During a recent Breakthrough T1D Facebook Live event, Tandem Diabetes Care Senior Director of Behavioral Sciences Stephanie Habif and Tandem Product Manager of Clinical Solutions Christina Pamelar discussed outcomes from real-world Tandem insulin pump users, the t:connect® Mobile App (launched July 7), the remote insulin pump training process and other practical insights about Tandem Diabetes Care product use.
Breakthrough T1D Vice President of Corporate Development Joe Watterson delivered introductory remarks.
Habif holds Doctor of Education (EdD) and Master of Science (MS) degrees; Pamelar holds a Master of Science degree in Nursing (MSN) and is a Registered Nurse (RN), Certified Pediatric Nurse Practitioner (CPNP) and Certified Diabetes Care and Education Specialist (CDCES).
During the event, Habif and Pamelar covered numerous topics, including:
- Features of the t:slim X2.
- The differences between the two predictive insulin delivery technologies that are available when the t:slim X2 is used with the Dexcom G6 continuous glucose monitor (CGM): Control-IQ Technology and Basal-IQ Technology.
- Real world examples of use of the t:slim X2 with each of the predictive technologies.
- The advantages of the recently-launched t:connect® Mobile App.
The event wrapped up with a Q&A session during which Habif and Pamelar answered questions submitted in the comments.
Watch the full, recorded livestream:
A New Breakthrough T1D-Tandem Partnership
Find out about how Breakthrough T1D and Tandem Diabetes Care are teaming up to help drive awareness and education on topics related to technology innovation, diabetes management and healthcare for people with diabetes.
Smart insulin; what is that? It’s the type of insulin that Chandra Bhattacharya, Ph.D., has spent her Breakthrough T1D-funded postdoctoral fellowship—the time right after you receive your doctoral degree, but are not ready for primetime as an assistant professor, yet—to pursue. And she has a lot of shoes to fill. At the Massachusetts Institute of Technology (MIT), she’s a postdoc in the laboratory of Bob Langer, Sc.D.—a scientist known for educating a generation of researchers and a prolific inventor and patent holder—and Dan Anderson, Ph.D.—himself once a postdoc of Dr. Langer and, today, a professor.
So, let’s get back to the topic. Smart insulin is a form of insulin that turns on when it’s needed to lower blood sugars, and turns off when blood sugars are at safe levels. It’s like a pancreas (well, a healthy one), but in drug-form. Some with type 1 diabetes (T1D) think that this will be a functional cure. Take it once a day, and you’re all set.
But making smart insulin is really hard. There are two proteins (for the most part) that regulate blood sugar: insulin, which makes blood sugar go down, and glucose, which makes blood sugar go up. A very small change in glucose levels, however, is very difficult to measure, so we need a very sensitive and very selective sensor to gauge it. And that’s where Chandra fits in. Find out from her video why making a good smart insulin is both difficult and, possibly in the end, rewarding.
Breakthrough T1D’s early-career funding enables promising scientists, like Chandra, to focus their talent, ambition and passion on improving life with T1D. Learn more about the researchers we fund, and learn how you can support their work to prevent, treat and—one day—find cures for T1D.
Thanks to a new, national partnership, Breakthrough T1D and Tandem Diabetes Care are teaming up to help drive awareness and education on topics related to technology innovation, diabetes management and healthcare for people with diabetes.
“Together, we will show how innovations in type 1 diabetes (T1D) management and care will continue to minimize the intrusions of T1D on day-to-day life and provide better health outcomes for people living with the disease,” said Joe Watterson, Breakthrough T1D Vice President of Corporate Development.
The partnership will utilize Breakthrough T1D’s community facing platforms—including local chapter events, digital channels and education programs—to highlight the benefits of devices such as insulin pumps, continuous glucose monitors, and novel treatment approaches like hybrid closed loop therapy.
People living with T1D have a variety of needs that span from reducing the daily burden of disease management to finding a trusted technology partner who can deliver solutions that work reliably, provide clinical benefits and improve their overall quality of life.
The Breakthrough T1D and Tandem partnership aims to actively support the growing needs of the T1D community and help address the gap in diabetes education on these topics through regional and local activities, including select Community Summits and Breakthrough T1D Walk events.
“Education is the foundation for improved quality of life and clinical outcomes for people with type 1 diabetes, and we are proud to partner with Breakthrough T1D in furthering our shared mission to support the diabetes community,” said Brian Hansen, Executive Vice President and Chief Commercial Officer at Tandem Diabetes Care.
