something, Ducat.’” And after a doctor told her that with enough money, scientists could cure the disease, Lee put away her tissues. “I said, ‘Well, if money is all you need, we’ll get the money to cure diabetes.’”
At the same time, Carol and Erwin Lurie were seeking out families in their community who had T1D after their ten-year-old son was diagnosed at a summer camp. Soon, Ducat and the Luries found each other and Breakthrough T1D was born.
Since its founding, Breakthrough T1D has become the largest private funder of T1D research in the world, with affiliates in five countries and chapters throughout the U.S. In the early years, Breakthrough T1D was intensively focused on funding basic research to advance a cure for the disease. (Coincidentally, Herold’s family knew Lee Ducat. When he returned to Philadelphia after his diagnosis and was searching for an opportunity to work in T1D research, it was Ducat that helped point him in the right direction.)
But by the late 1990s, Breakthrough T1D was supporting Bluestone and Herold’s anti-CD3 research, and over the next decade, it would play a critical role as a continuing research funder, an investor, an FDA regulatory resource, and a policy advocate to advance teplizumab to the doorstep of the FDA. In fact, Herold has received more than 15 research grants from Breakthrough T1D, many of them focused on anti-CD3 studies and trials.
Herold actually spent a brief stint as Breakthrough T1D’s scientific director before moving to Columbia University in 1998; by 2000, Bluestone had moved to the University of California in San Francisco (UCSF). But they remained passionate believers in teplizumab, determined to demonstrate its efficacy. “The first thing I did when I got to Columbia was to figure out how to do a trial with teplizumab. I had about $30,000 from Breakthrough T1D.” Herold went to Columbia‘s businessperson and said, “‘I am going to do this,’ and she just laughed at me. ‘That’s not money, that’s pocket change,’” she said. But Herold managed to scrape additional funding together through a grant from the NIH; and he and Bluestone launched a small human trial testing teplizumab in 12 recently diagnosed patients with T1D.
In 2002, the prestigious New England Journal of Medicine published the first evidence that teplizumab could be the drug that could delay the progression of T1D in humans. The 2002 paper was followed in 2005 by a trial Herold and Bluestone ran in 42 patients that further solidified the evidence that teplizumab could delay T1D. Importantly, this trial also established that teplizumab generated minimal side effects.
With these exciting findings, the first human evidence emerged that teplizumab could be the drug that could change the course of diabetes without the need for chronic immune suppression—the administering of a lifetime of additional, often dangerous, drugs to prevent the immune system from overreacting.
Equally exciting, Herman Waldmann at Oxford University had created yet another T1D anti-CD3 candidate called otelixizumab, and he, Chatenoud, and Bart Keymeulen reported in 2005 that this molecule showed great promise in preserving beta cell function, similar to the findings of Herold and Bluestone. The only cautionary note was the finding that moderate flu-like symptoms, reminiscent of the symptoms of Epstein-Barr viral mononucleosis, were experienced by some drug recipients.
Herold’s 2002 and 2005 studies and the otelixizumab study fueled tremendous excitement in the T1D research and patient community. More importantly, the promising results ignited interest in these mAbs among major drugmakers.
Chapter 4: The Race Is On
In 2009, a small company called Tolerx aligned with the large global company GlaxoSmithKline (GSK) to test otelixizumab in T1D patients. It was a phase III trial—the final, largest, and most costly stage of drug testing before seeking FDA approval. It would test the drug in hundreds, not dozens, of newly diagnosed patients with T1D. They named the trial DEFEND.
Another player soon jumped in. MacroGenics was formed to develop immune-based therapeutics for autoimmune diseases and cancer. Scott Koenig, one of its founders, previously worked at MedImmune, where he met Bluestone, who was on the company’s Scientific Advisory Board. By now, Bluestone had moved from Chicago to the University of California at San Francisco (UCSF), and, in 2005, he received a visit from Koenig.
“I went out to California, and he told me the history of the molecule and it fit into what we were doing in developing molecules for autoimmune diseases,” Koenig recalled. “The fact that it already had some clinical data (from the 2002 and 2005 human trials) was an attractive way to accelerate our company. So, in June 2005, we did the deal to acquire teplizumab from Jeff.”
MacroGenics promptly went to work on a phase III trial called PROTÉGÉ, which would test teplizumab in hundreds, not dozens, of newly diagnosed patients with T1D. It was a prelude to applying to FDA for approval of teplizumab as a drug that would preserve beta cell function in newly diagnosed patients, staving off the onset of full insulin dependence. Eli Lilly came aboard as a partner to help fund the trial and manufacture the drug. Breakthrough T1D supported the trial with a relatively modest grant.
Suddenly, four companies were in the T1D anti-CD3 race: MacroGenics/Lilly, GSK/Tolerx. Optimism was high. If both trials went as expected, two anti-CD3 drugs would be on their way to the FDA for approval and the first immune therapy for T1D would soon be available to patients.
But that is not how things worked out.
