One of our most promising avenues toward cures for type 1 diabetes (T1D) is cell therapies. Cell therapies replace destroyed beta cells with functional, insulin-producing cells to restore insulin therapy independence. Right now, there are nearly a dozen cell therapies in clinical testing—and OPF-310 is one of them.
OPF-310 cells are derived from porcine (pig) islets. This approach is different from other cell therapies in the pipeline, which are either harvested from human deceased donor pancreases or manufactured in the lab.

Xenotransplantation
Xenotransplantation involves transplanting organs, tissues, or cells from a non-human animal into a human. This approach holds promise to address the shortage of organs from deceased donors and is being investigated in clinical trials for kidney transplantation—and now islet transplantation, too. Yet, this isn’t the first time porcine-derived islets have been tested in T1D. In fact, before human insulin was widely available, insulin derived from pigs was commonly used to manage diabetes.
Porcine islets, like other cell therapies that come from a source other than the recipient, have the potential to activate an immune response after transplantation. To overcome this, OPF-310 uses an encapsulation device that protects the islets from destructive immune cells. This means that no immunosuppression is needed.
Cell therapies and immunosuppressants
Right now, the only approved cell therapy for T1D (Lantidra®) and most cell therapies in clinical testing require immunosuppression to prevent the recipient’s immune system from attacking the transplanted islets. This can often be a barrier to trial participation because standard immunosuppressants can come with side effects. Besides OPF-310, another cell therapy in the pipeline that does not require immunosuppression is Sana Biotechnology’s gene-edited islets.
Read on to learn more about the trial, who may be eligible for OPF-310, and the team in the Windy City performing these transplants.
Essential information about the trial
The OPF-310 clinical trial
- Sponsor: Otsuka Pharmaceutical Factory
- Trial site spotlight: University of Illinois Hospital & Health Sciences System
- Co-Investigator spotlight: Benito Valdepeñas III, Pharm.D., Co-Investigator
- Team member spotlight:
- Miranda Kipp, MSN, RN, Clinical Research Nurse
- Memunat Ogunmefun, MPH, BDS, Clinical Research Coordinator
The goal of this first-in-human, phase 1/2a study is to assess the safety, tolerability, and efficacy of OPF-310 transplantation and to define the recommended phase 2 dose. Right now, the trial is only enrolling at a single location: the University of Illinois Hospital & Health Sciences System (UI Health) in Chicago, IL. For eligible participants that live out of state or more than 100 miles from UI Health, reimbursement for travel expenses (such as flights, hotels, and food) is available.
A glance at the inclusion criteria
- 35 to 65 years old
- At least five years have passed since T1D diagnosis
- Unstable T1D despite using a hybrid closed-loop automated insulin delivery (AID) system under the care of a qualified diabetes team for at least 6 months prior to enrollment
- History of severe hypoglycemic episodes and/or hypoglycemia unawareness
People who enroll in the trial will need to meet with the team at UI Health before and after the transplant as a part of their participation in the study.
These visits include:
- Four onsite visits prior to surgery
- Follow-up visits with the endocrine team within the first four weeks post-transplant
- 18 follow-up visits within the first year after the transplant
- Long-term follow-up visits that continue throughout each participant’s lifetime
These visits include bloodwork and other tests that can let researchers know how well the cells are functioning and making insulin and ensure participants’ continued safety. Also, for a year after the transplant, participants are required to use a second continuous glucose monitor (CGM) so researchers can track changes in blood sugar levels.
As with all new therapies and treatments, there are some risks involved. For xenotransplantation in particular, there is the risk of developing zoonotic infections (those that can be transferred from animals to humans), or passing these infections to close contacts. Safety is the top priority for the study team at UI Health. To help mitigate these risks, the porcine islets undergo testing to ensure that they are sterile before transplantation, and participants are routinely tested for possible infections.
The risks associated with surgical transplantation, such as infections at the transplant site, are managed by detailed pre-operative care and assessments, a top-notch and highly experienced surgical team, and close monitoring post-transplant.
“The OPF-310 trial is an important first step toward new treatments for type 1 diabetes. It uses pig islet cells to help people who have trouble controlling blood sugar or experience severe lows,” explains Benito Valdepeñas, a Co-Investigator on the trial. “If you join, you will be closely monitored by a team of specialists who will guide and support you every step of the way.”
Get involved!
Right now, there are three people enrolled in this study—and ten more are needed. The team needs your help! If you or someone you know may be interested in the OPF-310 trial, learn more about the study or email bvalde2@uic.edu to connect with Dr. Valdepeñas and the study team to see if you are eligible to enroll.
Support OPF-310 clinical trial participation
Spread awareness and encourage participation by circulating information about the OPF-310 clinical trial widely in your communities: on social media, within your networks, at community events, and any other platforms where you can reach people who may be interested. Even more, help reach our community members who might be hesitant by addressing clinical trials misconceptions—and reminding people that safety is always the top priority.
Clinical trials are the key to cures
The team at UI Health agrees: the future is looking bright for T1D. “Every one of these innovations began as an idea and relied on volunteers willing to help bring it to life,” says Miranda Kipp, a Clinical Research Nurse. From the perspective of Memunat Ogunmefun, a Clinical Research Coordinator, “…the work we do now could lead to new and improved treatment options for patients in the years to come.” It’s true: the only way to bring therapies like OPF-310 to life is through clinical trial participation, which can transform the lives of the T1D community now and in the future.

“I’m inspired by the possibility that emerging trials, like OPF-310, could give patients a level of stability and freedom they’ve never had before. Being part of research that could potentially ease their daily burden, prevent complications, and offer a more hopeful future is what drives me. Ultimately, my inspiration comes from the patients themselves.” – Benito Valdepeñas III, Pharm.D., Co-Investigator
“…participation in this trial goes beyond contributing to data, it is about being a part of an effort to explore therapies that could potentially change how we treat type 1 diabetes. Every participant plays a vital role in helping us evaluate both the safety and effectiveness of OPF-310, and their involvement directly shapes the future of care. We recognize that joining a clinical trial is a big commitment, so we prioritize patient safety, transparency, and support at every stage.” – Memunat Ogunmefun, MPH, BDS, Clinical Research Coordinator


“…your safety and support are our top priorities throughout this study. After your transplant, you will receive very close follow-up from the UI Health team to make sure you’re healing well and have everything you need. Once this early period is complete, your care will smoothly transition back to your regular healthcare provider, with our teams staying closely coordinated. Our goal is to make you feel supported at every step while ensuring you continue to receive care from the providers who know you best.” – Miranda Kipp, MSN, RN, Clinical Research Nurse
We’ve come a long way: cell therapies with curative potential, like OPF-310, are in clinical trials. The only way to get these therapies through the testing process and into the hands of people with T1D is through clinical trial participation. We need volunteers from the T1D community to help us get there. Consult our clinical trials web page to learn more about how you can get involved. Use our Clinical Trials Matching Tool to find a trial near you that you or a loved one may be eligible for. Connect with a Clinical Trial Education Volunteer in your area to have all your questions answered. We need your help!
Breakthrough T1D extends a heartfelt thank-you to Dr. Valdepeñas, Ogunmefun, and Kipp for taking the time to speak with us about the OPF-310 trial and what inspires them to do T1D clinical research. Breakthrough T1D also thanks Otsuka Pharmaceutical Factory for making this connection and driving research into cures for T1D.
This article was written by Alecia Wesner (pictured above, right), a Participant Advisory Council (PAC) member, a Clinical Trial Education Volunteer, and long-time Breakthrough T1D supporter. Alecia’s story details her lived experiences with type 1 diabetes, participation in clinical trials, and her journey to joining the PAC. The views expressed by the author are her own and are not necessarily representative of Breakthrough T1D or our leadership, employees, or supporters.
Where it all began

I was diagnosed with type 1 diabetes (T1D) in 1979 in suburban Philadelphia. I had just finished kindergarten, learned to ride a bike, and spent summer days pedaling the block. I was thirsty all the time. I was a new little cyclist and it was summer, of course I was thirsty! One day I drank an entire quart of iced tea, within mere minutes, at a neighbor’s house. That was the red flag. My pediatrician sent us to the local hospital, and multiple nurses and a doctor held me down as they repeatedly struggled to find a vein for a blood draw. My Dad took me for pancakes afterwards—definitely not ideal without insulin. Later, there was a phone call and my Dad left the house. I saw him sitting outside with his head in his hands. It was the first time I saw him cry. My Mom pulled me away from the window.
I spent the next week and a half in a children’s ward of a Philadelphia hospital. My parents were trained to care for me, and I practiced giving insulin shots on an orange while arts and crafts kept me busy. I spoke to my parents every night on a pay phone, whispering, while crying that I wanted to come home. My parents would say the other children in the ward needed me to be brave. This was long before continuous glucose monitors (CGMs) or even portable blood glucose meters. My parents learned to check my glucose by testing my urine with chemicals in test tubes, a non-exact way to measure glucose range. When I came home, neighbors were anxious because many believed diabetes was contagious.
A designer’s perspective on T1D clinical trials

