Closing the gap between access to and adoption of type 1 diabetes (T1D) therapies is a mission priority for Breakthrough T1D.
Our newly created Medical Affairs unit provides education and resources for the entire T1D community, from clinicians to people living with the condition, so that everyone impacted by T1D is empowered to make informed decisions about healthcare and emerging therapies.
Chris Dunn represents our efforts in action.

Chris is a parent to four children, two of whom live with T1D. Knowing the increased risk of T1D in immediate family members, Chris and her children without T1D participated in screening. In what she could only describe as “surreal,” Chris’ screening results revealed the presence of multiple T1D antibodies; further testing indicated she was in stage 2.
For decades, Breakthrough T1D funded discovery research, supported clinical development, and worked with regulators to bring Tzield—the first disease-modifying therapy for type 1 diabetes—to market. Tzield is available to people in stage 2 type 1 diabetes—like Chris—and can delay progression to stage 3, or clinical onset of T1D, for up to three years. This means potentially years without blood-sugar monitoring, insulin administration, and the fear of short- and long-term complications.
Watch Chris’s story now:
Part 1: Screening and Early Detection
Part 2: Early Detection and Disease-Modifying Therapies
Advocacy is critical to advancing breakthroughs for the entire type 1 diabetes (T1D) community. Through nonpartisan and grassroots support, we advance treatments, influence policy, and improve global access to care.
Breakthrough T1D advocates strengthen the voice of the T1D community. They engage with their Members of Congress to share their personal connections to T1D, articulating the emotional, financial, and physical costs of the condition, and the importance of Federal funding for T1D research.
Our advocates have been the driving force behind renewal of the Special Diabetes Program (SDP), a critical government program that provides $160 million annually to the U.S. National Institutes of Health (NIH) exclusively for T1D research.
Together, our advocates change how people see T1D.
Share your story with us!
If you’re inspired by the stories above, share your story too. What do you wish people understood about life with T1D? How have T1D breakthroughs changed your life? Post on social media, tag @BreakthroughT1DHQ, and use the hashtag #SeeT1D.
What is the pipeline?

Every new medical device, therapy, treatment, and drug—including those for type 1 diabetes (T1D)—goes through the drug development pipeline. Getting a new therapy or device from the earliest stages of research eventually into the hands of people with T1D is a complicated process. Science takes time (from years to decades!), money (from hundreds of millions to billions!), and brainpower (lots and lots of brilliant scientists, doctors, researchers, and more)—and lots of it.

That’s what drives us and our work in the pipeline: people with T1D doing better.
This process is complex, to say the least. There are safety checks at every step of the way. Data are scrutinized, and preclinical and clinical testing must meet ethical standards. In fact, many new drugs and devices don’t make it very far in the pipeline—and those that do take a long time to get there.
Breakthrough T1D is unique in that we work across the entire pipeline—from start to finish—for every promising therapy or device that we invest in. Between Research, Advocacy, and Medical Affairs, we work at every single step to accelerate progress and get new treatments to people with T1D faster than ever.
That’s our value proposition. That’s what makes supporting us different than supporting an individual researcher, or a university, or a company.
Let’s dive a little deeper into the pipeline—and how we’re turbocharging it.
Time and money
Total money spent in the diabetes space in FY24:
$146 million
By Breakthrough T1D
$521 million
Total T1D research support, including by Breakthrough T1D
$160 million
By the Special Diabetes Program
$412.9 billion
In healthcare
Total investments in new drugs and devices:
12 to 15 years
Estimated time it takes a new drug to get to the clinic
$1 billion
Estimated total cost to bring a new drug from discovery research to the clinic
3 to 7 years
Estimated time it takes a new medical device to get to the clinic
$522 million
Estimated total cost to bring a new medical device from discovery research to the clinic
The pipeline in action: Automated insulin delivery systems

Automated insulin delivery systems vs. artificial pancreas systems
At the very beginning, these devices were called artificial pancreas (AP) systems. Today, they are called automated insulin delivery (AID) systems. We’ll be referring to them as AID systems going forward.