Recently Expand Choice for Prospective Tandem Users
Thanks to a concerted effort that included mobilizing Breakthrough T1D’s grassroots network of T1D champions across the nation, UnitedHealthcare (UHC) recently announced it would cover the t:slim X2 insulin pump from Tandem Diabetes in network, starting July 1, 2020.
This came on the heels of FDA clearance of an expanded pediatric indication for the Tandem t:slim X2™ insulin pump with Control-IQ™ technology in children ages six and older. Initially, the device was only authorized for use in people 14 and older.
Thanks to a concerted effort that included mobilizing Breakthrough T1D’s grassroots network of T1D champions across the nation, UnitedHealthcare (UHC), the nation’s largest insurer, has increased choice and will begin to cover the t:slim X2 insulin pump from Tandem Diabetes in network on July 1st.
This gives people living with type 1 diabetes (T1D) more choice in deciding which technology they use to manage their T1D.
“UHC’s decision to cover Tandem Diabetes’ insulin pumps is a direct result of the T1D community’s advocacy,” said Aaron J. Kowalski, Ph.D., Breakthrough T1D President and CEO. “Our Coverage2Control campaign brought together thousands of T1D champions demanding UHC give people with T1D the freedom to choose the insulin pump that’s right for them.”
“Today’s decision reflects the power of the collective voice of our community,” Kowalski emphasized. “We thank UnitedHealthcare for ensuring that its members have access to the technology they need to stay as healthy as possible until we have cures for T1D.”
Another T1D Triumph
Through its Coverage2Control campaign, Breakthrough T1D rallies members of the T1D community to encourage health insurers to provide affordable options, give people more choices, and offer coverage for all therapies to help ensure that people with T1D have what they need to survive and lead healthy lives.
As a result, the 25 largest insurance companies in the United States covered the first artificial pancreas system, and many put into place options to lower out-of-pocket costs for insulin.
Unfortunately, UHC had a pre-existing policy that limited pump choice for adults. On February 1, 2019, UHC announced the policy would also apply to children.
Breakthrough T1D saw this as an unacceptable step backward and mobilized the community to respond.
Using Breakthrough T1D’s campaign tools, more than 27,000 supporters sent upwards of 145,000 messages to UHC leadership and shared their concerns on social media, urging them to change their decision.
In October 2019, Kowalski and other members of the Breakthrough T1D senior leadership team met with UHC’s Chief Medical Officer, Dr. Richard Migliori, to make the case directly.
Importance of Insulin Pump Choice
Finding the best insulin pump is a personal decision that affects how people manage their T1D. Pumps have different features and capabilities and different insulin pumps work best for different people.
A person living with T1D may choose to go without a pump if they are unable to find or afford one that works for them. This can lead to diabetes being managed less successfully and increase the cost of care as health outcomes are negatively affected. Providing people with diabetes the full range of pump options increases the likelihood that they will use insulin pumps, will better manage their T1D and achieve improved health outcomes.
While Breakthrough T1D works with its partners and researchers to unlock the science for cures, we are fighting for policies that increase access to life-saving T1D treatments—such as insulin, insulin pumps, artificial pancreas systems, and continuous glucose monitors—and are sharing the most up-to-date information on programs that help with costs.
“My son and I are thrilled to learn that UHC is opening the door to covering new technologies so those living with T1D can choose the management tools that are right for them,” said Jill Folsom. “As a young adult, my son was essentially forced off of his preferred pump, and—in using a pump that didn’t fit his needs as well—he’s seen poor outcomes as a result. I am so grateful for Breakthrough T1D’s continued advocacy efforts to ensure people with T1D are empowered to make these important, personal decisions, and that health plans follow suit by covering a wide range of tools to manage the disease.”
During a recent Breakthrough T1D Facebook Live event, elite type 1 diabetes Athlete/Ultramarathoner and Dexcom Warrior Eric Tozer and Dexcom Certified Diabetes Educator (CDE) Cher Pastore, MS, RD, discussed Dexcom CLARITY technology.
Leaders from Breakthrough T1D and Dexcom delivered brief remarks before Tozer’s and Pastore’s discussion:
- Joe Watterson, Breakthrough T1D Vice President of Corporate Development
- Molly Donahue, Dexcom Associate Marketing Manager
- James McIntosh, Dexcom Senior Manager of Public Relations
During their discussion, Pastore and Tozer covered a variety of topics, including:
- How CLARITY can help people living with T1D focus conversations with their healthcare providers.