Clinical trials and research have been central to my life. Clinical trials are essential to proving a concept, where ideas are tested rigorously and then refined.
My training as an industrial designer has always been about the intersection of people and systems. Whether designing a lighting fixture, a user interface, or an educational program, I think first about human needs, usability, and empathy.
That design lens informs how I view research. A study protocol is not just a list of procedures. It is a system that has users, interfaces, and outcomes. Participants are the users. Devices and consent forms are the interfaces. The data is the outcome. When design thinking is applied to research, studies become more accessible, clearer, and better aligned with the realities of daily life.
A defining experience
For those of us living with type 1 diabetes, research is not just academic—it is the path to a better quality of life. I have participated in nine clinical trials and numerous studies. I tested early automated insulin delivery (AID) systems over many years, assessing software and hardware which led to commercially available AID systems. I tested an implantable continuous glucose monitor, and I took part in studies that examined complications and diabetes biomarkers. Each trial taught me about technology, the human body (specifically mine), and how research truly impacts lives.
The experience that defined my clinical trial pathway was in my mid-20’s. I developed retinopathy. Due to a great macular specialist, I received aggressive laser treatments for years. It was scary, especially as a young professional starting a career in product development and visual design. The treatments that preserved my vision were possible because others had volunteered for research for years, even decades, before me. That reality gave me enormous gratitude, and it also gave me a sense of responsibility. Participating in research is how I have been able to embrace a life philosophy of “Do good, feel good” and ultimately pay it forward. Clinical trials and research studies are how we learn and design better solutions for everyone who follows.
Joining the Participant Advisory Council

My designer’s perspective led me to join the Breakthrough T1D Participant Advisory Council (PAC). I had been involved with Breakthrough T1D for many years as a Clinical Trial Education Volunteer, an advocate, and former board member. The PAC offered a new opportunity to bring lived experience directly into the research process.
On the council, we review trial designs, advise on participant burden, critique consent language, and recommend practical changes in the hope of lowering barriers to participation. My concern initially was that I’d be a token voice, but we are not. We are advisors who help researchers see the experience through the eyes of the people who will take part in the studies—people who may ultimately benefit from the data gained.
One of the most meaningful ways I contributed as a PAC member was in June 2025, when I spoke on a panel about disease-modifying therapies (DMTs) at the American Diabetes Association Scientific Sessions in Chicago. The panel focused on disease-modifying therapies designed to preserve C-peptide (a biomarker for insulin production) and slow or stop the progression of T1D. I represented the lived experience, and I brought the perspective of a long-term clinical trial participant and an educator who helps others understand and navigate research opportunities.
Using my voice to spread awareness about clinical trials

There were numerous questions to be addressed. One question asked how patients view trials targeted at specific subsets of the T1D population compared to all open trials. My answer was that subsets are still part of the whole community, and a win in one group can expand to broader benefits. At the same time, being excluded from any study can feel deeply personal. People who have made the decision to participate and then hear they do not qualify may feel a sense of loss and frustration. That is where clear communication and education matter. Explaining why inclusion and exclusion criteria exist, offering information about other potential trials, and keeping lines of communication open helps people stay engaged rather than feeling shut out.

Recruitment, especially for new onset trials, is another challenge the panel addressed. Speed matters because opportunities for early intervention are time-sensitive. Effective recruitment requires a network approach. Endocrinologists and clinicians are critical, but so are community organizations, advocacy groups, and peer networks. Many newly diagnosed people search for help and connect with the T1D community to find resources. When trusted peers share information about trials, it not only demystifies the process and reduces the emotional burden for families facing a new diagnosis—it opens doors.
We also discussed patient-relevant endpoints in DMT trials. Clinical metrics matter, but they do not capture everything people live with. Participants prioritize outcomes that reduce daily burden, delay disease progression, improve long-term health, lower the risk of severe hypoglycemia, preserve endogenous insulin, enhance glycemic control, potentially reduce lifetime treatment costs, and sustain hope. These are the practical changes that matter in everyday life.
Education and implementation were the final themes I emphasized. Clinicians and patients alike need clear information about DMTs and about how early interventions would be implemented in real-world settings. Linking DMTs to screening efforts ensures that people in the early stages of T1D are identified and connected with options. Volunteers, advocacy groups, and community programs are essential for amplifying success stories and helping clinicians and patients understand how to access emerging therapies.

“Research is deeply personal”
All of this ties back to design. When we include lived experience in research design, the results are more humane, relevant, and practical. We reduce friction that prevents people from participating, and we improve outcomes by focusing on the needs of real users. My perspective as both a person who lives with T1D and an industrial designer helps me see where small design decisions have large effects.
Looking forward, I remain committed to this work because research is deeply personal. It saved my vision. It offers better tools and brighter possibilities. It is also a way to honor those who volunteered before me and to give others the chance to benefit. Whether I am advising on study protocols, speaking on national panels, or teaching people how to find trials, I see it as part of the same lifelong commitment: to make life with T1D easier, to improve the design of treatments and systems, and to help guide us toward the ultimate goal: cures.

If you or someone you know may be interested in joining the Participant Advisory Council, please reach out to Michelle Simes-Kennedy at MSimes-Kennedy@BreakthroughT1D.org.
Learn more about clinical trials
Visit Breakthrough T1D’s clinical trials web page to learn more about how you can get involved in clinical trials. Use our clinical trial matching tool to find recruiting trials near you that you may be eligible for. Connect with a Clinical Trial Education Volunteer in your area to better understand the process and get your questions answered.
What is the pipeline?

Every new medical device, therapy, treatment, and drug—including those for type 1 diabetes (T1D)—goes through the drug development pipeline. Getting a new therapy or device from the earliest stages of research eventually into the hands of people with T1D is a complicated process. Science takes time (from years to decades!), money (from hundreds of millions to billions!), and brainpower (lots and lots of brilliant scientists, doctors, researchers, and more)—and lots of it.

That’s what drives us and our work in the pipeline: people with T1D doing better.
This process is complex, to say the least. There are safety checks at every step of the way. Data are scrutinized, and preclinical and clinical testing must meet ethical standards. In fact, many new drugs and devices don’t make it very far in the pipeline—and those that do take a long time to get there.
Breakthrough T1D is unique in that we work across the entire pipeline—from start to finish—for every promising therapy or device that we invest in. Between Research, Advocacy, and Medical Affairs, we work at every single step to accelerate progress and get new treatments to people with T1D faster than ever.
That’s our value proposition. That’s what makes supporting us different than supporting an individual researcher, or a university, or a company.
Let’s dive a little deeper into the pipeline—and how we’re turbocharging it.
Time and money
Total money spent in the diabetes space in FY24:
$146 million
By Breakthrough T1D
$521 million
Total T1D research support, including by Breakthrough T1D
$160 million
By the Special Diabetes Program
$412.9 billion
In healthcare
Total investments in new drugs and devices:
12 to 15 years
Estimated time it takes a new drug to get to the clinic
$1 billion
Estimated total cost to bring a new drug from discovery research to the clinic
3 to 7 years
Estimated time it takes a new medical device to get to the clinic
$522 million
Estimated total cost to bring a new medical device from discovery research to the clinic
The pipeline in action: Automated insulin delivery systems

Automated insulin delivery systems vs. artificial pancreas systems
At the very beginning, these devices were called artificial pancreas (AP) systems. Today, they are called automated insulin delivery (AID) systems. We’ll be referring to them as AID systems going forward.
The process at a glance:
$171 million
The amount of money Breakthrough T1D spent on AID system-related research from 2005 to 2024
12 years
The amount of time it took for next-generation AID systems to go from discovery research to clinical adoption after Breakthrough T1D became involved
Hundreds of millions of dollars
Total investment in AID systems from all stakeholders
60+ years
The amount of time it took for AID systems to become a reality, starting with the first experimental AP system in 1964
A deeper dive into the process:

Breakthrough T1D x AID systems
Breakthrough T1D has played a significant role in the evolution of AID systems. Doug Lowenstein, a long-time Breakthrough T1D volunteer and supporter, detailed the history of AID systems from the very beginning, and how we accelerated progress at every step of the way.
Discovery research
It’s the early 2000s. I’m a new AID system, but I don’t exist yet—I’m just an idea. I live in the minds of some scientists and researchers who think that I have the potential to one day become a reality. But, turning an idea into reality can be hard. It’ll take a lot of people, time, and effort for me to exist—but it’s not impossible. The question is: how? Answer: it starts with funding.
2005: Breakthrough T1D launches the Artificial Pancreas Project
Breakthrough T1D dedicates funds to scientists and researchers who have compelling ideas to turn AID systems into a reality, marking the start of the decades-long Artificial Pancreas Project (APP). These investments were key in jump-starting research into all the components needed to make an AID system work.
“[The goal was] to keep people alive and healthy until we find a cure. We were losing people…overnight of low blood sugars. If we could automatically dose insulin and have everybody go to sleep and all wake up, that was an incredible victory.” -John Brady, member of Breakthrough T1D Executive Committee in the early 2000s, former Chair of Breakthrough T1D’s International Board of Directors, and father of a son with T1D
Translational research
It’s 2008. Scientists are making progress on the three key parts that need to work in harmony for me to become a reality: a continuous glucose monitor (CGM), an insulin pump, and an algorithm that lets them talk to each other. But, like most new ideas, before I’m allowed to be tested on humans, I need to be tested in animal models first. Lucky for me, I skipped this step because someone made a tool that could simulate how I would act if I was attached to a human being.
2008: Breakthrough T1D-funded scientists create simulator tool to bypass animal studies
The simulator tool, developed from the initial Breakthrough T1D grants, allowed scientists to model how an AID system would respond to real-life scenarios, like eating a certain amount of carbs or exercising, and how this would translate to blood sugar outcomes like time in range or hypoglycemic events. The FDA accepting the use of this tool was a major win—without animal studies, AID system development was accelerated by years.
“The simulator saved at least five years of animal studies because we didn’t require an algorithm to be tested in an animal model to be deemed safe and effective before going into human studies. That entire chunk was eliminated.” -Sanjoy Dutta, Ph.D., Breakthrough T1D Chief Scientific Officer
Clinical trials
It’s 2012. Scientists have come up with different versions of my components that are ready to be tested in humans. For the next four years, I’ll be attached to people with T1D around the world who courageously volunteered themselves to test if I actually work. Turns out, I do a pretty good job at managing blood sugar—and I’m safe!
2012 to 2016: AID systems make headway in clinical trials
While clinical trials for AID systems started in earnest in 2008, they kicked into high gear in 2012, when investigators started conducting trials using different AID components and algorithms in real-life settings. Progress moved quickly, and results from numerous studies supported the idea that AID systems are both safe and effective. Breakthrough T1D funding—along with the Special Diabetes Program—helped move these trials forward.
“Getting involved in AID system trials to me was my chance to pay it forward for somebody else. I have lived 45 years with T1D…I think there’s something comforting in knowing that my body was used for something that not only had the potential to make me healthier, but really was for other people. I do think there’s something to be said for doing good, feeling good, and this is what it felt like being part of trials.” – Alecia Wesner
Regulatory approval
Flashback to 2009. While scientists are busy figuring me out, people at Breakthrough T1D are already thinking about and planning for my future—and how to work with the decision-makers at the FDA who will ultimately decide my fate. The FDA has also given their two cents about the best ways researchers can test me in clinical trials to get the data needed for me to get approved. Fast forward to 2016, it finally happens: I get FDA approval!
2009 to 2016: From APP roadmap to FDA approval
In 2009, Breakthrough T1D published the AP Roadmap, detailing what the future of AID systems will look like—and how we plan to get there. Two years later, we worked with the FDA on clinical trial design for AID systems so that there was a clear path to approval. After human clinical trials, the FDA approved Medtronic’s hybrid closed-loop MiniMed 670G—officially marking the first AID system to be available in the U.S.
“I do give [Breakthrough T1D] credit for pushing, for saying there’s a real need for this.” -Jeff Shuren, M.D., Head of the Center for Devices and Radiological Health at the FDA at the time
Access
Flashback to 2008. At this time, people were still unsure if one of my main components—a CGM—was a reliable way to measure blood sugar. This all changed when a first-of-its-kind clinical trial showed that CGMs are better than finger pricks and glucose meters, and they were covered by insurance shortly after. This decision paved the way for my future: after my approval in 2016, insurers began offering to cover me.
2008 to 2017: Insurance coverage evolves
The landmark Breakthrough T1D-funded clinical trial in 2008 provided the data to convince private insurers to cover CGMs. Nearly a decade later, Breakthrough T1D launched its “Coverage2Control” campaign to advocate for insurance coverage of T1D treatments, therapies, and devices—including AID systems—ultimately resulting in all major private insurers offering coverage of AID systems by the end of 2017, followed by Medicare shortly after.
“Seeing the artificial pancreas go from concept to reality…is what makes Breakthrough T1D and all of the advocacy volunteers—who sent an email, made a call, signed an action alert, or met with their Member of Congress—very proud of this historic achievement and the impact that these will have on the individual lives of those with type 1 diabetes.” -Cynthia Rice, former Chief Mission Strategy Officer at Breakthrough T1D
Adoption
It’s present day. There are tons of iterations of me, and even more coming. People get to choose which version of me they like best. I’m covered by most health insurance. I’ve come a long way since I was just another thought in the minds of a few scientists who had a vision… and now I’ve come to life! Even so, not everyone has chosen to use me yet—and I will continue to evolve and grow until I can make the lives easier of as many people with T1D as possible.
2017 and on: More and more AID systems come to life
After the first hybrid closed-loop AID system was approved, the flood gates were opened. More and more systems are coming to market each year, and they keep getting better. They’re smaller, easier to use, and better at managing blood sugar. They’re covered by both government and private insurance plans. They’re an integral part of routine discussions between people with T1D and their healthcare providers, and people have options to choose which system is best for them. This is a future that was difficult to imagine two decades ago—and now it’s a reality. Even so, the work continues until AID systems are a reality for more and more people with T1D.
“What we brought to bear is resulting in a safer and easier life for hundreds of thousands, and soon millions, of people with T1D, including my son, that is going to keep them safe until something like a cure comes along,” -Jeffrey Brewer, one of the APP founders
The final stage: Cures and improved lives
It took tons of time, money, people, and effort to get us where we are today, but we’re not at the finish line yet. “The end game for AID systems,” says Breakthrough T1D CEO Aaron Kowalski, Ph.D., “is to have multiple compatible pumps, glucose sensors, and algorithms, so that patients can mix and match what they prefer.”
The end game for T1D as a whole, however, is cures. AID systems have greatly improved the lives of those with T1D—and will continue to do so now and in the future—while we continuously work toward cures that are one day available to everyone with the condition.
The path followed for AID systems is a roadmap for other therapies coming down the pipeline. Breakthrough T1D’s Project ACT is taking a page from the AP roadmap and applying it to cell therapies, so that functional cures can get to people with T1D who want them as quickly and safely as possible.

While I’m proud of my work as a scientist at Breakthrough T1D on AID systems (and my brother and I currently wear AID systems that are derived from Breakthrough T1D-supported work), more than anything else I want to take off my diabetes devices and achieve what our founders set out to do—find cures for T1D.”
Breakthrough T1D successfully took an idea and turned it into a reality—and we’ll do it again and again until T1D is a thing of the past.
What’s happening?
Today, Bayer shared data from the phase 3 FINE-ONE clinical trial. These results, which were presented at the American Society of Nephrology Kidney Week in Houston, TX, showed that finerenone (Kerendia™/Firialta™) significantly reduces urine albumin-to-creatinine ratio (UACR), a measure of kidney damage, in people with chronic kidney disease (CKD) associated with type 1 diabetes (T1D). This is an exciting win for the T1D community!

Finerenone
Finerenone is a drug that has already been approved for the treatment of CKD in type 2 diabetes. In CKD, the hormone aldolsterone is overactive, leading to kidney damage. Finerenone blocks this hormone’s activity to protect the kidneys from further damage.
What did the FINE-ONE study show?
The global, phase 3 FINE-ONE clinical trial investigated the use of finerenone in people with CKD associated with T1D. This trial achieved its primary outcome: finerenone significantly reduced UACR—a measure of kidney damage—compared to placebo.
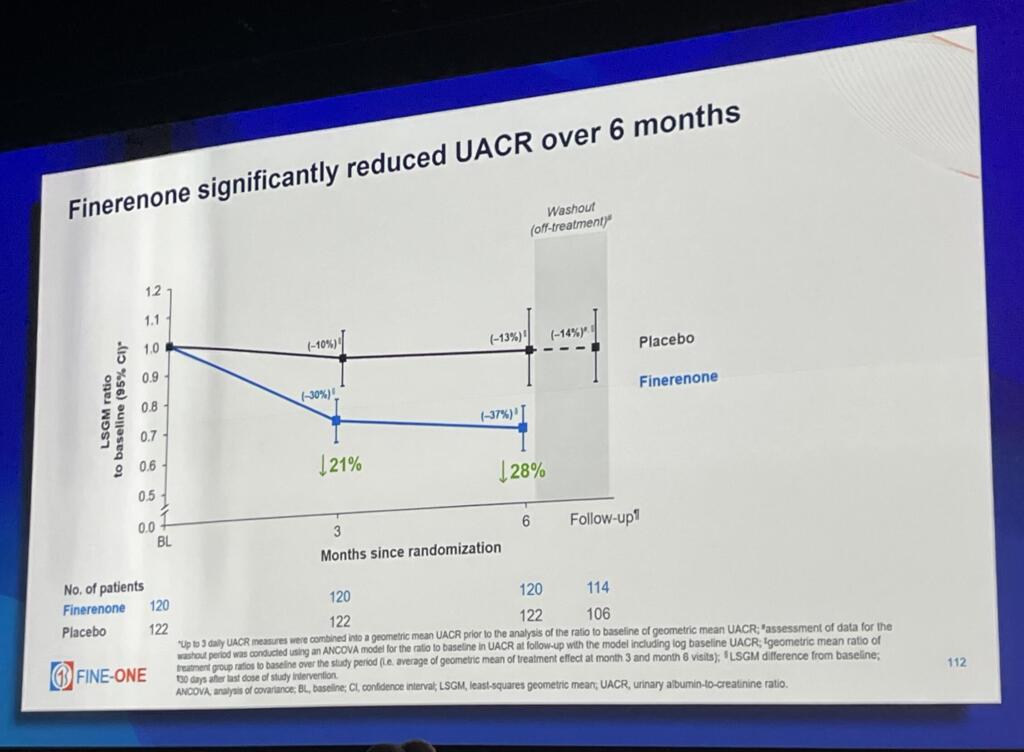
So, what does this mean? Previous clinical trials with people who have type 2 diabetes and CKD have shown that reductions in UACR from finerenone are associated with positive long-term health outcomes. In people with T1D, reductions in UACR caused by finerenone are likely indicative of lower risk of kidney disease progression, kidney failure, and cardiovascular events.
Finerenone was well-tolerated, with no new safety issues reported and few serious adverse events. Based on these results, Bayer intends to submit the data for regulatory review, with the goal of expanding finerenone’s indication to include treatment of CKD in people living with T1D. If finerenone gets approved, this would be an incredible step forward for people living with the burden of both kidney disease and T1D.
Why does this matter for T1D?
CKD is one of the most common complications of T1D. Nearly a third of people living with T1D will develop CKD, increasing the risk of both kidney failure and cardiovascular disease. Right now, the treatment options for people with T1D and CKD are limited—and there is a major need for therapies that can address this.
Finerenone is the first therapy in three decades to achieve positive outcomes for CKD in people with T1D. This is a huge step forward in the right direction, and this exciting data has the potential to transform the lives of people living with T1D with CKD if finerenone gets regulatory approval.
The bigger picture
Breakthrough T1D strategically collaborated with Bayer to support the FINE-ONE clinical trial, and we are committed to further collaboration with Bayer to advance therapies for people with T1D. Working together with Bayer and other companies will allow us to deliver more treatments to people with T1D to address complications like kidney disease.