Chapter 5: Abandoned Again
In a matter of a few years, both trials would be deemed as failures—though for different reasons. The PROTÉGÉ trial “failed” for two reasons. First, the FDA and the European Medical Agency (EMA), the continental equivalent to the FDA, had required MacroGenics to combine two separate success metrics, called “endpoints” in clinical trial parlance. Neither of the required FDA/EMA endpoints had been used in the earlier Herold-Bluestone trials. Second, it turned out that many trial participants enrolled outside the United States may have had different forms of T1D that inadvertently biased the results. In any event, when the results were revealed, teplizumab failed to meet the mandated endpoints, and the trial and the entire MacroGenics teplizumab program shut down. Lilly immediately walked away from teplizumab. The PROTÉGÉ results became teplizumab’s third near-death experience.
And right behind this discouraging development came the fourth one. The GSK/Tolerx trial failed as well. Because Keymeulin’s smaller 2005 trial had shown some risk of activation of Epstein-Barr Syndrome which can cause infectious mononucleosis, the sponsors decided to lower the dose given to patients in its phase III trial. In 2011, it was reported the drug did not impact T1D progression because the actual dose used was simply too low to have an effect.
The results were also a blow to Breakthrough T1D. The reality was that without an inexhaustible research budget Breakthrough T1D could not afford to keep funneling grant money to a drug that was perceived as a lost cause. Breakthrough T1D’s Chief Scientific Officer at the time, Richard Insel, recalled “a meeting with MacroGenics, Jeff, and Kevan, and the question was: should Breakthrough T1D take this on completely? But there was no way that we were going to be able to pay for it in its entirety. By then, we had lost multi-millions of dollars on the MacroGenics and Tolerx trials.”
MacroGenics maintained ownership of teplizumab but moved on to other drug candidates. Koenig continued to advocate for the drug and he was in touch regularly with Bluestone and Herold. “We would talk constantly,” he recalled. “We would go to meetings together and separately. I spent a lot of time on the road looking for a home for this. We came close with a few companies, but we just couldn’t get it over the finish line.”
Bluestone pushed Koenig to give him an opportunity to sell teplizumab to another company. Koenig agreed and Bluestone went to work. But it was tough sledding. “People thought it was not going to work,” said Bluestone. “I couldn’t raise money; we couldn’t start a company.”
That should have been enough to end the story. The golden age of T1D anti-CD3s appeared to have come and gone.
Chapter 6: Glimmers of Hope
It probably would have died there except he and Herold were not buying the negative data from the PROTÉGÉ trial. Herold and Bluestone believed that the data from their own 2002 and 2005 trials, as well as from other trials with teplizumab in the mid-2000s, proved the drug’s positive effects on newly diagnosed people with T1D. “I knew we had all these positive results from the clinical trials I was doing. I was a little surprised that there was an absolute ‘zero’ on PROTÉGÉ I have to admit. I remember being really shocked and I couldn’t figure that out.”
But soon enough, they did. As they dived deeply into the PROTÉGÉ data, they saw what they believed all along. Patients receiving teplizumab produced more insulin than those receiving the placebo as measured by the breakdown product of insulin, called C-peptide. This is exactly what had excited them and Chatenoud, in all their earlier trials. But the FDA had not agreed to use C-peptide as the trial’s endpoint, and, in so doing, it had obscured the drug’s efficacy. The chronically understated Herold recalled, “When I saw the C-peptide data from the trial I thought, ‘Well, yeah, of course it worked.'”
Still, though, hope does not pay the cost of clinical work. Industry had lost interest; Breakthrough T1D lacked resources to fund a new trial by itself. And while the NIH’s National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) had continued to provide funding to Herold for T1D research evaluating teplizumab in newly diagnosed patients, there was no guarantee it would have the will or the resources to do so in the future.
The Special Diabetes Program (SDP) would help change that.
Chapter 7: Strange Bedfellows
“It was the summer of 1997 and Breakthrough T1D had just established our D.C. office a few years earlier,” recalled Bill Schmidt, Breakthrough T1D’s first head of Government Relations. “We were making a lot of noise in Congress and the Clinton Administration about the underfunding at NIH of T1D research, and diabetes in general. It was pretty concerning. And then Speaker Gingrich really developed a strong interest in diabetes.”
“Woody (Robert Wood) Johnson came in to see me about the importance of Breakthrough T1D,” Gingrich said in a recent interview. “He convinced me T1D was a pervasive and life-altering disease and we should have a more dramatic funding program than we had.” This set the stage for Erskine. (Ironically, Johnson, currently the owner of the New York Jets, was the great grandson of one of the three brothers that founded Johnson & Johnson, the very company that helped discover teplizumab and abandoned it way back in 1994.)
“Erskine” was Erskine Bowles, President Clinton’s Chief of Staff who had a child (eventually two) with T1D. While Gingrich and Bowles had little in common politically, the former Speaker recalled that “we had a very good working relationship, and Erskine brought the idea of a T1D funding program to me.”