- Why telemedicine visits may be more convenient for people with T1D and how CLARITY can enhance that convenience.
- How CLARITY can help people living with T1D establish goals in between visits with their healthcare providers (either via telemedicine or in-office visits).
- Tozer’s experiences with CLARITY and telemedicine visits.
Watch the full, recorded livestream:
Most people with type 1 diabetes (T1D) and their caregivers are well aware of the dangers of hypoglycemia (low blood sugar)—especially should it become severe, which can result in seizures or unconsciousness.
Many families don’t talk about the reality of a severe hypoglycemic episode, much less plan for it as the topic can lead to anxiety for all involved
This increased anxiety is so common that there’s even a term for it: Fear of Hypoglycemia (FoH).
According to research published in the June 2019 supplement issue of the journal of the American Diabetes Association, FoH is associated with substandard diabetes management and reduced health outcomes.
Breakthrough T1D and our partners at Beyond Type 1 want T1D families to know that it doesn’t have to be this way.
With proper planning, preparation and provisions to quickly treat severe hypoglycemia, you can overcome FoH and handle emergencies with confidence.
Why now?
The coronavirus pandemic has upended the lives of most Americans. For many families, remote learning and working has created a new, stress-filled normal in which caregivers are always on and stretched thin. New routines present additional challenges. In the process, steps to prevent hypoglycemia may accidentally be skipped, increasing the chances for severe hypoglycemia.
But quarantine life also offers the opportunity to talk about and plan for important issues like severe hypoglycemia.
How should you prepare?
For starters, each member of a T1D family should be aware of the different causes and symptoms of hypoglycemia, as well as what mild/moderate hypoglycemia looks like vs. severe hypoglycemia.
A type 1 diabetes family should always have glucagon on hand to be prepared to handle a severe hypoglycemia emergency—which can be associated with seizures and unconsciousness. Each family member should know where the glucagon it is kept, how to administer it and the steps to take after administering it:
- Turning the person on their side. This is important because glucagon may cause nausea and vomiting and turning the person on their side will prevent them from choking if they do vomit.
- Calling 911 and staying by the person’s side until emergency responders arrive are taking care of the person.
Glucagon comes in different forms:
- Baqsimi—a nasal spray (Lilly)
- GlucaGen (glucagon) for Injection (Novo Nordisk)
- GvokePFS (glucagon injection) Pre-Filled Syringe (Xeris)
Families should check their glucagon at the same time each year to make sure it has not expired. This includes glucagon that may be kept in the car or is part of a travel emergency kit.
Don’t forget communication
Because severe hypoglycemia can limit someone’s ability to communicate clearly—even if they  remain conscious—T1D families may want to establish some kind of signal that their loved one with T1D can make to let others know they need help right away.
remain conscious—T1D families may want to establish some kind of signal that their loved one with T1D can make to let others know they need help right away.
This could be as simple as a specific word, blinking or stomping one’s feet two or three times. Whatever the signal is, it needs to be something the person with T1D feels they could likely do even if they are feeling very low, and something everyone in the family will remember.
You should work together to test out the signal when your loved one with type 1 diabetes has a stable blood sugar level and is feeling normal. That way, you will see if the signal makes sense for everyone involved. Don’t be afraid to have “practice drills” to make sure everyone knows how to respond.
You may also want to map out responses in different scenarios. For instance, a plan for what to do at home as well as a plan for what to do when you are away from home (does everyone know where the glucagon is kept in the car, on vacation, at a relative’s house, etc.?).
Don’t hesitate to ask for help
People can be reluctant to ask for help—even when they most need it. Sometimes, they may not even be aware that they need help.
Remind your loved one with type 1 diabetes that they should never hesitate to ask for help and emphasize to other family members that they should be ready to help at a moment’s notice.
Fear of Hypoglycemia is a real thing and quite common. But it can be overcome.
For additional advice about severe hypoglycemia emergency plans, T1D families should consult with their healthcare providers, and ask about which glucagon option is best for them.
Editor’s Note: This educational content is made possible with support from Lilly Diabetes and BD. Breakthrough T1D produces this content to provide information to our supporters about their options for managing their T1D and not as an endorsement of products. Editorial control rests solely with Breakthrough T1D.
On June 15, 2020, Abbott announced that the FDA has authorized the FreeStyle Libre 2 System. The Libre 2 gained its CE Mark clearance in Europe late 2018 and is now authorized for adults and children 4 and up in the United States. The FreeStyle Libre 2 is a 14 day continuous glucose monitor (CGM) system that transmits data every minute and now includes customizable high and low alerts without the need to scan the device. The Abbott announcement says it will be “a third of the cost of other CGMs.”