“People with type 1 diabetes and chronic kidney disease face an immense burden due to their increased risk for both kidney and cardiovascular events,” said Jonathan Rosen, Ph.D., Research Director at Breakthrough T1D. “Breakthrough T1D remains committed to collaborating with Bayer to improve kidney care for people with type 1 diabetes.”
As a part of our Improving Lives portfolio, we aim to identify and support treatments and therapies that can address complications that arise from T1D, like CKD. The FINE-ONE trial opens the door for more therapies to come that could transform the landscape of available therapies for people with T1D, and industry investment in this space helps drive progress forward faster than ever. The T1D community needs more options, and our strategic industry partnerships will help make this possible.
We look forward to seeing the regulatory decisions that stem from FINE-ONE, and we are incredibly excited for what the future holds for CKD and T1D.
What’s happening?
Earlier today, Eli Lilly and Company announced that they are launching two new clinical trials for baricitinib in type 1 diabetes (T1D). These phase 3 trials will investigate whether the drug can delay T1D onset or progression and will open for recruitment soon. Read on to learn more about the trials, why this matters, and what led to this exciting news.
Baricitinib has shown promise for T1D
Baricitinib is a JAK inhibitor that interferes with signaling pathways critical to both immune cells and beta cells. This drug has the potential to extend the “honeymoon period” of T1D, meaning that it could preserve remaining insulin-producing beta cells earlier in disease progression. More beta cells mean better blood sugar management—and potentially reduced long-term complications.

JAK inhibitors
JAK inhibitors, including baricitinib, are already FDA-approved for other autoimmune diseases, such as rheumatoid arthritis, alopecia, and more. JAK signaling pathways are associated with overactive immune responses, so blocking this pathway may turn down the immune response.
The phase 2 Breakthrough T1D-funded BANDIT study was key in showing that this drug is safe and effective in T1D. This study was led by Professor Thomas Kay, MBBS, Ph.D., and his team at St. Vincent’s Institute of Medical Research in Australia. In this trial, more than 90 newly diagnosed individuals with stage 3 T1D between the ages of ten and 30 were treated with either baricitinib (as a single, daily oral tablet) or placebo for 48 weeks.
The results were positive: people treated with baricitinib had increased insulin production, spent more time-in-range, needed less external insulin, and experienced minimal side effects. These data were published in the prestigious New England Journal of Medicine and inspired the launch of another clinical trial (JAKPOT T1D) investigating different JAK inhibitors in preserving beta cells in T1D.
Thanks to the BANDIT study, which stemmed from years of work with Lilly to supply the drug for the trial, we got the convincing data needed to move baricitinib to phase 3—and one step closer to people with T1D.
The new studies
The new studies will be recruiting soon at medical centers around the globe. Participants in either trial will randomly receive either baricitinib or placebo. The studies are also double-blinded, meaning that neither the study team nor individuals will know whether they are administering or receiving baricitinib or a placebo. Importantly, baricitinib is a once-daily oral pill—meaning its use is simple and easy.
BARICADE-DELAY
- Purpose: To determine if baricitinib can delay the onset of clinical stage 3 T1D in people who are at high risk
- Eligibility: Must be between one and 36 years old and be in stage 2 T1D
- Time commitment: Up to approximately five years
- Primary outcome: Time to diagnosis of stage 3 T1D
BARICADE-PRESERVE
- Purpose: To determine if baricitinib can preserve beta cell function in people with newly diagnosed T1D
- Eligibility: Must be between one and 36 years old and diagnosed with stage 3 T1D within the last 100 days
- Time commitment: About 60 weeks
- Primary endpoint: C-peptide preservation (a measure of insulin production)
Breakthrough T1D is a supporter of baricitinib
Breakthrough T1D has been a tireless advocate for the use of JAK inhibitors in T1D. In conjunction with our Australia affiliate, we funded the BANDIT study, which generated the positive data that provided the basis for these new clinical trials.
Baricitinib is a cornerstone of our Disease-Modifying Therapies Program, which aims to find treatments that can delay, halt, reverse, or prevent T1D. The progression of baricitinib through the pipeline is a perfect example of what we do best: identify a promising therapy, collaborate with industry to support or fund initial clinical trials that establish a therapy’s effects in T1D, facilitate the path towards an industry-sponsored phase 3 trial, and help recruit trial participants to accelerate progress.
We will continue to follow these studies closely and look forward to seeing the results. We encourage our community and supporters to spread the word—if you or anyone you know may be eligible for these trials, share this information!
Finally, we thank Lilly for their shared commitment to our mission: a world without T1D. Industry investment in clinical trials for T1D helps us progress faster—and with partners like Lilly, we’ll get more therapeutic options to people with T1D sooner.

“Breakthrough T1D has been a tireless advocate for the use of JAK inhibitors and has seen the promise of baricitinib as a therapy for type 1 diabetes to preserve insulin production and delay disease progression,” said Breakthrough T1D Chief Scientific Officer Sanjoy Dutta, Ph.D. “We’re thrilled that Lilly is advancing clinical research with baricitinib and thank them for moving us one step closer to making it available for people with type 1 diabetes.”
“In the BANDIT trial we tested for the first time whether baricitinib could preserve insulin-producing cells under autoimmune attack in type 1 diabetes. Years of laboratory work indicated that this drug should be effective. Baricitinib given for 12 months maintained insulin production compared with its loss with placebo. We are grateful to Breakthrough T1D and Lilly for their support of the trial. This was a very exciting outcome from our bench to bedside research. Current insulin therapy is life-saving in diabetes but it is imperfect, and new treatments such as baricitinib show promise. Preservation of insulin production allows the body’s own amazing processes to control blood glucose and reduce the burden of type 1 diabetes. We hope that the BARICADE trials will confirm our findings and extend them to people with earlier diabetes, before insulin is required, and lead to the use of baricitinib in clinical practice for type 1 diabetes.” – Professor Tom Kay, Director, St Vincent’s Institute of Medical Research (SVI) in Melbourne, Australia

Get involved in clinical trials
Clinical trials are key to bringing new treatments, devices, and therapies from labs and into clinics. People who choose to participate in clinical trials help drive progress forward for the entire T1D community. If you or anyone you know is interested in participating in a clinical trial, use our Clinical Trials Matching Tool to find a trial that you may be eligible for. You can also reach out to a Clinical Trial Education Volunteer in your area to connect and answer any questions you may have. Information about the new baricitinib clinical trials will be available on our clinical trials web page when enrollment begins.
Breakthrough T1D had the pleasure of speaking with Patti Columbe, a participant in Vertex’s pivotal trial for the manufactured islet therapy zimislecel (formerly VX-880). Patti has been living with type 1 diabetes (T1D) for nearly 39 years. This is one of the most exciting T1D trials in terms of getting us closer to cures for T1D.
In this piece, we’ll talk about Patti’s T1D journey and what led to her successful enrollment in this landmark clinical trial.
The opinions, views, and experiences detailed in the interview below are those of Patti and are not representative of other participants’ experiences in this clinical trial, nor are they representative of Vertex Pharmaceuticals or Breakthrough T1D.
The conversation has been edited for length and clarity.
Q: Patti, can you tell us about your diabetes journey?
A: I was diagnosed with T1D in October 1986. I’ve lived with it for nearly 39 years. As anyone who knows this disease can tell you, it’s very hard. I’ve used every possible management system—pumps, continuous glucose monitors (CGMs), inhaled insulin, and I even have a diabetic alert dog named Otto. Despite these tools, I always struggled with low blood sugars, and I struggled with the mental burden of this disease. I was the kid at diabetes camp who had to get a glucagon injection in the middle of the night.