That same year, there was a pitched battle over the federal budget. Eventually, President Clinton and Republicans, led by Gingrich, reached a form of a grand bargain that went by the prosaic name of The Balanced Budget Act of 1997. The bill retained a package of tax hikes favored by Democrats and coupled these with reduced spending on Medicare and Medicaid.
Tucked deep inside the hundreds of pages of legislative language, Gingrich and Bowles had slipped into the bill a five-line section called The Special Diabetes Program, or SDP, providing $30 million annually for five years to fund T1D research through NIH.
In 2000, Congress increased annual funding for SDP to $100 million. But just two years later, SDP was set to expire. Unless it was renewed, this increasingly important T1D funding stream would abruptly end. What ensued was a high-wire, gut-wrenching battle that on multiple occasions seemed destined to end with the death of the program. Breakthrough T1D would not go down without swinging. It waged a full-court, high-profile campaign involving families, celebrities (Breakthrough T1D Chair Mary Tyler Moore who had T1D, actor Kevin Kline who had a son with T1D), and dozens of major corporate CEOs who had T1D in their families.
The effort culminated in the wee hours of the morning on November 20 when the last holdout, Senator Don Nickles of Oklahoma, a senior member of the Senate Budget Committee, capitulated under a barrage of Breakthrough T1D-generated calls to his office from Oklahomans with T1D, and pressure from numerous colleagues supporting SDP’s renewal who surrounded him on the Senate floor. At 1 am on November 20, 2000, SDP was renewed for $150 million per year for five years.
None of those involved in SDP’s creation or renewal imagined that SDP would eventually provide some of the key funding for the clinical trial that would eventually transform the world of T1D science and treatment.
Chapter 8: The Game Changer
Jay Skyler was a well-known and accomplished T1D researcher with a strong commitment to T1D prevention, and he was a long-time ally of Bluestone and Herold in T1D prevention research. In fact, Skyler was the lead investigator in what had been the largest T1D prevention trial ever conducted—the NIH’s National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) 2000-2005 Diabetes Prevention Trial (DPT), which sought to determine if administering oral insulin to nondiabetic relatives at risk of getting T1D would delay disease onset. The results did not pan out.
By 2010, Skyler was the head of the NIDDK’s TrialNet program which had grown out of the DPT trial he led. Herold and Bluestone proposed to Skyler that TrialNet fund a teplizumab trial in those at risk of T1D—patients who had blood tests that showed they would eventually get clinical, symptomatic, insulin-dependent T1D. In other words, instead of testing the drug in newly diagnosed patients who were already symptomatic, as all the prior studies had done, why not try it in patients before they even reach that point? Skyler readily agreed.
Fittingly, Herold, by now at Yale University School of Medicine, was the lead, or primary investigator, overseeing the trial.
Things got off to a promising start when FDA agreed to allow TrialNet to use the safety data from all the earlier teplizumab trials instead of requiring new studies to prove safety, a requirement that would have taken years longer and would have set the bar too high to even try. And most importantly, FDA agreed to a much more relevant endpoint than it had mandated for PROTÉGÉ. “There was a single endpoint: do you get diabetes or don’t you get diabetes?” said Bluestone. While this was great news, it also meant the trial would take years to complete to allow time to compare whether and how long those receiving teplizumab progressed to T1D compared to those not receiving the drug (placebo arm).
But the FDA’s flexibility didn’t clear the field entirely. In fact, the trial’s advocates ran into a buzzsaw of skeptics at NIDDK and some diabetes specialists in the community. Voices said it was premature, they wondered about the drug’s safety, and they were concerned the trial would take too long and cost too much. Judy Fradkin, who served at the time as Director of NIDDK’s Division of Diabetes, Endocrinology, and Metabolic Diseases, made the call to proceed; in July 2011, the first trial to delay onset of T1D in at-risk patients got underway. It was called simply TN-10, or TrialNet 10.
MacroGenics, thanks to Koenig, also played a key role in getting TN-10 off the ground. It provided some of the remaining supply of teplizumab for use in the trial (Lilly had stopped making it but MacroGenics retained leftover inventory). It insisted that the trial be “randomized” meaning that some participants would receive teplizumab and others would not. “We would not have proceeded unless there was agreement about the [randomized] study design,” said Koenig. “If that had not occurred, there would have not been an approved drug today.”
As it turned out, this would not be the last word on trial design. The subsequent debate would indeed almost kill TN-10.
After the TN-10 launch, it was immediately clear that one of the major challenges would be enrolling subjects. This was because unlike conducting a trial of people who already had a T1D diagnosis as every other study had done, TN-10 needed to identify at-risk participants that had blood tests that had shown the disease had started and likely would eventually have clinical T1D.
In essence, TrialNet had to persuade people who were not actually sick to participate in a medical trial with some risks. That was hard enough. But because all participants had to come to a trial site for a 14-consecutive-day infusion, it placed an added set of practical burdens on recruitment. Asking kids to miss two weeks of school, their parents to miss time at work, and, in some cases, to travel long distances, was a heavy lift.