The process at a glance:
$171 million
The amount of money Breakthrough T1D spent on AID system-related research from 2005 to 2024
12 years
The amount of time it took for next-generation AID systems to go from discovery research to clinical adoption after Breakthrough T1D became involved
Hundreds of millions of dollars
Total investment in AID systems from all stakeholders
60+ years
The amount of time it took for AID systems to become a reality, starting with the first experimental AP system in 1964
A deeper dive into the process:

Breakthrough T1D x AID systems
Breakthrough T1D has played a significant role in the evolution of AID systems. Doug Lowenstein, a long-time Breakthrough T1D volunteer and supporter, detailed the history of AID systems from the very beginning, and how we accelerated progress at every step of the way.
Discovery research
It’s the early 2000s. I’m a new AID system, but I don’t exist yet—I’m just an idea. I live in the minds of some scientists and researchers who think that I have the potential to one day become a reality. But, turning an idea into reality can be hard. It’ll take a lot of people, time, and effort for me to exist—but it’s not impossible. The question is: how? Answer: it starts with funding.
2005: Breakthrough T1D launches the Artificial Pancreas Project
Breakthrough T1D dedicates funds to scientists and researchers who have compelling ideas to turn AID systems into a reality, marking the start of the decades-long Artificial Pancreas Project (APP). These investments were key in jump-starting research into all the components needed to make an AID system work.
“[The goal was] to keep people alive and healthy until we find a cure. We were losing people…overnight of low blood sugars. If we could automatically dose insulin and have everybody go to sleep and all wake up, that was an incredible victory.” -John Brady, member of Breakthrough T1D Executive Committee in the early 2000s, former Chair of Breakthrough T1D’s International Board of Directors, and father of a son with T1D
Translational research
It’s 2008. Scientists are making progress on the three key parts that need to work in harmony for me to become a reality: a continuous glucose monitor (CGM), an insulin pump, and an algorithm that lets them talk to each other. But, like most new ideas, before I’m allowed to be tested on humans, I need to be tested in animal models first. Lucky for me, I skipped this step because someone made a tool that could simulate how I would act if I was attached to a human being.
2008: Breakthrough T1D-funded scientists create simulator tool to bypass animal studies
The simulator tool, developed from the initial Breakthrough T1D grants, allowed scientists to model how an AID system would respond to real-life scenarios, like eating a certain amount of carbs or exercising, and how this would translate to blood sugar outcomes like time in range or hypoglycemic events. The FDA accepting the use of this tool was a major win—without animal studies, AID system development was accelerated by years.
“The simulator saved at least five years of animal studies because we didn’t require an algorithm to be tested in an animal model to be deemed safe and effective before going into human studies. That entire chunk was eliminated.” -Sanjoy Dutta, Ph.D., Breakthrough T1D Chief Scientific Officer
Clinical trials
It’s 2012. Scientists have come up with different versions of my components that are ready to be tested in humans. For the next four years, I’ll be attached to people with T1D around the world who courageously volunteered themselves to test if I actually work. Turns out, I do a pretty good job at managing blood sugar—and I’m safe!
2012 to 2016: AID systems make headway in clinical trials
While clinical trials for AID systems started in earnest in 2008, they kicked into high gear in 2012, when investigators started conducting trials using different AID components and algorithms in real-life settings. Progress moved quickly, and results from numerous studies supported the idea that AID systems are both safe and effective. Breakthrough T1D funding—along with the Special Diabetes Program—helped move these trials forward.
“Getting involved in AID system trials to me was my chance to pay it forward for somebody else. I have lived 45 years with T1D…I think there’s something comforting in knowing that my body was used for something that not only had the potential to make me healthier, but really was for other people. I do think there’s something to be said for doing good, feeling good, and this is what it felt like being part of trials.” – Alecia Wesner
Regulatory approval
Flashback to 2009. While scientists are busy figuring me out, people at Breakthrough T1D are already thinking about and planning for my future—and how to work with the decision-makers at the FDA who will ultimately decide my fate. The FDA has also given their two cents about the best ways researchers can test me in clinical trials to get the data needed for me to get approved. Fast forward to 2016, it finally happens: I get FDA approval!