Q: What did you use to manage your T1D?
A: I’ve done everything—pumps, injections, CGMs, everything. But now I use multiple daily injections. I also rely on my diabetic alert dog—Otto! Otto, my service dog would often jump in my bed 20 minutes before a CGM would go off. And he consistently does that!
How did you hear about the clinical trial?
A: Having been around T1D for so long, I knew about the concept of islet transplants. I also knew that the end goal is making these cells. It was exciting but not something I thought was a reality today.
About two years ago, I heard about a clinical trial being done by Vertex. I found out about it on Instagram, actually. I then reached out to them and began the screening process.
Breakthrough T1D x Vertex Pharmaceuticals: Where it all began
Breakthrough T1D has played a role in the evolution of zimislecel for decades. In 2000, Breakthrough T1D awarded a grant to Douglas Melton, Ph.D., who developed a cutting-edge protocol to grow beta cells in a lab. Dr. Melton later founded Semma Therapeutics to turn these cells into curative therapies for T1D. In 2017, The T1D Fund made a catalytic investment in Semma, which was then acquired by Vertex in 2019—accelerating zimislecel’s development and leading to where we are today. We’ve been working closely with them ever since.
Q: What was the screening process like?
A: The screening was extremely thorough. I answered a ton of questions about my insulin usage, diet, blood type, history of hypoglycemia, and more. I even had to prove I had had T1D for all these years. They tested me for autoantibodies. They tested me for seemingly everything!
Q: How did it feel to make it into the trial?
A: Clinical trials like this are important. Clinical trials have to happen. We can’t get a cure without them, so somebody has to step up. I am lucky enough that I can step up—and I’m so glad to.
Participate in clinical trials
Clinical trials are an essential step in bringing new treatments and devices from labs to clinics. People who courageously choose to participate in clinical trials, like Patti, help drive progress for the entire T1D community. If you’re interested in participating in a clinical trial, use our Clinical Trials Matching Tool to find a trial near you. You can also connect with a Clinical Trial Education Volunteer in your area to answer any questions you may have.
Why this matters
Breakthrough T1D extends a heartfelt thank you to Patti for courageously sharing her story with us so we can truly understand why the work we’re doing is critically important. If you choose to participate in a clinical trial, like Patti, you have the potential to provide transformative benefits to the entire T1D community by accelerating the pace of clinical research. Learn more about clinical trials and how you can get involved. If you’re interested in the zimislecel clinical trial, check out the trial sites and eligibility criteria.
To accelerate T1D cures faster than ever, Breakthrough T1D’s Project ACT (Accelerate Cell Therapies) is simultaneously advancing research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression. We will not stop until these therapies are available to everyone with T1D who wants them—and clinical trial participants like Patti are getting us there faster.
We are well on our way to seeing manufactured cell therapies like zimislecel come to life. We owe a huge thank you to our supporters and the broader T1D community for inspiring us every day to keep pushing forward until functional cures for T1D are a reality.
Disclaimer: Vertex Pharmaceuticals has donated to Breakthrough T1D.
This article was written by both Brian Herrick and Sandy Vogt.
Partners in clinical trials
Clinical trials are key to bringing new treatments, therapies, and devices for type 1 diabetes (T1D) from the lab to the clinic. Pharmaceutical and biotech companies, which conduct research and help develop and manufacture new therapies, are a critical player in accelerating this process. Companies often partner with clinical researchers at medical centers and hospitals to test their new therapies through clinical trials. These collaborative partnerships are essential for new drugs to move through the pipeline and get to people with T1D.
Clinical trials terms you need to know
Sponsor
The trial sponsor is the person, organization or company that initiates, manages, and oversees a clinical trial, ensuring it is conducted ethically and safely.
Trial site
A trial site is the medical center, hospital, or clinic where clinical trial participants receive the study drug, treatment, or intervention and receive care throughout the duration of the trial.
Principal investigator
Within a particular trial site, the Principal Investigator (PI) is the lead researcher that oversees the trial and the research team. The PI ensures participant safety, supervises data collection and analysis, and maintains scientific and ethical integrity during the research process.
Spotlight on the CATT1 clinical trial
The CATT1 clinical trial is investigating whether cadisegliatin (TTP399), a glucokinase activator, can reduce hypoglycemic events (low blood sugars) in people with T1D when used alongside insulin therapy. Read on to learn more about the people making the CATT1 clinical trial happen, the science behind cadisegliatin, and how you can get involved.
The CATT1 clinical trial
Sponsor
vTv Therapeutics (a company with support from the T1D Fund: A Breakthrough T1D Venture)
Trial site spotlight
The University of North Carolina (UNC) Medical Center located in Chapel Hill, N.C.
Principal investigator spotlight
Klara Klein, M.D., Ph.D., Assistant Professor of Medicine and Director of the Endocrinology, Diabetes, and Obesity (EnDO) Clinical Research Unit (CRU) at the UNC School of Medicine, Division of Endocrinology and Metabolism
Clinical research team member spotlight
Alex Kass, MSN, MBA, RN, CDCES, Research Program Director for the EnDO CRU
Breakthrough T1D x vTv Therapeutics
Breakthrough T1D has a long-standing relationship with vTv Therapeutics. After partnering with Breakthrough T1D in 2017, vTv pivoted from type 2 diabetes to T1D. With Breakthrough T1D’s support, vTv launched a phase 2 clinical trial (SimpliciT1) investigating cadisegliatin as an adjunctive therapy for T1D, showing that it improved blood glucose control. Eager to keep the momentum going, the T1D Fund has supported vTv from early 2024 onwards, most recently participating in the company’s latest round of financing to continue to support the ongoing phase 3 trial for cadisegliatin in T1D.