Chapter 9: The Patients
Madison Buff’s older brother was diagnosed with T1D around 2013 when he was 15. Madi and her two siblings were screened through TrialNet to see if any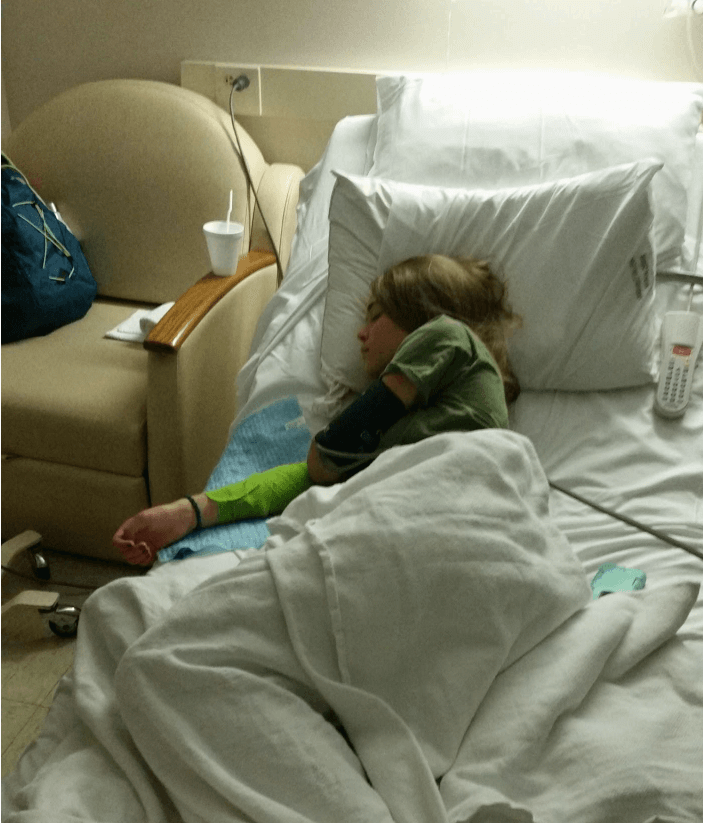 of them were on the path to developing T1D. “I was the only one that came back multiple autoantibody positive (meaning she would eventually progress to T1D),” said Madi. “And just hearing that I had the autoantibodies was scary,” recalled Madi. By her junior year in high school, her test results suggested the disease might be starting to progress.
of them were on the path to developing T1D. “I was the only one that came back multiple autoantibody positive (meaning she would eventually progress to T1D),” said Madi. “And just hearing that I had the autoantibodies was scary,” recalled Madi. By her junior year in high school, her test results suggested the disease might be starting to progress.
“And that’s when they told me about the TN-10 trial. We were hesitant about it because my mother would have to take two weeks off from work and me from school, and it was a six-hour drive to Nashville where we would get the infusion and my mom didn’t like to drive. But we decided even if it didn’t help me, it would help the research world; and, eventually, it would help us, because we knew T1D ran in our family.” So, they went off to one of the trial sites in Nashville where Madi would receive teplizumab or the placebo for the two-week infusion.
Just outside of Rochester, NY, Cory Wirt, a pediatrician, and her husband were routinely screening their daughter Claire for T1D from age 2. Both parents had T1D in their families, so they were quite familiar with the disease. In fact, the family was even involved in the local Breakthrough T1D chapter. All went well with Claire until her test at age four came back autoantibody positive. “In 2015, we got a call from our local endocrinologist who said, ‘Hey, there’s this guy at Yale, Kevan Herold, and he has an interesting trial going and I wonder if you’d like to talk to him?’” And she did. Soon thereafter, Claire enrolled in the TN-10 trial and headed to New Haven where a TN-10 trial site was running under Herold’s supervision.
T1D from age 2. Both parents had T1D in their families, so they were quite familiar with the disease. In fact, the family was even involved in the local Breakthrough T1D chapter. All went well with Claire until her test at age four came back autoantibody positive. “In 2015, we got a call from our local endocrinologist who said, ‘Hey, there’s this guy at Yale, Kevan Herold, and he has an interesting trial going and I wonder if you’d like to talk to him?’” And she did. Soon thereafter, Claire enrolled in the TN-10 trial and headed to New Haven where a TN-10 trial site was running under Herold’s supervision.
Still, TN-10 recruitment moved glacially. The goal was to enroll a total of 160 people, 80 in the placebo group and 80 in the drug arm. In time, it became clear that TrialNet would take years to hit its enrollment target. “I remember some people felt we should just pull the plug. It was just costing too much and taking too long,” said Skyler.