2009 to 2016: From APP roadmap to FDA approval
In 2009, Breakthrough T1D published the AP Roadmap, detailing what the future of AID systems will look like—and how we plan to get there. Two years later, we worked with the FDA on clinical trial design for AID systems so that there was a clear path to approval. After human clinical trials, the FDA approved Medtronic’s hybrid closed-loop MiniMed 670G—officially marking the first AID system to be available in the U.S.
“I do give [Breakthrough T1D] credit for pushing, for saying there’s a real need for this.” -Jeff Shuren, M.D., Head of the Center for Devices and Radiological Health at the FDA at the time
Access
Flashback to 2008. At this time, people were still unsure if one of my main components—a CGM—was a reliable way to measure blood sugar. This all changed when a first-of-its-kind clinical trial showed that CGMs are better than finger pricks and glucose meters, and they were covered by insurance shortly after. This decision paved the way for my future: after my approval in 2016, insurers began offering to cover me.
2008 to 2017: Insurance coverage evolves
The landmark Breakthrough T1D-funded clinical trial in 2008 provided the data to convince private insurers to cover CGMs. Nearly a decade later, Breakthrough T1D launched its “Coverage2Control” campaign to advocate for insurance coverage of T1D treatments, therapies, and devices—including AID systems—ultimately resulting in all major private insurers offering coverage of AID systems by the end of 2017, followed by Medicare shortly after.
“Seeing the artificial pancreas go from concept to reality…is what makes Breakthrough T1D and all of the advocacy volunteers—who sent an email, made a call, signed an action alert, or met with their Member of Congress—very proud of this historic achievement and the impact that these will have on the individual lives of those with type 1 diabetes.” -Cynthia Rice, former Chief Mission Strategy Officer at Breakthrough T1D
Adoption
It’s present day. There are tons of iterations of me, and even more coming. People get to choose which version of me they like best. I’m covered by most health insurance. I’ve come a long way since I was just another thought in the minds of a few scientists who had a vision… and now I’ve come to life! Even so, not everyone has chosen to use me yet—and I will continue to evolve and grow until I can make the lives easier of as many people with T1D as possible.
2017 and on: More and more AID systems come to life
After the first hybrid closed-loop AID system was approved, the flood gates were opened. More and more systems are coming to market each year, and they keep getting better. They’re smaller, easier to use, and better at managing blood sugar. They’re covered by both government and private insurance plans. They’re an integral part of routine discussions between people with T1D and their healthcare providers, and people have options to choose which system is best for them. This is a future that was difficult to imagine two decades ago—and now it’s a reality. Even so, the work continues until AID systems are a reality for more and more people with T1D.
“What we brought to bear is resulting in a safer and easier life for hundreds of thousands, and soon millions, of people with T1D, including my son, that is going to keep them safe until something like a cure comes along,” -Jeffrey Brewer, one of the APP founders
The final stage: Cures and improved lives
It took tons of time, money, people, and effort to get us where we are today, but we’re not at the finish line yet. “The end game for AID systems,” says Breakthrough T1D CEO Aaron Kowalski, Ph.D., “is to have multiple compatible pumps, glucose sensors, and algorithms, so that patients can mix and match what they prefer.”
The end game for T1D as a whole, however, is cures. AID systems have greatly improved the lives of those with T1D—and will continue to do so now and in the future—while we continuously work toward cures that are one day available to everyone with the condition.
The path followed for AID systems is a roadmap for other therapies coming down the pipeline. Breakthrough T1D’s Project ACT is taking a page from the AP roadmap and applying it to cell therapies, so that functional cures can get to people with T1D who want them as quickly and safely as possible.

While I’m proud of my work as a scientist at Breakthrough T1D on AID systems (and my brother and I currently wear AID systems that are derived from Breakthrough T1D-supported work), more than anything else I want to take off my diabetes devices and achieve what our founders set out to do—find cures for T1D.”
Breakthrough T1D successfully took an idea and turned it into a reality—and we’ll do it again and again until T1D is a thing of the past.

Breakthrough T1D envisions a world where the burden of type 1 diabetes (T1D) no longer exists.