“Breakthrough T1D has long been a supporter of adjunctive therapies, meaning medicines that can be taken alongside insulin to improve glucose control and other outcomes. Previous clinical trials have demonstrated the potential for cadisegliatin to reduce hypoglycemia and improve HbA1C in people with T1D, and we are excited to support the phase 3 program to advance this therapy toward the clinic where it can be used to address major unmet clinical needs in our community.”
Jonathan Rosen, Ph.D., Director of Research at Breakthrough T1D
Meet the team at UNC
Within the UNC Medical Center, the EnDO CRU is a hub for endocrinologists, clinical researchers, and other experts who work together to lead clinical studies at cutting edge facilities that help bring new transformative therapies to the people who need them most. Within the EnDO CRU, Dr. Klara Klein and Alex Kass are taking the lead on T1D clinical trials—a passion they both share for different reasons.
T1D has been on Dr. Klein’s mind since she was a little girl. “I grew up hearing about diabetes—my mom is an endocrinologist, and it was a frequent topic at the dinner table,” she said. After following in her mother’s footsteps and becoming an endocrinologist herself, Dr. Klein is shocked that insulin—which was discovered over 100 years ago—is still the gold standard for T1D management. In this sense, not much has changed since she was sitting at the dinner table with her parents.
This sentiment inspires her work. “I’ve always been struck by the daily challenges people with T1D face. Even with incredible advances in technology, managing T1D is a constant effort—every meal, every workout, every illness,” she explained. Her true passion is bringing new therapies to fruition that can ease the daily burden of living with T1D until everyone has access to cures. “Being part of research that could improve the lives of people living with diabetes in meaningful ways is both exciting and deeply rewarding.”
Dr. Klein is supported by a passionate and experienced team composed of five endocrinologist sub-investigators, a nurse coordinator, and a research assistant. She also works closely with Kass, who leads the operational side of many different trials within the EnDO CRU.
Kass is one of a few other staff on the team who is also living with T1D, giving him an inside perspective and inspiring his career to help people with diabetes live better lives. “Having type 1 diabetes myself has fueled my passion for both patient care and finding better treatment options,” he explained. “Together, our team’s mission is simple: to run high-quality studies that give people with diabetes access to promising therapies and make their day-to-day lives easier. This shared passion in our group inspires me!”
The team is working hard behind-the-scenes to recruit and care for participants of the CATT1 clinical trial, which is just one of the many studies they are running to help people with T1D.
Everything you need to know about the CATT1 clinical trial
The phase 3 CATT1 clinical trial is investigating whether cadisegliatin can reduce moderate to severe low blood sugar (hypoglycemic) events when used alongside insulin therapy over a period of six months. Hypoglycemia is the most common short-term complication of T1D, and we need to do more to help people with T1D manage their blood sugar to reduce dangerous lows. This clinical trial is trying to do just that.
Based on observations from previous clinical studies, cadisegliatin may be able to lower the risk of hypoglycemia. Participants in the phase 2 SimpliciT1 study showed clinically meaningful improvements in blood sugar control in addition to a 40% reduction in hypoglycemic events compared to the placebo. Nearly 600 participants have received cadisegliatin so far—it’s been well-tolerated and hasn’t increased the risk of diabetic ketoacidosis (DKA).
Unexpected clinical trial results
Sometimes, clinical trials have unexpected results. In this case, researchers may pivot to change the design of future trials to ensure they’re looking for the right thing. For cadisegliatin, earlier trials primarily looked at improvements in control of high blood sugar, but researchers noticed significant reductions in hypoglycemic events, too. Now, this is the primary outcome for the CATT1 trial—while this wasn’t the original intention, it’s the most clinically meaningful assessment for people with T1D.
Dr. Klein, Kass, and their team are actively recruiting for the CATT1 trial over the next three or so months.
Some key eligibility criteria are:
- Minimum of five years since T1D diagnosis
- At least one moderate to severe hypoglycemic event (Level 2 or 3) in the last two months prior to screening
- 18 years of age or older
- HbA1c levels < 9.5%
- Use of a continuous glucose monitor (CGM) for at least three months prior to screening (excluding closed-loop systems)
Clinical classification of hypoglycemic events
| Level | Severity | Blood glucose range and characteristics |
|---|---|---|
| 1 | Mild | 54 to 70 mg/dL |
| 2 | Serious/moderate or clinically significant | < 54 mg/dL |
| 3 | Severe | An altered mental and/or physical state requiring immediate assistance to prevent progression to loss of consciousness, seizure, coma, or death. |
Participants, investigators, and care providers will all be unaware of whether the participants are receiving cadisegliatin or the placebo, which will both be given orally. Participants can expect a combination of in-person and virtual visits over a 26-week period. During these visits, the clinical research team will check blood sugar levels, perform other lab tests to assess safety, and provide top-tier T1D care.
At UNC, for the entire length of the trial, participants will have access to a dedicated study team including a nurse coordinator, research assistant, and diabetes specialists. The clinical research team will guide participants through every step of the process—answering questions, reviewing results, providing support—so that people with T1D and their families can feel confident that they are in good hands.
Interested in participating?
Complete a pre-screener questionnaire to see if you qualify!
The science behind cadisegliatin
We know that cadisegliatin has been shown to reduce the risk of hypoglycemia in people with T1D (SimpliciT1 study). The next question is: how?
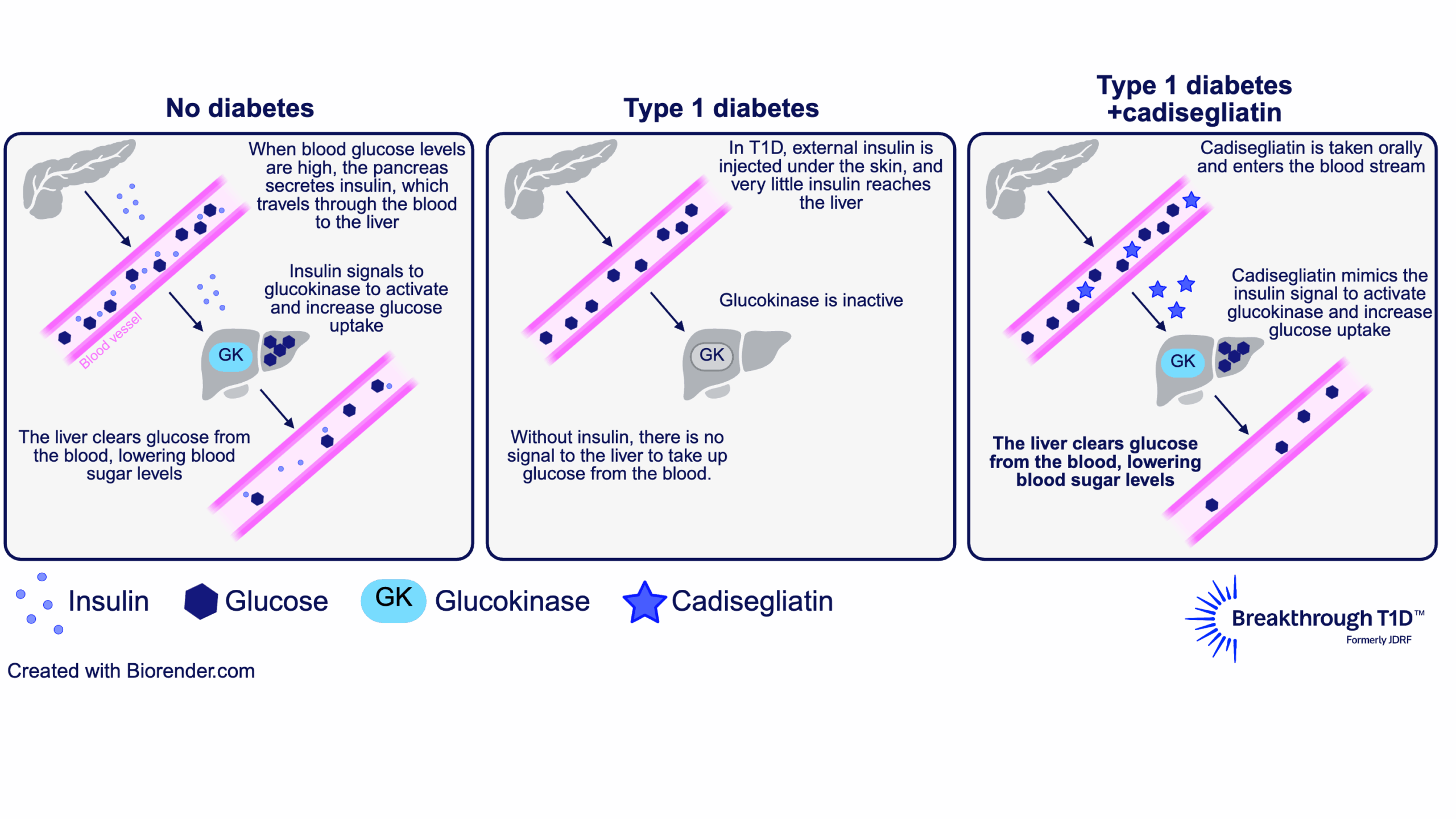
The answer to this question lies in the liver. The liver plays a major role in blood glucose control by taking up excess sugar from the blood and storing it until it’s needed again. To facilitate this, an enzyme called glucokinase in liver cells is activated in response to higher levels of insulin (indicating high blood sugar). This allows liver cells to remove glucose from the blood, bringing blood sugars down to a normal range.
Some of the glucose in the liver is also converted into glycogen, a storage form for glucose. This way, the liver can help increase blood sugar levels when needed, such as in between meals, overnight, or during exercise, to avoid low blood sugar. In this way, glucokinase helps reduce both hyperglycemia and hypoglycemia.
In people with T1D, the pancreas produces little to no insulin, and injected insulin doesn’t reach the liver the same way insulin from the pancreas does. So, glucokinase doesn’t get the signal that allows liver cells to respond to high blood sugar—making hyperglycemia and hypoglycemia worse.
This is where a glucokinase activator like cadisegliatin comes in. It keeps glucokinase in an active state, which restores the ability of liver cells to respond to (and reduce) high blood sugar. Then, in response to low blood sugar, the liver can release some of the sugar it has stored as glycogen into the blood stream. This is why cadisegliatin is being studied in people with T1D— it has the potential to improve blood glucose control in the liver and reduce the risk of highs and dangerous lows, with the goal of easing the daily burden of living with the disease.
Make a difference for yourself and others by participating in clinical trials
How to get involved
If you’re interested in getting involved at UNC, visit their Research for Me website to learn more about the CATT1 clinical trial at this location and other diabetes trials that may be the right fit for you. Reach out to the study team directly, and they will follow up with you to answer your questions and see if you qualify.
You can also register to be contacted by clinical researchers at UNC about participating in future studies for T1D. A research team member will reach out if a study becomes available that matches your profile criteria.
The CATT1 trial is also enrolling at other locations in the U.S. Find the trial site closest to you.
The clinical research team at UNC encourages trial participation as an opportunity to contribute to the body of research that may improve T1D care not only for yourself, but for the T1D community. Participants receive continued support at every step of the way—and safety is the highest priority.
By choosing to participate in a trial, people with T1D may gain access to research treatments, drugs, or devices that may make daily life with T1D easier, while receiving top quality diabetes care delivered by renowned medical centers. The data generated from these trials—thanks to the courageous and brave T1D community members who choose to participate—will shape the treatment landscape for future generations of people with T1D. Dr. Klein is one of many investigators who finds the dedication of the T1D community inspiring. “…I am consistently amazed and moved by the generosity of our participants who dedicate their time and energy to help move science forward,” she explains.

