
There is always breaking, inspiring news in the world of type 1 diabetes (T1D) research—and that news was front and center at the 2025 European Association for the Study of Diabetes (EASD) Meeting.
This annual event is one of the largest diabetes conferences in the world, and Breakthrough T1D staff, partners, funded researchers, and more were there to participate, share new data, and stay current on the state of T1D and the path to cures.
At EASD 2025, we heard the best and the brightest in T1D present on every area of our Mission priorities, from advances in cell therapies to building consensus around general population screening for T1D.
Check out the highlights below!
Long-awaited updates on two disease-modifying therapies: ATG and verapamil
Excitingly, the results from the MELD-ATG and Ver-A-T1D clinical trials were presented at EASD, co-chaired by our very own Sanjoy Dutta, Ph.D., Chief Scientific Officer at Breakthrough T1D. Each of these studies were made possible through a collaboration between INNODIA, Breakthrough T1D, the Helmsley Charitable Trust, industry, the European Commission’s Innovative Medicines/Health Initiative (IMI/IHI), and investigators around the world. Each of these studies incorporated the INNODIA master protocol, an effort to align on clinical trial design and evaluation for investigational therapies for newly diagnosed T1D.
The phase 2 MELD-ATG study, presented by Chantal Mathieu, M.D., Ph.D., investigated whether a minimal low dose of anti-thymocyte globulin (ATG) could preserve beta cells in children, adolescents, and young adults (5-25 years old) newly diagnosed with clinical stage 3 T1D. ATG works by blocking the immune cells that destroy beta cells. Here’s a summary of the recently published data:
- Using an adaptive clinical trial design, the study examined multiple doses of ATG at once, eventually moving forward with 0.5 mg/kg and 2.5 mg/kg in comparison to placebo.
- The minimal effective low dose for ATG is 0.5 mg/kg. This dose had fewer side effects compared to the higher doses, and it was generally well-tolerated.
- Participants who received low dose ATG demonstrated clinically significant higher C-peptide levels during the treatment period compared to placebo. This was accompanied by lower HbA1c levels.
The phase 2 Ver-A-T1D study, presented by Thomas Pieber, M.D., investigated whether verapamil could preserve beta cells in adults (18-44 years old) with newly diagnosed stage 3 T1D. Verapamil is a blood pressure medication that may also reduce beta cell stress, and initial clinical studies suggest that it may protect beta cells in newly diagnosed T1D. The key findings presented are:
- At 12 months, there was no statistically meaningful difference in C-peptide levels between those who were treated with verapamil versus placebo. This was accompanied by no differences in insulin dose or continuous glucose monitor (CGM) metrics. There was a trend toward better C-peptide preservation in the treatment arm compared to placebo but missed statistical significance marginally.
- Verapamil was generally safe and well-tolerated, with no unexpected adverse events but some reported mild and reversible cardiac disorders.
- The study investigators concluded that future studies testing verapamil in T1D require a larger population and longer duration—and that verapamil may be a candidate to combine with immunomodulatory disease-modifying therapies.
- The Ver-A-Long extension study will follow people who continued with verapamil treatment after this trial ended.
Key takeaways
The horizon is bright for ATG—investigators have now identified the lowest dose needed to preserve beta cells in newly diagnosed children and young adults with T1D, with the fewest side effects possible. BT1D and the T1D Fund are supporting the development of a next generation ATG with a manageable/lower side effect profile compared to the medication used in the current trial. On the other hand, the future is open for verapamil—more studies are needed to determine if it can delay beta cell loss and T1D progression. A biotechnology company, TiXiMed, is developing a next-generation verapamil (TX-100), with a more favorable pharmacological profile.
Emerging consensus for screening the general population for T1D
Breakthrough T1D spotlight: Early detection and screening
Breakthrough T1D’s Vice President of Medical Affairs, Anastasia Albanese-O’Neill, Ph.D., APRN, CDCES, spearheaded an effort to assemble a working group of nearly 30 experts to establish a consensus on T1D screening guidance. This session—which Dr. Albanese-O’Neill chaired—included presentations by some of these experts, who gave the audience a sneak peek on how to integrate population-level screening for T1D into clinical practice.
In this session, Marian Rewers, M.D., Ph.D., Anette Ziegler, M.D.,and Chantal Mathieu, M.D., Ph.D., provided an overview of soon-to-be-published consensus screening guidance for the general population. These presentations highlighted an urgent need for population-level screening given the rising global incidence of T1D and the known benefits of early detection, including time to prepare and potentially delay disease progression.