Fradkin and the team ultimately made the tough call to cut the trial down in half to 80 subjects evenly divided between the two arms. Chatenoud, who served as an advisor on the TrialNet Steering Committee, recalls being at a meeting when the trial size was reduced and flying home to Paris from Washington, D.C. feeling “discouraged.” She said she felt “if you decrease the number, you increase the challenge to show that the difference between the two groups (the one receiving the drug and the one receiving the placebo) is stark [statistically meaningful], and when they went down with the numbers a lot of people in the community felt it was not going to work.” It meant that the trial would have to hit the clinical trial equivalent of a grand slam home run by showing the drug’s effectiveness far beyond the levels needed in the other positive teplizumab trials.
The second threat to TN-10 came when some pushed to abandon the placebo arm entirely to accelerate the trial and reduce cost. Once again, Fradkin made the final call. “There was some talk about getting rid of the placebo arm which to me would have been a non-starter,” she said. “If we had gotten rid of the placebo arm there would have been no point in doing the trial, it would have been totally unconvincing.” Skyler added: “What Judy did, when recruitment was tough, and there was a vote to get rid of the placebo group, Judy said ‘No.’ Had she not, we would not be talking today. She stood firm and was the savior of the trial.”
“It was the hardest clinical trial I have ever been involved with,” recalled Fradkin. But teplizumab had survived two failed industry trials, the disappearance of four drug companies, and a touch and go debate over whether to do a prevention trial at NIH, and somehow, it was back on the clock more than 20 years after its creation back in the early 1990s.
By now, Madi and Claire had completed their TN-10 infusions and waited. Did they or did they not get the drug?
“I wasn’t getting any symptoms,” recalled Madi. “We heard some people were getting rashes and we were looking every day at my arms to see if we had any rashes and nothing was ever coming up. And it was just disheartening.”
Cory Wirt had heard something similar. “Every day we’d walk back and forth to the infusion center, we’d get back to the room, and I would literally sit and cry. Tears of joy, tears of sadness because you don’t know. I still get emotional thinking about it. I think on day six or seven there was a faint rash that was fleeting and you just want to hang onto that. Maybe that was it. But you don’t know, you’re grasping.”
Chapter 10: The New Kids on the Block
Ashleigh Palmer and Francisco Leon met through LinkedIn. Palmer had established a reputation as a drug company turnaround and startup specialist; Leon had built an extensive career as an immunologist. “We decided to start a disease prevention company,” Palmer recalled, and by 2017, they formed a company with three assets: two gastrointestinal autoimmunity drugs (licensed from J&J subsidiary Janssen) and a vaccine against the Coxsackie B virus. The Coxsackie B virus is believed to be a common trigger of T1D. They named their new company Provention Bio, “pro” for being proactive at disease interception, and “vention” for disease prevention.
Soon after Provention was formed with the goal of preventing T1D and other autoimmune diseases, it benefited from two major strokes of luck. Palmer was contacted by a financial firm called MDB Capital because it had heard of his turnaround expertise and wanted him to join a Board of a company in which it had invested. Palmer declined but mentioned his new company and MDB expressed interest in hearing more. In January 2017, Palmer and Leon met with MDB during the annual JP Morgan Conference, a major gathering of biotech companies, scientists, investors, and others looking for the “next” major therapy. “I will never forget the meeting,” said Leon. “We pitched them and they said they could get our company funded around the vaccine asset, which surprised me because it was our riskiest asset with the longest lead time (to profitability).”
The MDB meeting was lucky enough, but the second stroke of luck came at 7 am the following morning when the pair were at what Leon called a “speed dating” session where attendees could stop by and quickly check out companies to see if anything was of interest. “One guy was at the table, and he said he ran a pipette company,” remember Leon.
The pipette man was Mark Fischer-Colbrie who had a son with T1D; and he just happened to be a volunteer leader at Breakthrough T1D. “I was standing at a table with Francisco and Ashleigh, and I always wear my Breakthrough T1D pin and always ask people if they are working on anything related to autoimmunity. And they told me they had just started a company and had a vaccine against Coxsackie B virus and I knew that it is strongly implicated in T1D. So, I immediately connected them to the Breakthrough T1D T1D venture philanthropy fund.”
The Breakthrough T1D T1D Fund was itself brand-new. It was the brainchild of John Brady, a former Chair of the Breakthrough T1D International Board. Brady was desperately looking to expand Breakthrough T1D’s core funding from galas and walks to something that could supercharge Breakthrough T1D revenue so it could make bigger bets on more ambitious, potential game-changing therapies. Brady recruited a bright, dynamic executive named Sean Doherty, at the time the General Counsel for the giant private equity firm Bain Capital, who had a son with T1D, to design and launch the innovative venture philanthropy initiative.
Five months after the chance meeting with Fischer-Colbrie, and just a few months after the T1D Fund was launched, it made an investment in Provention Bio. J&J Innovation soon followed, and MDB Capital completed the Series A round. In June 2017, Provention’s first capital infusion closed with $28.4 million. As part of the deal, Provention committed to becoming a public company within 18 months.
The Breakthrough T1D T1D Fund imprimatur was a game changer. Leon said that without it, “the Provention we know would not have existed. Their commitment was important to make us ‘real.’”