“The T1D community is incredibly generous with their time and motivated to help advance care—not just for themselves, but for the type 1 diabetes community and future generations of people who will live with type 1 diabetes. It’s really an honor to be able to work with them.”
-Klara Klein, M.D., Ph.D.
For Kass, it’s personal. By having T1D himself, he can form deeper connections with trial participants and their families. This allows him to keep conversations honest, participate in shared problem-solving, and understand the same day-to-day challenges of living with T1D—all a part of why he’s excited to be a member of the team providing top-tier diabetes care.
“Some of my favorite moments are when a participant or parent says, ‘You get it,’ and we can talk not just about the study, but about what it means to live well with diabetes. Whether it’s troubleshooting a CGM issue, celebrating a small win in blood sugar control, or simply being a dependable point of contact, I value the trust our participants place in us. That trust is what keeps me passionate about this work.”
-Alex Kass, MSN, MBA, RN, CDCES

Learn more about clinical trials
Visit Breakthrough T1D’s clinical trials web page to learn more about how you can get involved in clinical trials. Use our clinical trial matching tool to find recruiting trials near you that you may be eligible for. Connect with a Clinical Trial Education Volunteer in your area to better understand the process and get your questions answered.
Without clinical trials, progress wouldn’t be possible. All new therapies, devices, and treatments need to be tested to get from labs and into clinics as quickly and safely as possible. Thank you to every trial participant—in the past, present, and future—for helping us get closer to a world without T1D.
A new study funded by Breakthrough T1D and published in the journal Diabetes Therapy shows that people with type 1 diabetes (T1D) and caregivers are largely willing to try next-generation therapies, drawn by the promise of reduced insulin reliance, greater normalcy, and freedom from constant disease management.
The findings highlight the importance of ensuring patient and caregiver perspectives guide regulatory decisions around the balance of a therapy’s risks and benefits.
The promise of next-gen therapies
Advances in T1D management, such as automated insulin delivery (AID) systems, mark major progress over the past decade. Despite these gains, insulin therapy remains physically, mentally, and emotionally demanding, and substantial unmet needs persist.
Next-generation therapies aim to move beyond insulin replacement through two approaches:
- Disease-modifying therapies (DMTs): Preserve insulin production by slowing or halting autoimmune destruction of beta cells.
- Cell Therapies: Restore insulin production by delivering new functional cells, with the potential for partial or complete insulin independence.
For the study, researchers interviewed people with T1D and caregivers to better understand their perspectives on the risks and benefits of these therapies.
What participants said
Participants expressed willingness to try new therapies, with 92% of adults, 70% of caregivers, and 100% of adolescents saying they would try a DMT at the time of T1D diagnosis. Every participant said they would try cell therapies.
Participants also shared their thoughts on top benefits of both DMTs and cell therapies.
- Top benefits: DMTs: Reduced insulin reliance and an extended period when a
person with T1D produces their own insulin; cell therapies: insulin independence,
more normalcy, and freedom from constant demands on diabetes.
Why this matters
To approve a product, regulators must judge whether the therapy’s benefits outweigh its
risks. This study shows that , people living with T1D and their caregivers have strong view
about which benefits matter and how much they matter. Because DMTs and cell therapies
bring significant benefits compared to insulin therapy, regulators should incorporate these
patient perspectives into regulatory decision-making.
About the study
The Breakthrough T1D Patient Preference Study is being conducted in two phases. In the first phase, researchers interviewed 26 individuals—12 adults, 4 adolescents, and 10 caregivers—about their views on DMTs and cell therapies. These insights are now shaping a large-scale survey that will measure how the T1D community prioritizes different risks and benefits. Results from that survey are expected later this year.

The short version
Breakthrough T1D’s newest publication outlines what the future of beta cell replacement therapies looks like—and how we can make these therapies a reality for everyone with type 1 diabetes (T1D) who wants them through innovative clinical trial design and expanding the pool of eligible trial participants.
Breakthrough T1D, in collaboration with other leading experts in the field, recently published an article titled “Future Directions and Clinical Trial Considerations for Novel Islet β-Cell Replacement Therapies for Type 1 Diabetes” in the journal Diabetes. Breakthrough T1D Research and Advocacy staff who contributed to the publication include Sanjoy Dutta, Ph.D., Chief Scientific Officer, Esther Latres, Ph.D., Vice President of Research, and Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs.
Beta cell replacement therapies have the potential to cure type 1 diabetes (T1D) by removing the need for external insulin. While donor islet transplantation can result in insulin independence and results from early trials with manufactured islets are encouraging, people with T1D need better tools and therapies. This publication serves as a roadmap for the entire field—including researchers, product developers, industry, regulators, clinicians, and people with T1D—to address these needs and ensure that beta cell replacement therapies can get to people with T1D as quickly and safely as possible.
Read on to learn more about what the future of beta cell replacement therapies looks like.
Key Takeaways
- Currently available beta cell replacement therapies are limited to people with T1D with unstable blood sugar management, typically measured by HbA1c levels, in addition to dangerous lows (hypoglycemic events) that require immediate assistance. These therapies also require lifelong immunosuppression.
- New and improved beta cell replacement therapies are on the way, and we need to make sure that they are available for people with T1D beyond those with unstable blood sugar management and severe hypoglycemic events.
- Clinical trials for these therapies must be strategically designed to include more people with T1D given the potential for insulin independence.
- Shared decision-making between people with T1D and their care team will be critical for evaluating the risks versus benefits of a beta cell replacement therapy, especially as more options become available.
Where we are now
People living with T1D depend on external insulin to manage their condition throughout their lives. Despite advancements in diabetes technology, such as continuous glucose monitors (CGMs) and automated insulin delivery (AID) systems, most people with T1D are unable to achieve blood sugar targets, rendering them at a higher risk for complications, reduced quality of life, and lower life expectancy.
It’s simple: we need to do more for the T1D community.
One promising avenue to meet these needs is through cell replacement therapy. Lantidra®, the first FDA-approved donor-derived islet replacement therapy for T1D, has proven to be safe and effective in eliminating severe hypoglycemia, providing insulin independence, and improving quality of life. However, this therapy is limited to people with severe hypoglycemia and requires immunosuppression to prevent rejection of the transplanted cells, which can have side effects. Even more, these donor-derived islets are in limited supply.
Alternative beta cell replacement therapies are emerging—and we can no longer limit their development to people experiencing severe hypoglycemia.
As stated in the publication:
Given the proven benefits of islet transplantation extending far beyond the amelioration of severe hypoglycemia that has been documented and the understanding of the risk profile gained over the past 20 years, consideration must be given to broadening the application for islet beta cell replacement.
Developing cell therapy strategies to meet unmet needs of the T1D population
Breakthrough T1D’s vision for the T1D community includes beta cell replacement therapies with no immunosuppression that are available to everyone who wants them. We are committed to making this a reality through our Project ACT (Accelerate Cell Therapies) initiative, which will accelerate the development of these therapies to achieve our vision as quickly as possible.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
We are entering an exciting era of beta cell replacement. Emerging therapies are addressing challenges such as cell source and scalability, resulting in the development of islets derived from sources other than donors, including manufactured islets and porcine islets. Up-and-coming therapies are also testing different transplantation sites, methods of delivery, and cell protection strategies to prevent immune rejection with the fewest side effects possible (and ideally no immunosuppression). Learn more about what scientists are doing to optimize beta cell replacement therapies.
Breakthrough T1D’s continuous support of many of these therapies, such as Vertex’s manufactured islet therapy zimislecel, has been critical to accelerating them through the clinical pipeline. Explore emerging beta cell replacement therapies in clinical trials now—and see how Breakthrough T1D’s commitment to these therapies helped make this possible.
When successful, the advent of new, safe, scalable, and effective beta cell replacement therapies will provide the T1D community with options. As these therapies are moving their way through the pipeline, we need to ensure they are being studied in a broader T1D population who stand to benefit.
So, how do we make this happen?
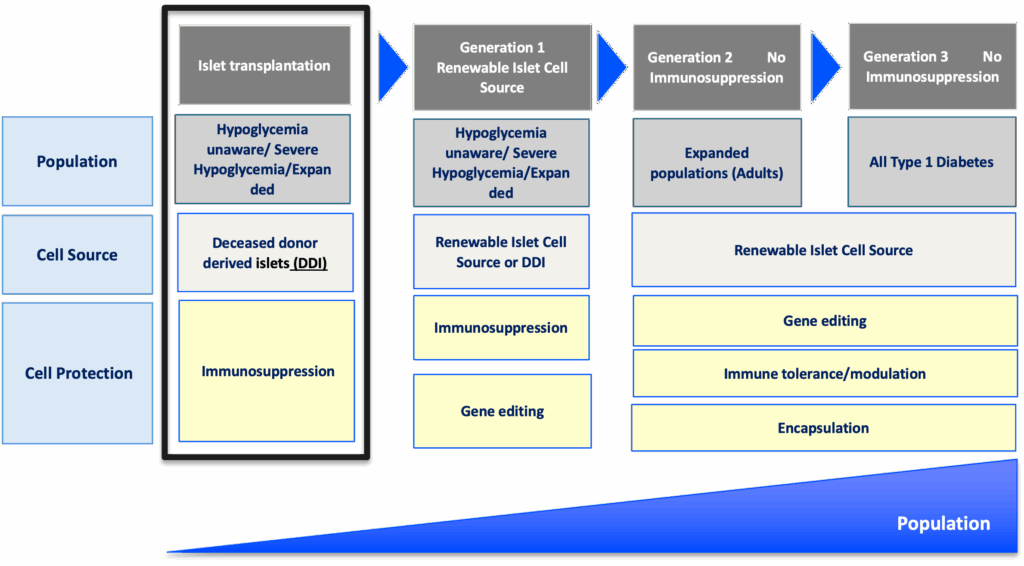
The roadmap for emerging beta cell replacement therapies
The goal
Accelerate availability of emerging next-generation beta cell replacement therapies for every person with T1D who wants them by designing clinical trials that speed their development, regulatory approval, access, and adoption.
Expanding the T1D population eligible for beta cell transplantation
Current trials testing beta cell therapies necessitating immunosuppression require participants to have elevated HbA1c levels and recurrent severe hypoglycemic events. This limits the pool of participants to people who meet the requirements and are most likely to benefit, given the side effects associated with chronic, broad immunosuppressants.
Clinical trials for emerging beta cell replacement therapies should broaden eligibility criteria so more people with T1D can participate—and experience the potential benefits. When designing new clinical trials, sponsors and regulators should consider including a broader range of HbA1c levels, clinically important or serious hypoglycemic events, and other complications.
Studies have found that the T1D community is generally open to beta cell replacement therapies as a potential solution to T1D, and people are willing to accept the associated risk versus benefit considerations for the possibility of becoming insulin independent. A Breakthrough T1D assessment also found that physicians are interested in recommending beta cell replacement therapies to people with T1D—especially if they don’t require immunosuppression.
“Clinical trials to support the development of islet cell replacement therapies need to evolve to include a broader representation of people living with T1D who could benefit from these novel therapies. This includes expanding the outcomes used to assess the benefits of cell replacement that reflect how people with T1D feel and function.”
Marjana Marinac, Pharm.D., Associate Vice President of Regulatory Affairs