Also notable with this clearance is the FDA’s decision to permit the marketing of the FreeStyle Libre 2 as an integrated CGM (iCGM). This allows the CGM to be part of an interoperable system and work with other medical devices like insulin pumps, blood glucose monitors and other devices. This makes the FreeStyle Libre 2 only the second CGM on the market to receive this designation.
This also marks the first time a FreeStyle Libre CGM has been approved for pediatrics in the United States, widening the scope of potential users.
According to Abbott, the Libre 2 adds easy-to-use, customizable alarms for high and low glucose levels using Bluetooth. Users can also receive notifications should the sensor and reader fail to link up to ensure that a dropped connection does not go unnoticed. Alerts come in the form of sounds or vibrations, according to preference. Libre 2 users can still scan their sensor as often as desired to see current blood glucose levels, trends and patterns and a graph of the last eight hours.
“Another iCGM provides people with diabetes more choice to improve diabetes outcomes,” says Campbell Hutton, Vice President, Regulatory and Health Policy, Advocacy at Breakthrough T1D, “and also will facilitate diabetes device interoperability.”
Content from this article was sourced from Beyond Type 1.
Scientists have uncovered a potential breakthrough in developing a faster acting insulin in an unusual place: snails. Now, Breakthrough T1D-funded researchers are using the molecular features of the snail insulin to create ultra-rapid insulins for people with type 1 diabetes (T1D) and have published their findings in .
As discussed in last year’s blog, certain sea snails shoot insulin at their prey, sending them into hypoglycemic shock and allowing the snails to eat them. This immediately piqued the interest of T1D scientists, and catalyzed research into better understanding how this insulin works, with the potential to transform the treatment of type 1 diabetes.
The human and rapid-acting analog insulins available today leave much to be desired. They are slow to lower blood sugar and remain in the bloodstream for hours after administration. Faster acting, or ultra-rapid insulins, would help people with T1D better manage their disease. They could correct high blood sugars more quickly, allow for more flexibility at mealtimes, prevent hypoglycemia caused by insulin lingering too long in the body, and crucially, help to “close the loop” in artificial pancreas technology, i.e. create an artificial pancreas system where the user doesn’t have to manually enter any insulin doses (for example, at mealtime).
This paper, whose authors include lead investigator Xiaochun Xiong, Ph.D., a Breakthrough T1D postdoctoral fellow; Breakthrough T1D-funded senior author Danny Chou, Ph.D.; and previously Breakthrough T1D-funded collaborator Helena Safavi-Hemami, Ph.D., details their approach. These scientists identify the molecular characteristics of the snail insulin that make it work quickly and potently and incorporate them into human insulin. This creates an insulin that is part human and part snail that will serve as a basis for new ultra-rapid insulins.
In order for people with T1D to achieve better outcomes, insulin needs to work faster. Breakthrough T1D is excited by the findings in this paper, and looks forward to continuing to support this science to achieve novel, innovative insulins.
Read the paper here and learn more about Breakthrough T1D’s research into glucose control here.
During a recent Breakthrough T1D Facebook Live event, a panel from Medtronic Diabetes discussed Medtronic’s efforts to support the T1D community during the coronavirus disease (COVID-19) pandemic.
Members of the panel included Executive Vice President and President of Medtronic Diabetes Sean Salmon, as well as other leaders from Medtronic. Breakthrough T1D Vice President of Corporate Development Joe Watterson and Senior Director of Marketing for Medtronic Diabetes Charles Cush delivered introductory remarks.
The panelists addressed a variety of topics, including:
-
Medtronic’s COVID-19 response, including how they have been listening to and working with the T1D community and health care professionals (presented by Salmon)
-
Medtronic Assurance Program (presented by Vice President of Health Economics, Reimbursement & Government Affairs, Medtronic Diabetes, Jeff Farkas)
-
Optimizing Remote Telemedicine Appointments (presented by Chief Medical Officer, Medtronic Diabetes, Robert Vigersky, M.D.)