T1D autoantibodies
Autoantibodies are proteins that may signal that the body’s immune system is attacking insulin-producing cells in the pancreas, and they can easily be measured by a blood test. If a person has two or more persistent autoantibodies, it’s very likely they’ll develop T1D.
The presenters discussed benefits, harms, and methods of T1D screening; who should be screened and how often; and how to effectively communicate screening results. Some notable takeaways:
- Early detection of T1D—prior to clinical onset and at the time of diagnosis or later—has enormous benefits, including prevention of diabetic ketoacidosis, opportunities to delay progression with approved therapies or experimental therapies in clinical trials, early control of hyperglycemia that can reduce the risk of complications, and more.
- Providing support during the screening process will be important, such as mitigating pain associated with obtaining blood samples or reducing anxiety.
- Every initial screening result must be confirmed with another test before diagnosis.
- Infrastructure and policy must be in place before population-level screening can be implemented into clinical practice, including established processes for confirming positive test results, monitoring people with diagnosed early-stage T1D, and referring people to specialists.
- General population screening is suggested starting at 2-4 years old, and again at 6-8 years old and 10-15 years old if negative for autoantibodies.
- Healthcare providers (HCPs) need to be knowledgeable on key aspects of the screening process—and be able to effectively communicate what screening results mean.


Breakthrough T1D spotlight: Pediatric screening for T1D
To increase pediatric T1D screening in the UK, Breakthrough T1D is co-funding the ELSA (EarLy Surveillance of Autoimmune disease) study with Diabetes UK. At EASD, Parth Narendran, FRCP, Ph.D., presented how the data from this study was used to assess the acceptability and feasibility of general population screening for presymptomatic T1D in children who are 3-13 years old. The research team found that an initial finger-prick dried blood spot test—which can be performed at home or school and mailed to the team—was acceptable as an initial autoantibody screen, followed by a confirmatory blood test. Most families found out about this screening opportunity through social media, school, or their general practitioner—and none regretted participating, regardless of the outcome. This study will continue with an expanded population of children who are 2-17 years old.
Key takeaways
Consensus guidance from T1D experts suggests that population-level screening for T1D, starting at a young age, will be critically important for detecting T1D early so families have ample time to prepare and potentially delay disease progression. This consensus guidance will soon be published in a peer-reviewed journal, with the goal of integrating T1D screening into clinical practice. Breakthrough T1D is committed to expanding T1D early detection on a global scale.
Cell therapies are coming
Cell Therapies replace destroyed beta cells with external cells that make insulin and protect them so that they can function for a very long time. It’s one of our primary strategies to achieve cures for T1D—as exemplified by Project Accelerate Cell Therapies.
Our goal is to grow these cells in a laboratory and discover a way to keep them safe inside the body without the need for chronic immunosuppression. EASD provided several reminders that these therapies are not science fiction—they’re working in clinical trials, and we must continue to push these first-generation therapies—and future generations—across the finish line.
Vertex and zimislecel
Attendees at EASD were treated to another presentation demonstrating the efficacy of Vertex’s zimislecel, a manufactured islet cell replacement therapy. Michiel Nijhoff, M.D., presented some highlights from the FORWARD study, all of which were previously presented at other diabetes conferences and published in the New England Journal of Medicine [subscription required] this summer.
This chart does an excellent job illustrating the drastic improvement in glucose control in the year following transplantation of the cells. As does this stat: the 12 individuals in the first phase of the study went from 55.5% time in range and 36.1 units of insulin per day at the start of the study to 95% time in range and 0 units of insulin per day after one year.
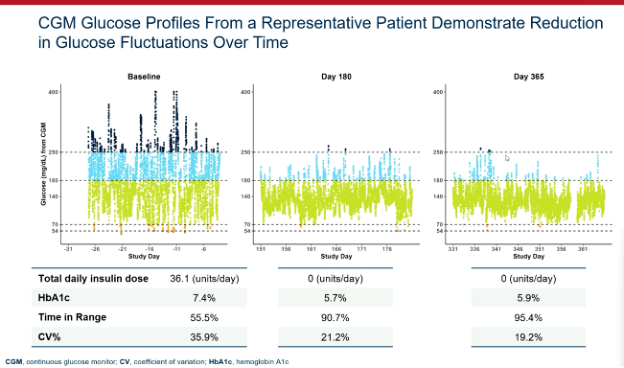
Breakthrough T1D’s support for Doug Melton, Ph.D.—whose proprietary lab-created beta cells are now being advanced by Vertex—goes back decades, both via research grants and an investment from the T1D Fund: A Breakthrough T1D Venture. We can’t wait to see more data when the Phase 1/2/3 trial is completed in the coming months.