It’s a world where people don’t have to manage their diabetes—don’t take insulin, don’t wear devices, don’t have blood sugar highs and lows, and don’t develop complications. That’s how we define cures for type 1 diabetes, and there are three ways we can get to cures faster:
Cell therapies
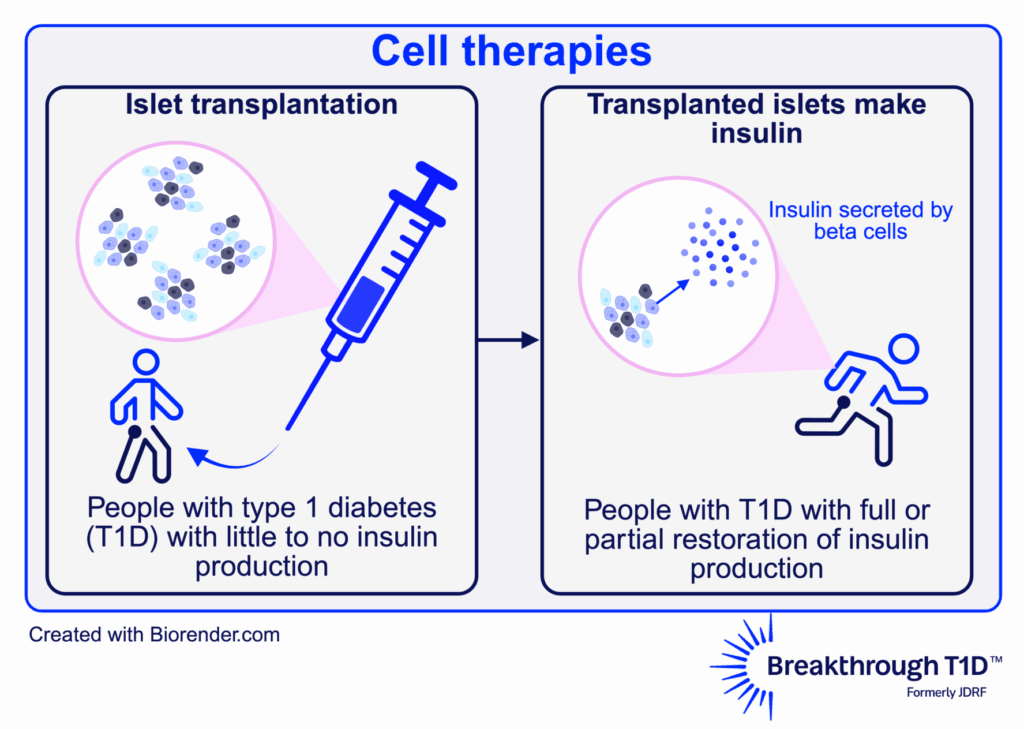
What are cell therapies?
In people with T1D, the immune system destroys the beta cells in the pancreas, which produce insulin. One known way to cure T1D is through cell therapies, which replace destroyed beta cells with external cells that make insulin and protect them so that they can function for a very long time.
What work is Breakthrough T1D doing in this area?
117
Number of preclinical cell therapies research projects we are supporting
5
Number of cell therapies clinical trials we are supporting
We are funding research to:
- Develop a renewable beta cell source so that every person with T1D who wants this treatment can get it.
- Find ways to maintain these beta cells so they stay functional in the body for years—or even decades.
- Shield these beta cells from immune attack to prevent the need for anti-rejection immunosuppressants or the recurrence of T1D.
What does the future hold for cell therapies?
Our ultimate goal is large-scale cell therapies with the fewest side effects possible, accessible by anyone with T1D who wants them. Our Project ACT initiative will get us there faster than ever. We believe that, in the future, these therapies will create a path for people with T1D to walk away from the condition for good.
Project ACT
Scientific progress takes time, resources, collaborations, and effort. To get cell replacement therapies to people with T1D faster than ever, Breakthrough T1D launched Project ACT (Accelerate Cell Therapies) to simultaneously advance research, development, regulatory policies, access, and adoption of manufactured islet therapies that do not require broad immunosuppression.
Early detection

What is early detection?