Amazingly, at the time the financing closed, teplizumab was not on the radar of any principals. The Breakthrough T1D investment was all predicated on the Coxsackie B vaccine. That would soon change, and it changed because of yet another appearance by Lady Luck.
Chapter 11: TN-10 Earthquake
Eleanor (Leni) Ramos started her career in academia as an immunologist, but eventually decided she wanted to get involved in hands-on drug development. She became a kidney transplant physician and joined Bristol Myers Squibb (BMS). While at BMS she met Leon around 2000. She soon moved on to join Bluestone at the Immune Tolerance Network (ITN), which was supporting an early teplizumab trial led by, surprise, Kevan Herold. Over the years, she and Leon would occasionally catch up.
In 2017, after a career break, Ramos was about to take a new job when Leon called and asked for her input on the assets Provention was putting together. “When she told me she was about to take a new job, I suggested she join us at Provention as Chief Medical Officer,” Leon said. Ramos was intrigued. “Ashleigh and Francisco were coming out to LA the next week, and I agreed to meet in a relaxed setting at Los Angeles International Airport (LAX),” she remembered. “But my plane was delayed and delayed.” It looked like the vagaries of air travel would prevent Ramos from making the meeting. But Palmer and Leon convinced her to fly to LAX for even a short meeting. The “relaxed” meeting finally took place over an hour with the trio sitting on the floor of a crowded baggage claim at United’s Terminal 7. Ramos signed on as CMO. The trio didn’t know it at the time, but it would turn out to be a very serendipitous hire.
Provention Bio’s now also included the founders, Ramos, and Andrew Drechsler. Drechsler had joined the company as Chief Financial Officer a few months earlier. An experienced drug industry financial executive, he also has three children with T1D. The team realized that to complete a successful public stock offering (Initial Public Offering, or IPO) it would be prudent to find other drug assets. “One of the tenets of Provention Bio, and a lesson I learned from turnaround work, is that a company with limited assets would be dead” if those few assets didn’t succeed, said Palmer. So, the small team started reaching out to their networks to see if it could find other promising autoimmune therapeutic candidates to acquire.
One of Ramos’ first calls was to an old colleague she had worked with 15 years earlier: Jeff Bluestone. “I said, ‘What’s going on?’ and he asked if I remembered the T1D drug we worked on at ITN. Honestly, from 2002 when I left ITN, to when I called him in 2017, I had not followed the diabetes space. Jeff said that MacroGenics had put it on the shelf because the pivotal phase III PROTÉGÉ trial failed but we should look at it.”
Ramos reported back to the team that teplizumab was available and soon the principals met with Bluestone. Bluestone gave them a mixed view of their other assets and then, in his words, “I blurted out that I had a better drug than all the ones in their pipeline.”
The Provention team vigorously debated the pros and cons of teplizumab but eventually agreed to try to acquire it from MacroGenics. Finalizing the deal was complicated because so many other companies, including Lilly and Celltech had acquired limited royalty rights to teplizumab over its history. Eventually the deal closed. And Scott Koenig, still CEO at MacroGenics and still a believer even though his company was no longer developing teplizumab, played a key role. “When we were at the 11th hour, Scott stepped in and made some compromises to make sure the deal got over the finish line even though he knew there might never be a profit made from the drug,” Bluestone said.
Armed now with teplizumab and the other assets, Provention Bio, or PRVB as its ticker symbol said, went public in July 2018 at $4 per share for a total raise of $64 million. Once they had teplizumab, the company moved to set up the PROTECT trial, another trial of newly diagnosed T1D patients to see if they would experience an extended period of insulin production before becoming symptomatic. “We planned to repeat the failed PROTÉGÉ study with lessons learned,” Palmer said. But they wanted to move fast for one major reason.
“We were concerned about the TN-10 study,” Drechsler remembered. “It was a wild card. And what if we start enrolling 300 newly diagnosed patients and the TN-10 study reads out negatively? How do you deal with that? How do you position it to Wall Street? More importantly, how do you position it with the clinical investigators we needed to run the trial?”
“We were prepared for TN-10 to fail,” said Leon. “And the reason was it was an underpowered study,” exactly the concern Chatenoud expressed when the enrollment was cut in half. “There might not be enough patients to show efficacy,” said Leon. “And then, of course, everything changed.”
What changed is that TN-10 turned out to be a stunning success despite its relatively small size.
In June 2019, Herold stood up at the annual ADA Scientific Sessions in San Francisco and announced that TN-10 had produced astonishing data. Those that received teplizumab in the trial took a median of two years longer to progress to full symptomatic T1D (follow up work has since shown that the median is now closer to three years and that some trial participants have gone a decade and remain asymptomatic). In the New England Journal of Medicine paper published months later, the conclusion simply read: “Teplizumab delayed progression to clinical T1D in high-risk participants.” Put with a bit more dramatic flair, T1D had its first ever potential disease-modifying immune therapy, the first major advance in T1D therapy since insulin a century earlier.