Multiple strategies moving forward
Several other investigators shared data on their approaches to bringing scalable cell therapies to people with T1D. This includes:
- Matthis Hebrok, Ph.D., a longtime Breakthrough T1D collaborator, showed his research on hypoimmune, gene-edited islets. These cells are genetically engineered to avoid the immune system.
- Maria Nostro, Ph.D., another Breakthrough T1D-funded researcher, is using a different protection strategy: macrophages. Macrophages are a type of white blood cell that we have learned are present in the developing pancreas. The idea here is that we can implant the macrophages alongside the beta cells during islet transplant and it will help protect and vascularize the cells. This research is still pre-clinical but represents another “shot on goal” to keep these cells safe.
- Orizuru Therapeutics (a T1D Fund Company) presented on OZTx-410, an investigational cell therapy composed of human manufactured islets derived from adult cells that were reprogrammed to precursor cells and eventually islets. This is one of the first cell therapies for T1D derived from reprogrammed adult cells, and this therapy is suitable for manufacturing at large scale. Earlier this year, these cells were implanted into a person with T1D as multiple “sheets” in the abdominal wall. Orizuru expects to share results in the coming months.
- Allarta, a company with Breakthrough T1D funding, shared pre-clinical data on their hydrogel, which allows islet transplants without the use of immunosuppression. Their product is showing efficacy in animal models, and they are progressing towards human clinical studies.
Breakthrough T1D spotlight: Personal journeys and emerging T1D therapies
Breakthrough T1D’s Chief of Global Medical Affairs, Thomas Danne, M.D., Ph.D., hosted an interactive workshop for participants to better understand the personal journeys of people with T1D through videos from T1D community members and their families. Dr. Danne—along with other Breakthrough T1D staff and in conjunction with Eelco J.P. de Koning, M.D., Ph.D., and Valera Sordi, Ph.D.—fielded questions from the audience and covered topics spanning immunosuppression, how islets are derived from donor pancreases, T1D screening, and more.


Key takeaways
Cell therapies are real and advancing, with promising results in clinical trials, including dramatic improvements in glucose control. Also moving forward are ways to keep them safe and happy without the use of immunosuppressants.
Continuous ketone monitoring is coming – for good reason!
Diabetic ketoacidosis (DKA) is a serious and, unfortunately, too-common complication of life with T1D. This dangerous condition happens when the body does not have enough insulin. DKA is life-threatening and costly.
Breakthrough T1D-funded research Jennifer Sherr, M.D., Ph.D., presented this, outlining that this is a problem and that there is room for improvement in identifying DKA and agreeing on the interventions. We need better mitigation strategies, and if we get them, people will do better.

Breakthrough T1D Medical Affairs: CKM
Breakthrough T1D has prioritized the development of continuous ketone monitors, which we believe can be lifesaving. Our Medical Affairs team is working to build consensus around these and how they can be useful in the clinic and in the lives of people with T1D.
Key takeaways
Peter Bergstal gave a great summary, where he noted the CKM can be incredibly useful if we hit a few key checkpoints. These include developing an accurate CKM, receiving regulatory approval, finding the people most likely to benefit, and they’re covered by payers.
T1D Index 3.0 is here
The T1D Index—a computational tool launched in 2022 that measures the human, public health, and economic impact of T1D around the world—was developed by Breakthrough T1D in collaboration with Life for a Child, the International Diabetes Foundation (IDF), and the International Society for Pediatric and Adolescent Diabetes (ISPAD). Renza Scibilia, part of the Global Responsibility team at Breakthrough T1D, presented about exciting new upgrades for the T1D Index version 3.0. Some important updates:
- The newest version of the T1D Index has been updated with new published and unpublished data to ensure it remains the most comprehensive picture of T1D in the world.
- The new-and-improved dashboard allows for easier usability for researchers, healthcare professionals, and T1D community members to explore the data and computationally model the effects of care and diagnosis.
- The next version of the T1D Index in the works will further break down T1D statistics, focusing on regions and states within countries.
- The T1D Index is critically important for global advocacy and collaboration efforts by providing evidentiary data that support initiatives to change the trajectory of T1D.
9.5 million
The number of estimated people living with T1D across the globe.