Type 1 diabetes doesn’t show up overnight (even though for many people it feels like it does). The condition develops in stages over time:

From a therapy perspective, early detection is important for identifying people who could benefit from the FDA-approved disease-modifying therapy Tzield, which can delay T1D progression for years.
From a research perspective, early detection can open doors to research opportunities, like clinical trials of other disease-modifying therapies in development.
From the clinical perspective, early detection can prevent complications at diagnosis, like diabetic ketoacidosis (DKA), and help families and individuals prepare for T1D onset.
Screening is done through a simple blood test to detect T1D autoantibodies, which signal that the immune system may be attacking the body’s insulin-producing cells.
What work is Breakthrough T1D doing in this area?
52
Number of preclinical early detection research projects we are supporting
26
Number of early detection clinical trials we are supporting
Through our Advocacy and Medical Affairs efforts, we are:
- Supporting the SCREEN for Type 1 Diabetes Act
- Leading a campaign to expand access to early detection of T1D in the United States
- Providing accredited education and resources on T1D and early detection to healthcare professionals
What does the future hold for early detection of type 1 diabetes?
We expect to improve ways to detect T1D early and prevent it from advancing by attaining a better understanding of the natural progression of T1D, which may include additional biomarkers for the condition. We will continue to expand access to early detection and find ways to improve the standard of care for early-stage T1D.
Disease-modifying therapies
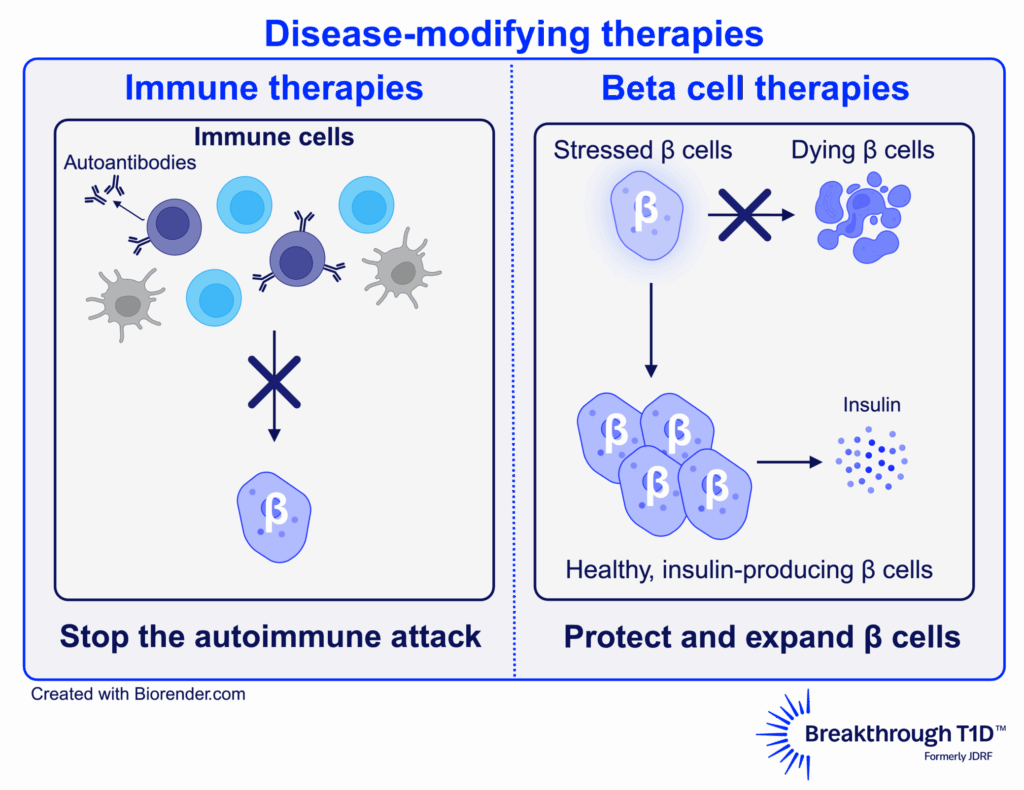
What are disease-modifying therapies?
