
Medtronic Panel Discusses Supporting T1D Community During COVID-19
During a recent Breakthrough T1D Facebook Live event, a panel of leaders discussed Medtronic’s efforts to support the T1D community during the pandemic.

Meet the Scientist: Local Immune Suppression with a Novel Protein
Mark Poznansky, M.D., Ph.D., wants the seemingly impossible—a person with type 1 diabetes (T1D) has a procedure where beta cells are placed in their body, the doctor stitches it up and the person has functioning insulin-producing beta cells, for life. Of course, we’re not there yet, but, with Dr. Poznansky—and many, many other researchers—Breakthrough T1D […]

Improving the Lives of Veterans With Diabetes
How to access the latest diabetes management tools through the Department of Veterans Affairs with Armed For Diabetes .
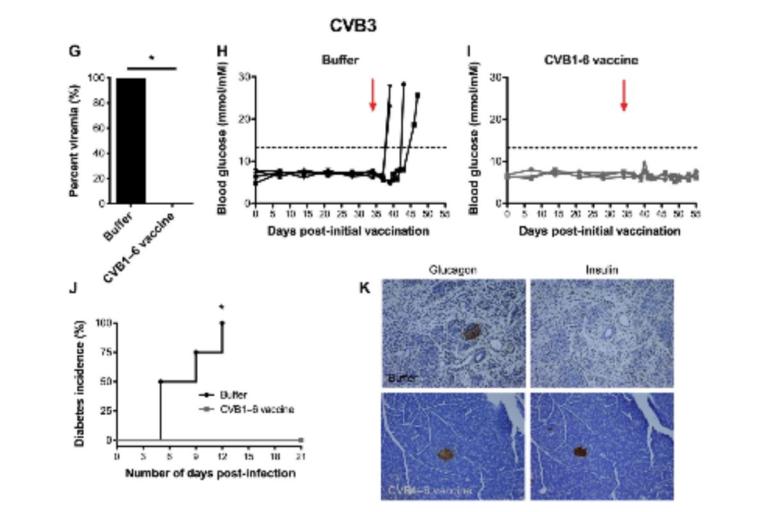
A Promising (Preclinical) Vaccine for Type 1 Diabetes
Wouldn’t it be nice if you could take a vaccine in infancy, and type 1 diabetes would not develop? Preclinical research suggests it might not be that far away.

Dr. Carla Greenbaum Talks about T1D TrialNet During the COVID-19 Pandemic
Carla Greenbaum, M.D., chair of TrialNet—an international Breakthrough T1D-supported network that is dedicated to finding cures for type 1 diabetes (T1D)—and a professor at Benaroya Research Institute, discussed T1D research in the time of COVID-19, including what TrialNet is up to, on Breakthrough T1D’s Facebook Live event. She chats about: What do we know now […]

Breakthrough T1D Plays a Leadership Role, Again, to Marshal Resources for T1D Research
It’s 2004. The California Institute for Regenerative Medicine (CIRM), which will fund $3 billion in stem cell research to advances new therapies, is on the ballot, if it gets approved. Fortunately, the patients, families and scientists were the voices that day, with nearly 60% voting to approve it. In the time since 2004, it has […]

At the Front Lines in COVID-19: Breakthrough T1D-Funded Scientist’s Company Developing a Vaccine
With coronavirus closing schools and forcing so many people to stay inside or wear facemasks every time they step outdoors, COVID-19 is threatening to make everyone stir-crazy well into summer. And unfortunately, the safety of herd immunity remains a long way off. In the meantime, scientists are battling coronavirus in a number of ways, and […]

Europe Supports Autoantibodies as Biomarkers for T1D
It was 2015—five years ago—that three organizations (BreakthroughT1D, the Endocrine Society and the American Diabetes Association) published a statement urging the adoption of a staging system of type 1 diabetes (T1D) that uses islet autoantibodies as biomarkers. (Biomarkers, in this sense, are indicators that a person is developing T1D but is still without symptoms.) It […]

In-Home Test Kits Make Screening Convenient and Easy
TrialNet—an international Breakthrough T1D-supported network of leading academic institutions, endocrinologists, physicians, scientists and healthcare teams dedicated to finding cures for type 1 diabetes (T1D)—has a new in-home test kit that makes screening far more easy and convenient. And FedEx will come pick it up, at no cost to you! If you have a relative with […]

Type 1 Diabetes Experts Unite In Virtual Summit for Families
Breakthrough T1D partners with Carb DM to make event a reality A long-running California type 1 diabetes (T1D) event can now be attended by anyone, anywhere in the world. The Bay Area Diabetes Summit on April 26 brings together some of the most renowned experts in T1D to discuss T1D research, psycho-social challenges and technology […]