1.5 million
The number of people at all ages living with T1D in the United States—the country with the highest prevalence of T1D.
Key takeaways
The T1D Index 3.0 is the most updated, accurate version yet, offering the best possible global coverage of T1D statistics and an enhanced dashboard to simulate changes in T1D incidence and prevalence.
The path to fully closed-loop automated insulin delivery systems
Hybrid closed-loop automated insulin delivery (AID) systems—a continuous glucose monitor (CGM) paired with an insulin pump, requiring mealtime and physical activity announcements—are estimated to be used by hundreds of thousands of people today. While these devices have been transformative for the T1D community, user input is still a daily burden, and fully closed-loop systems aren’t widely available for T1D yet. In this session, Moshe Phillip, M.D., Katrien Benhalima, M.D., Ph.D., and Charlotte Boughton, M.D., Ph.D., explored the benefits of currently available AID systems and fully closed-loop AID systems on the horizon.
- Based on clinical trial data and real-world evidence, hybrid closed-loop systems improve time-in-range (TIR) and reduce HbA1c levels without increasing the risk of hypoglycemia. This translates to better outcomes and quality of life for people with T1D.
- Different AID systems will work better for different people, depending on preferences for blood glucose management. Because there have been limited head-to-head trials comparing two or more AID systems, there is no evidence that any one system is definitively better than another.
- Right now, only one AID system is licensed for use in pregnancy (CamAPS FX). More research is needed to determine safety and efficacy of additional AID systems for use during pregnancy, and real-world evidence of women who become pregnant while using different AID systems may provide some clues.
- A clinical trial for the CamAPS HX fully closed-loop AID system in the U.K. increased TIR by 50% compared to 36% in people using standard pump therapy with a CGM—amounting to three additional hours each day of blood glucose in target range. Participants reported improved mood and sleep, less stress, and reduced diabetes burden. Similar improvements in blood glucose and daily life were reported in adolescents.
- Next-generation AID systems under investigation include two or more hormones for optimal glycemic control, artificial intelligence, and “digital twin” simulation tools.
Key takeaways
Fully closed-loop AID systems are coming, but they’re not here yet. Initial clinical studies using fully closed-loop systems in T1D demonstrate significant improvements in blood glucose control and quality of life. In the meantime, hybrid closed-loop systems offer substantial benefits for people with T1D—and there are many commercially available systems to choose from based on individual preferences.
Updates on heart and eye complications
Reducing T1D complications remains a major research focus area around the globe. Robert Humphrey presented on the LENS trial, which found that fenofibrate—a generic and affordable cholesterol-lowering medication—can reduce the progression of diabetic retinopathy in people with T1D and type 2 diabetes. A follow-up study found that fenofibrate is a cost-effective option for diabetic retinopathy in the U.K., especially for T1D. In conjunction with these studies, the recruiting Breakthrough T1D-funded Protocol AF trial is further investigating fenofibrate in preventing progression of diabetic retinopathy in people with T1D.
Rebecka Bergdal presented on a study examining the relationship between cumulative exposure to glycemia or lipids and heart failure, one of the leading causes of cardiovascular mortality in T1D. The study found that both cumulative glycemic exposure (defined as time spent with HbA1c levels > 7%) and cumulative lipid exposure (LDL, triglycerides, and cholesterol) are independently associated with increased risk for heart failure. The publication highlighting these findings includes a call to action for healthcare providers to help people with T1D minimize high blood sugar and lipid exposure, and support and encourage the best diabetes management possible to reduce risk for heart complications.
Key takeaways
Exciting clinical trials to prevent worsening of diabetic retinopathy in people with T1D are ongoing. To reduce the risk of heart complications, including heart failure, people with T1D and healthcare providers should aim for minimal exposure to high blood sugar and lipids, and more work is needed to find solutions for cardiovascular disease in T1D.

“We saw significant progress in each area of our Mission at EASD,” said Sanjoy Dutta, Ph.D., Chief Scientific Officer. “The MELD-ATG results are a noteworthy step forward for disease-modifying therapies; the screening consensus is a much-needed stake in ground around general population screening; we are moving closer to urgently needed adjunctive therapies; and cell therapies continue to perform in clinical and pre-clinical studies. It’s an incredibly exciting time in T1D and we will continue to support this work—and more—to bring cures to our community.”
The next big T1D meeting is ISPAD 2025, which will take place November 5-8 in Montreal, Canada. Stay tuned for more news!
This article was written by Sandy Vogt and Brian Herrick.