T1D is a progression. At earlier stages, the immune system has begun attacking the insulin-producing beta cells, but people are not symptomatic yet. Some people at later stages have had T1D for decades and have lost nearly all insulin production. Disease-modifying therapies, or DMTs, are aimed at helping everyone with T1D—regardless of where they are on the disease continuum—to slow, halt, prevent, or reverse the condition.
What is Breakthrough T1D doing in this area?
138
Number of preclinical disease-modifying therapy research projects we are supporting
16
Number of disease-modifying therapies clinical trials we are supporting
We recently published research on the importance of C-peptide—a biomarker for insulin production—as a key measure of how effective a DMT is in clinical trials. Use of C-peptide will be essential to speeding up development and approval of new DMTs—getting potential cures to people with T1D faster.
What does the future hold for disease-modifying therapies?
As we see more DMT clinical trials advance to later-stage human testing, we expect to see additional DMTs submitted for regulatory approval—giving people with T1D more options.
Improving Lives
Cures for type 1 diabetes are coming down the pipeline. While we look forward to their arrival, Breakthrough T1D is improving the lives of people with T1D today.
There has never been a more exciting time for T1D research—cures for type 1 diabetes and other breakthroughs are within reach. With the support of the T1D community, we will succeed!
Imagine a world where everyone understood what it means to live with type 1 diabetes (T1D). That’s our goal every November for Diabetes Awareness Month: to create a global movement that shines a light on T1D and the resilient people who face it.
Building awareness and understanding about T1D is more important than ever. When we see T1D, we move closer to life-changing breakthroughs in research, advocacy, and support—helping people with T1D live fuller, healthier lives as we drive toward cures.
Together, our stories change how people see T1D.












Share your story with us!
If you’re inspired by the stories above, share your story too. What do you wish people understood about life with T1D? How have T1D breakthroughs changed your life? Post on social media, tag @BreakthroughT1DHQ, and use the hashtag #SeeT1D.
Oh, the holiday season is upon us! The lights, the songs, the gifts, the food…and the stress.
It can make this time of the year a little tricky for those of us with type 1 diabetes (T1D). Can you enjoy the holiday treats and festive meals? Of course! Can you surround yourself with the hustle and bustle of the season? Definitely! But managing your blood sugar will take more work, including a bit more planning ahead and a bit more adjusting.
Here are a few tips to help you celebrate the holidays to the fullest, plus a recipe for a sweet treat from a Breakthrough T1D community member!
Get moving (and keep moving) to boost insulin sensitivity
Winter is no time to start sinking into the couch cushions! Daily exercise is one of the best tools for fighting rising blood sugars and insulin resistance during the holidays.
That daily walk can significantly boost your sensitivity to insulin and support your blood sugar goals. In other words, it can help insulin work better in your body! But you’ve got to be consistent—make it a daily part of your life.
It doesn’t have to be intense. Walking twice a day for 30 minutes is an excellent place to start. If that becomes your daily exercise routine, that’s a bonus. Even putting up the holiday lights can be a good workout!
If you’re interested in adding a little bit of strength training, it can be simple. Grab a pair of 8- or 12-pound dumbbells and do a set of standing shoulder presses, bent-over rows, or squats.
If you live in a very chilly place, grab your cold weather gear and get outside. Every day.
Learn more about exercise and T1D.
Plan ahead with lighter meals before the bigger holiday meals
You know there will be a wide range of indulgent, delicious, seasonal dishes and desserts at holiday gatherings. Just because you have T1D doesn’t mean you can take part in the tasty celebration—but you can plan ahead by eating lighter for the first part of the day.
Stick to whole-food, lighter meals at breakfast and lunch to make room for your holiday dinner. For example:
- Breakfast: egg and vegetable sauté
- Lunch: apple and salted nuts
- Snack: carrots and hummus
- Dinner: the holiday party!
This will also make blood sugar management easier for the first big chunk of the day. Managing your blood sugar with T1D is hard enough—set yourself up for success during the holidays by keeping some of the day’s meals light and easy!
Our Thanksgiving holiday guide has more helpful tips to make the holiday easier, including carb counts for foods and drinks!