-
Supporting People Living with T1D in a Virtual World (presented by National Clinical Director, Medtronic Diabetes, Kim Larson)
Watch the full, recorded livestream:
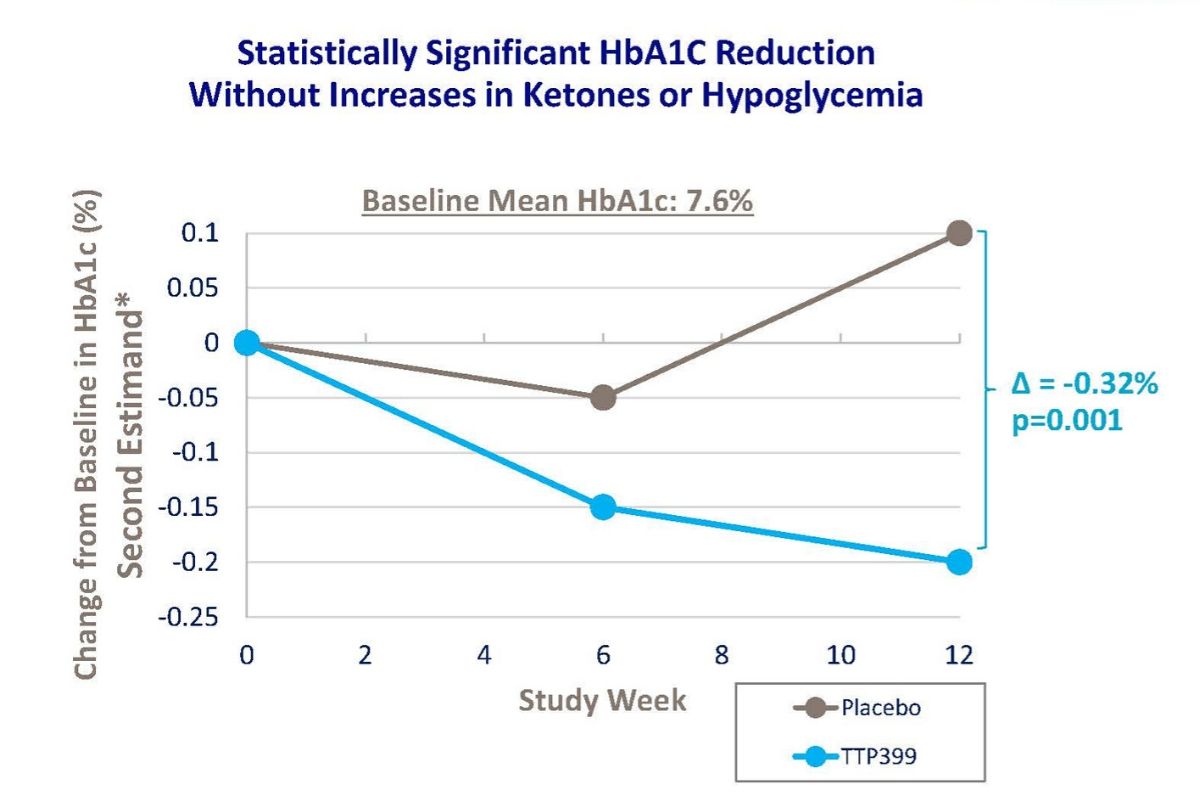
Nearly 80 percent of people with type 1 diabetes (T1D) fail to meet HbA1c goals defined by the American Diabetes Association. Despite more widely adopted diabetes technology and an increase in use, there is no improvement in clinical outcomes. This tells us that the current use of insulin alone is not enough, but vTv Therapeutics may have an answer.
In a Breakthrough T1D-funded clinical trial, they tested the safety and efficacy of TTP399, an oral therapy to be used in conjunction with insulin, in 85 people with T1D. The study successfully achieved its primary objective by demonstrating statistically-significant improvements in HbA1c, compared to placebo, at week 12.
The daily time-in-range was improved by approximately two hours in people treated with TTP399 relative to placebo. TTP399 was well tolerated, and importantly, there were no reports of diabetic ketoacidosis—a complication of T1D—reported in either group. There was no incidence of severe low blood sugar in the treated group and only one incident in the placebo group. People taking TTP399 experienced fewer symptomatic low blood sugar episodes: two subjects taking TTP399 reported at least one event compared to eight subjects in the placebo group (no TTP399).
TTP399 is a glucokinase (or GK) activator. GK acts as a key regulator of sugar levels in the body. If blood glucose levels are deemed too high, activation of GK in the liver has been shown to increase glucose utilization, which in turn lowers glucose levels in the blood.
vTv Therapeutics joined forces with Breakthrough T1D in 2017, to test TTP399 in people with T1D. The positive topline results from this phase II clinical trial follow the positive results obtained in the previous smaller clinical study reported by Breakthrough T1D in June 2019.
The next step: A phase III towards the future path of a registration trial. Stay tuned.