Bluestone was in the audience at ADA when Herold revealed the TN-10 results. “It was the first time I have ever been at an academic talk where the audience gave the speaker a standing ovation.”
Only days before Herold unveiled the results, Buff and Wirt learned that they had indeed received teplizumab, not the placebo.
Chapter 12: The Race to the FDA
Without the meeting between Palmer, Leon, and Ramos on the floor of LAX baggage claim, teplizumab likely would never have found a home at Provention Bio. But it didn’t take long after TN-10 for the company to realize it needed to switch gears immediately. “We didn’t expect this, but it was now about taking advantage of an opportunity and we began switching our priority from the PROTECT study to the prospect of being able to file for an application for approval of teplizumab to treat at-risk patients to delay the onset of full insulin dependence,” said Palmer. “What did we have to lose? It was all upside.” In January 2021, Provention filed a Biologics License Application (BLA) seeking FDA approval of teplizumab. The FDA set a decision on whether to approve it six months later.
By the time Provention filed, it had already been working to prepare for approval of the drug. One area of focus was manufacturing. Since Lilly had long ago stopped making teplizumab, Provention had to replicate the mAb in a modern manufacturing facility to distribute after approval. It chose to conduct a study to prove to the FDA that the new version was comparable to the version of the drug made by Lilly and that was used in the TN-10 trial. When it completed the study, Provention felt confident in the results and shared them with FDA. Unfortunately, FDA wasn’t convinced.
Nonetheless, FDA continued with the teplizumab review. One of the most consequential steps in any drug approval process is an Advisory Committee (AdComm), an expert panel that hears from the drug applicant, the FDA, and the public before voting whether to recommend that the FDA approve or disapprove the drug for commercial use. A positive vote can speed a drug to approval, and a negative one can be fatal.
The teplizumab AdComm took place in late May 2021. The AdComm lasted the entire day. Ramos and Herold led the Provention presentation, making the case that the drug was safe and effective. The FDA staff then made a strong case for approval, also expressing their view that the drug was safe and effective. But three of the four adult endocrinologists on the panel, those with the most experience dealing with diabetes patients, sharply pressed both the agency and Provention. They raised concerns about the potential long-term safety of the drug. They wondered whether the data was sufficiently robust, they wondered if the trial was too small to draw conclusions, and whether a larger trial should be required with more patients.
Palmer said the AdComm was “frustrating and disappointing” and “for the very first time I had an ‘Oh shit’ moment whereby this could be the end of the run of luck, this could be the squashing of the momentum.” Jason Hoitt, Provention’s Chief Commercial Officer, simply thought, “Oh my God, these people may not actually support the first disease-modifying treatment to address the underlying cause of the disease.”
But the afternoon presentations from those with the most at stake, those touched by T1D, began to regain the positive momentum. Fifteen of sixteen public witnesses offered their own powerful stories: people with T1D, doctors with T1D, academic researchers, patient advocates, all stressing how critical it would be to them and others to have two years without T1D even if they knew full insulin dependence with all its risks and burden was eventually inevitable.
Claire Wirt and Madi Buff, two of the patients who had enrolled in the trial, uncertain about their chances of getting the drug, uncertain if it would work, but willing to go to tremendous inconvenience and risk to be part of scientific experiment, were among these witnesses.
“During the past five years, I have been able to attend prom, graduate high school, I’ve moved out on my own, and I am set to graduate college in three-and-one-half years,” Madi said. “I am forever thankful to the team that helped make this such a positive experience for me and have given me the opportunity to lead a normal, young adult life without constant worrying about my blood-sugar levels and subsequent hospital visits.”
“At 9 years old, I had four positive autoantibodies,” Wirt said. “My parents and I were bracing for the inevitable. Then we got a phone call that changed everything for me. Now, I’m coming up on 66 months without T1D, five and a half years of my childhood free of this disease and from the burden that comes with it. I recently celebrated my fifteenth birthday without having to count carbs or administer insulin. I don’t have to track my blood sugar throughout the day or find the right balance for it when I go to dance or ski with my friends. My thoughts are not occupied with the stresses of going hypoglycemic [low blood sugar] or inadvertently giving myself a lethal dose of insulin. My life doesn’t have to exist in a state of careful calculation and continuous monitoring because teplizumab has given me the freedom and autonomy for five and a half years.”
After nearly eight hours, it was time for the AdComm to vote. The drama was intense. Those watching around the world on their phones or computers endured several agonizing minutes of silence. Then the mic crackled, and FDA’s Latoya Bonner announced, “For the record, the vote is 10 yeses, 7 noes.”
A win. But it was closer than ideal. Given the closeness of the vote, and the opposition of the adult diabetes experts on the panel, would that swing the FDA brass to a more skeptical view and lead to an overruling of the AdComm?
Chapter 13: The Stretch Run
Provention had no time to relax. The close AdComm vote was concerning. But an even bigger concern was that the comparability bugaboo could cause FDA to decline approval. That is exactly what happened. On July 6, 2021, the FDA told the company it had not proven that its version of the drug would work comparably to the Lilly version, and Provention’s application for approval of teplizumab was rejected.