Know the effects of alcohol on T1D
Eggnog and champagne toasts are often part of holiday festivities. Learning how to consume alcohol carefully and safely with type 1 diabetes is important so you can enjoy yourself without risking hypoglycemia later on.
Learn more about alcohol and type 1 diabetes.
Keep stress in check
The holidays can cause a lot of anxiety. T1D also never stops, and this comes with its own challenges. Stress of any kind can impact your blood sugar, causing unexpected highs or lows.
Focus on managing your stress this time of year. Exercise, eating a balanced diet, and getting enough sleep are all great ways to reduce stress.
It’s also a good time of year to find and embrace what you are grateful for. Whether it’s books, music, food, family and friends, pets, or nature, surround yourself with what brings your soul joy!
If you find that you are having difficulty managing stress, don’t be afraid to ask for help. Visit our Mental Health Guide for more information and support.
‘Tis the Season!
Navigating the holidays with type 1 diabetes isn’t always easy, but if you approach it with thoughtful intentions, you can enjoy it all!

Charlie’s Chocolate Chip Banana Bread Muffins
Donna Lynne Champlin’s son Charlie was diagnosed with T1D in 2020, at age 9. Donna’s family used this recipe for Chocolate Chip Banana Bread Muffins to teach Charlie how to bake and, inspired by kids’ baking shows, he’s spent years perfecting it! He came up with the idea of making banana bread muffins instead of a loaf to keep portion sizes consistent and make bolusing easier.
Prep time: 10 minutes
Cook time: 25 minutes
Servings: 12 muffins
Carb Count: 1 muffin = 45g carbs
Ingredients:
- 3 – 4 ripe bananas, smashed (52g carbs = 4 bananas)
- 1/3 cup melted butter (1g carb)
- 1 cup sugar (200g carbs)
- 1 egg, beaten (1g carb)
- 1 teaspoon vanilla (1g carb)
- 1 teaspoon baking soda (0g carbs)
- Pinch of salt (0g carbs)
- 1 ½ cups of all-purpose flour (142g carbs)
- 1 cup dark chocolate chips (144g carbs)
Instructions:
Preheat the oven to 350°F (175°C). With a wooden spoon (or your hands) mash bananas and mix with melted butter in a large mixing bowl. Mix in the sugar, egg, and vanilla. Sprinkle the baking soda & salt. Mix in flour. Fold in ¾ of the chocolate chips. Pour batter into a 12-cup muffin pan. Sprinkle the rest of chips on top of the muffins. Bake for 25 minutes.
*To make the bread loaf instead of muffins, just use a loaf pan and bake for an hour.
Paola Reynoso is a Development Assistant for Breakthrough T1D’s Northern California chapter. She has been integral in bringing together the Spanish-speaking type 1 diabetes (T1D) community. She hosts a Spanish-speaking E-Meet & Greet as well as a Facebook Group for Spanish Speakers. Paola also partnered with Breakthrough T1D’s national Culture and Belonging team to create a Spanish Bag of Hope for newly diagnosed families.
Paola is inspired by her son, Damian, who was diagnosed with T1D at the age of three. She recently released her first book, Damian: Living With Type 1 Diabetes, available in English and Spanish, to help educate newly diagnosed children and their families.

To be that person that people come to for support is reassurance that I’m where I’m meant to be.”
Widespread awareness brings us closer to advancing breakthroughs to cure, prevent, and treat type 1 diabetes (T1D) until it is a condition of the past. Visit our Diabetes Awareness Month page to learn more about how you can get involved.
The Cincinnati Bengals’ Orlando Brown, Jr. is a Breakthrough T1D Ambassador and Advocate. Orlando’s commitment to type 1 diabetes (T1D) advocacy stems from his family’s journey with the disease. It began with his grandmother’s diagnosis of T1D, followed by the devastating loss of his father, NFL player Orlando Brown, Sr., to diabetic ketoacidosis, a serious diabetes complication. His younger brother’s T1D diagnosis at just 11 years old fueled Orlando’s determination to advocate for more research, more education, and wider access to diabetes technologies and therapies that make life easier and safer for the more than 1.6 million Americans living with T1D.