Placing people with T1D at the center of clinical trial design
The outcomes used to assess the effectiveness of cell therapies currently in clinical trials, including those involving deceased donor islets, are acceptable for emerging beta cell replacement therapies. These include on-target HbA1c levels, absence of severe hypoglycemia, significant reduction or elimination of external insulin, and restoration of the body’s insulin production as measured by C-peptide.
Other endpoints that should be considered include CGM metric targets like time-in-range in addition to person-reported outcomes. Understanding how a beta cell therapy may affect a person’s health-related quality of life—such as diabetes distress, fear of hypoglycemia, or social and family dynamics—will be critically important for calculating the risk to benefit ratio of these therapies.
Read more about why person-reported outcomes are important for cell therapies.

“There are still significant unmet needs in the T1D community. Breakthrough T1D’s roadmap is supported by the assessment of clinically meaningful outcomes and driving research toward solutions that address key factors such as cell sources and protections strategies that will broaden the people with T1D who could benefit from emerging cell replacement therapies.”
Esther Latres, Ph.D., Vice President of Research
Innovative trial designs to accelerate development of cell therapies
Clinical trials for beta cell replacement therapies are generally based on a single-arm, open-label design—meaning there is no placebo group and both participants and researchers know which therapy is being administered. While this design can work for emerging beta cell therapies, single trials with multiple arms testing alternative transplant sites, immune protection strategies, or other methods have the potential to speed up the pace of development.
Similarly, adaptive trial designs use mid-trial interim analyses of study data to inform the remainder of the trial. This helps researchers learn what’s working (or not working) and adjust the design accordingly, with guidance from regulatory agencies, so the rest of the trial is a focused and efficient use of time and resources. Potential interim changes to trial design include reducing the number of participants required, eliminating doses, recruiting people who are most likely to benefit, or stopping the trial outright due to clear success or failure.
By applying guidance in therapeutic development and innovative trial designs to emerging beta cell replacement therapies, we can move early-stage trials along faster, thereby allowing regulators to make decisions sooner. To support quicker trials and reduce the possibility for delays, researchers, developers, and regulators around the world need to work together to achieve convergence on trial populations, endpoints, and innovative designs that will meet regional requirements.
Learning from past successes
People with T1D continue to live with unmet needs with still significant risk for long-term complications, and they need more therapeutic options. Right now, most clinical trials for beta cell replacement therapies requiring immunosuppression are limited to a small portion of the T1D population. This needs to change—especially given the potential for insulin independence. The T1D community must be put first when making decisions about beta cell replacement therapies, and Breakthrough T1D is making sure that this happens.
Adjusting how we approach clinical trials for emerging beta cell replacement therapies will be critical for ensuring we accelerate the research, development, regulatory approval, access and adoption of these novel therapies. Breakthrough T1D successfully accomplished this for AID systems—and we are confident that following a similar roadmap for cell therapies will get us to the finish line, faster.
The journey of AID systems
Learn more about the critical role of Breakthrough T1D in driving AID systems forward, recounted by Breakthrough T1D volunteer Doug Lowenstein.
Curative therapies for T1D are in reach. This roadmap, in conjunction with our Project ACT initiative, is key to bringing beta cell replacement therapies to every person with T1D who wants them.
To make this a reality, everyone needs to work together. As stated in the publication, “This requires a comprehensive strategy and a coordinated collaboration across stakeholders in every field relevant to islet cell replacement.” This roadmap is a guide for moving toward our common goal of a cure for T1D as soon as possible.
Breakthrough T1D will continue to lead the way until the T1D community can choose the beta cell replacement therapy that works best for them, regardless of blood glucose management or hypoglycemia status. Everyone deserves the chance to benefit.

Sana Biotechnology’s hypoimmune (HIP) donor-derived islets are genetically engineered to avoid immune destruction without the need for immunosuppressants.
Earlier this year, we reported on an update from the first person with type 1 diabetes (T1D) to receive a transplant of these cells, which showed that they were making insulin—without immunosuppression—after four weeks. Then, at the American Diabetes Association (ADA) 85th Scientific Sessions, we heard firsthand from Per-Ola Carlsson, M.D., Ph.D., the Principal Investigator at Uppsala University conducting the study, who provided more exciting data to a packed audience.
Now, these data have just been published in the prestigious New England Journal of Medicine in an article titled “Survival of Transplanted Allogenic Beta Cells with No Immunosuppression.” This study was supported by the Helmsley Charitable Trust, a long-time partner of Breakthrough T1D.
Read on to learn more.
Surviving and thriving
In this first-in-human study, the recipient of Sana’s HIP islets had no detectable insulin production at the time of the transplant (as measured by C-peptide). They received a small dose of islet cells—less than the eventual therapeutic dose—implanted into the forearm to evaluate safety.

C-peptide
C-peptide is an easily measurable biomarker of insulin production and islet function.
After 12 weeks, researchers found that the transplanted islets were successfully avoiding destruction by the immune system—meaning they are surviving AND making insulin!
The recipient had decreased HbA1c levels and stable insulin production between seven days and 12 weeks post-transplant, although, as expected with the small dose of cells used in this pilot study, they still required external insulin therapy. They experienced some non-serious side effects possibly related to the surgical transplant procedure, but not the islet cells themselves. Overall, the recipient is doing well and in good health—an exciting step for the T1D community.
Why this matters
To put it plainly: this is a huge step toward cures for T1D.
Immunosuppression is a major barrier to cell therapies because of the side effects associated with them. This is the first proof-of-concept evidence that we can genetically engineer islets to avoid immune destruction—eliminating the need for chronic, broad immunosuppressants. This means that more people may have the opportunity to benefit.
Importantly, the person in this study received only a fraction of the islet cells that would eventually be used therapeutically. Researchers are just beginning to understand the full potential of engineered HIP islets. Because more islet cells means more insulin production, there’s the possibility of insulin therapy independence if higher doses of HIP islets are implanted—something that will be addressed in future studies.
Thanks to this study, we’ve gained more evidence that forearm muscles may be a possible minimally invasive site for islet transplantation. This transplant site also provides researchers with the advantage of visualizing the islets—and making sure they’re alive—using magnetic resonance imaging (MRI), which isn’t possible with the current transplant site in the hepatic portal vein.
Excitingly, Sana is working to apply this technology to manufactured islets, opening the doors to producing HIP islets at large scale. They expect to begin phase 1 clinical trials for this next-generation HIP islet therapy as early as 2026.
Cell therapies without immunosuppression are a Breakthrough T1D priority
Cell replacement therapies—especially those that do not require immunosuppression—are a priority of Breakthrough T1D’s Cell Therapies Program and the goal of our Project ACT (Accelerate Cell Therapies) initiative.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To accelerate islet replacement therapies faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
To accelerate the development of cell therapies that do not require immunosuppression, The T1D Fund: A Breakthrough T1D Venture invested in Sana to help advance their HIP technology platform. Breakthrough T1D continues to support creative and promising approaches that may eliminate the need for immunosuppression with cell replacement therapies, and we are hopeful and excited about what the future may hold.
This wouldn’t have been possible without supporters like you. Thank you for all that you do to drive Breakthrough T1D’s mission, and together we celebrate each step towards a world without T1D.