The management team was not ready to give in. It believed it could eventually satisfy FDA on comparability, but it also knew the mountain ahead was steep and there was no guarantee of turning FDA around. Palmer called it “a gnarly and worrisome challenge.” Ramos worried that the only solution might be redoing the entire TN-10 trial which on its face seemed daunting and time consuming; it would take at least five years to repeat the TN-10 study.
The lifeboat came from an unlikely source: the PROTECT trial, once the centerpiece of the acquisition of teplizumab. Luckily, PROTECT started when there was still some supply of the Lilly drug so some of those enrolled were receiving it, and others were receiving the new drug. If Provention could use PROTECT data to prove the two drugs worked similarly it could save the day. There really was no other option: it was PROTECT or nothing.
Looking back at this fraught period and the perseverance and determination of the Provention team, the T1D Fund president Doherty observed, “You said to yourself, ‘Gosh, do you think this team can turn it around.’ And I will say to Ashleigh’s and Francisco’s great credit, they never gave up on it. It would have been an understandable decision to say ‘This is probably just not going to work.’ But that was never in the cards for them. They believed the two drugs were comparable and they believed in their results. They could have said they see the handwriting on the wall, but I remember Leni saying, ‘We can address this, let’s prove we are right.’”
In January 2022, seven months after the initial rejection letter, Provention shared its data with the FDA. After the meeting, it felt optimistic enough to refile its application for approval of teplizumab. The clock started ticking once again. The new decision date was set for August 2022. Frustratingly, in July, the FDA yet again extended the decision date for 90 days. Now, all anyone could do was wait.
Chapter 14: The Finish Line
On November 17, 2022, the FDA announced that it had approved what would hence forward be known as Tzield. “Today’s approval of a first-in-class therapy adds an important new treatment option for certain at-risk patients,” said John Sharretts, director of the Division of Diabetes, Lipid Disorders, and Obesity in the FDA’s Center for Drug Evaluation and Research. “The drug’s potential to delay clinical diagnosis of type 1 diabetes may provide patients with months to years without the burdens of disease.”
About 31 years after OKT3 was modified to become teplizumab, and 28 years on a roller coaster ride, teplizumab was on its way to patients.
Kevan Herold was in Chicago when the news came in: “It was the last day of a TrialNet meeting and I was to travel to give a talk the next day. Of course, I was quite nervous because even the last day I had gotten some questions from Provention Bio about the past trial experiences. I boarded my plane and still hadn’t seen anything. It was when I landed that I found out that the approval happened. I got a text from my wife.”
Jeff Bluestone was in his office. “My reaction was relief after so many challenges to getting across the finish line—questions about the manufactured product caused major delay, then there was a delay on July 1st due to concerns about comparability. So, when I finally heard in November, it was a relief. I emailed Kevan to tell him as well as Lucienne and Jean-Francois Bach. I heard from a few people—Margery Perry (a long-time Breakthrough T1D volunteer involved in research grants) stands out as she was so supportive over the years and continuously made the case at Breakthrough T1D. “I guess, I have lived by the mantra—do kick-ass science, collaborate like hell, and make a difference. That day, I finally felt like I had made a difference.”
Aaron Kowalski, Breakthrough T1D’s CEO, and someone who lives with T1D (as does his brother), was at home. “It’s one of those moments where you’re like, wow, here we are… a hundred years later [from the discovery of insulin] and we finally have something that’s not a treatment. For the first time we can actually do something about the disease. To me, that approval ushered in the optimism that I think our entire community is starting to feel that we are going to get this job done. We are going to walk away from diabetes, which is an amazing feeling.”
“My life would be completely different if I hadn’t gone into the trial,” Madi said recently. “Even if I develop [T1D] tomorrow, just being able to get through transitional years has helped me.” Today, Buff is working toward her MBA with a concentration in health management at the University of South Carolina. Both Madi’s and Claire’s words are the ultimate tribute to those who never doubted that teplizumab would eventually change lives and bring T1D closer to being prevented and cured, a dream that powered Lee Ducat and the Luries all the way back in 1970 when they started Breakthrough T1D, as well as Herold, Bluestone, Chatenoud, and dozens of other scientists all over the world who literally never gave up in the face of constant setbacks and skepticism right up to the end.
In mid-2023, the global French drug company Sanofi purchased Provention Bio for $2.9 billion.
Today, Buff and Wirt are both about eight years out from receiving their single course of teplizumab and neither one has symptomatic T1D.
Doug Lowenstein’s daughter was diagnosed with T1D in 2001 at age 14. Doug worked as a Breakthrough T1D volunteer for 23 years in the search for therapies to cure, prevent, and treat T1D. This work included consulting with Provention Bio, and continues with its acquirer, Sanofi. The work is entirely focused on expanding screening for T1D and does not involve marketing or selling teplizumab.