Knowledge is power, and I’m sharing my family’s story to educate and inspire others who are living with type 1 diabetes.”
Widespread awareness brings us closer to advancing breakthroughs to cure, prevent, and treat type 1 diabetes (T1D) until it is a condition of the past. Visit our Diabetes Awareness Month page to learn more about how you can get involved.
Op-ed by Breakthrough T1D Chief Global Advocacy Officer Lynn Starr
In my role as Chief Global Advocacy Officer, I lead an incredibly talented and dedicated team based in Washington, D.C., to support our mission—accelerating life-changing breakthroughs to cure, prevent, and better treat type 1 diabetes (T1D) and its complications.
The Advocacy team has many functions, all in pursuit of driving our mission progress forward faster. Some examples:
- We work with regulatory agencies around the world to establish roadmaps for approvals of advanced therapies.
- We advocate for insulin affordability so anyone who needs it can access the lifesaving medication they need at a reasonable, predictable price.
- We work with insurance payers to ensure there’s coverage for treatments.
- We push for government funding for T1D research.
The team works tirelessly in pursuit of our mission—a mission that’s deeply personal to me. I’ve lived with T1D for 36 years, and my daughter was diagnosed just last year. I’ve personally benefited from the relentless work of this organization, and it’s amazing that we’ve come so far. From automated insulin delivery systems to continuous glucose monitors, the daily management of this disease today would be unrecognizable to me 36 years ago. These advances have made my life, my daughter’s life, and the lives of so many in our community so much better. But the work is not done! Life with T1D remains extremely challenging and requires constant attention and vigilance.
We know this at Breakthrough T1D, and we’re dedicated to pushing further and speeding life-changing breakthroughs. We have many tools in our toolbox as we do this work. One of our power tools is the voice of our grassroots volunteer army of advocates. They are determined champions of our cause. Our grassroots advocates amplify our policy positions on a wide range of issues. In particular, this community voice in support of U.S. government funding for T1D research through the Special Diabetes Program (SDP) has been critical.
The SDP is the U.S. Federal program created in 1997 to fund T1D research through the National Institutes of Health. It’s currently funded at $160 million annually and has contributed nearly $3.5 billion (!) to T1D research since its creation. Breakthrough T1D funds significant amounts towards T1D research ($2.5 billion to date), but we can’t do this alone. We need the SDP funding, too.
The SDP funding has had a direct hand in so many T1D advances, such as automated insulin delivery systems and the first disease-modifying therapy to delay T1D onset. These simply would not have happened without the SDP. And, with the promise of cell therapies, increased research funding is crucial.
The SDP is set to expire at the end of the year. We cannot let that happen! We’re working hard to push forward not just for an extension of the SDP, but also an increase in its funding.
Can you join our advocacy army and help us power forward? It’s incredibly simple: sign up to be an advocate and take action when we email you to ask. Just look for emails from Breakthrough T1D and click through to our action alert, where you’ll be asked to contact your Members of Congress urging support for the SDP.
That’s it—it’s really that easy. It’s also that important.
I’m honored to lead Breakthrough T1D’s advocacy efforts on behalf of the T1D community, and I am so thankful for all the support we’ve received to help drive our mission forward. Especially during National Diabetes Awareness Month, let’s make sure all our voices are heard!
In it to end it,
Lynn Starr
Chief Global Advocacy Officer
Alecia Wesner has been part of the Breakthrough T1D family since her diagnosis with type 1 diabetes (T1D) in Philadelphia in 1979. She has participated in Walks and Rides and served on her Community Board in New York City.
Since 2014, Alecia has participated in numerous T1D clinical trials, many of which were early artificial pancreas system trials. She has also participated in an implantable CGM clinical trial, a complication prevention trial, and numerous studies addressing the psychological and sociological impact of living with T1D.

Clinical trials are so empowering. Participating is a great way to contribute to science and also learn.”
Widespread awareness brings us closer to advancing breakthroughs to cure, prevent, and treat type 1 diabetes (T1D) until it is a condition of the past. Visit our Diabetes Awareness Month page to learn more about how you can get involved.











