What’s happening?
Today, Breakthrough T1D co-hosted an event with leading international diabetes organizations to discuss cardiovascular disease (CVD) and type 1 diabetes (T1D)—where we are, challenges that remain, and how we can work together in Europe and beyond to address this need for the T1D community.
The focus of the event: Doing more for CVD and T1D

This event, titled “Type 1 Diabetes & Cardiovascular Disease: From Data to Solutions,” took place today on November 14, 2025, in Brussels, Belgium. Breakthrough T1D co-hosted this event in conjunction with the International Diabetes Federation (IDF) Europe and the International Society for Pediatric and Adolescent Diabetes (ISPAD). The goal was to bring together different stakeholders to address a critical concern: the lack of therapies for cardiovascular disease for people living with T1D.
The speakers at the event covered a range of topics. Jonathan Rosen, Ph.D., Director of Research at Breakthrough T1D spoke about the often-overlooked link between CVD and T1D. Others touched upon lessons we can learn from (T2D) diabetes and CVD, the gaps in evidence that still exist, new frontiers in CVD care for people living with autoimmune diseases, and the inclusion of T1D in CVD therapy development.
Attendees
From Breakthrough T1D:
- Sanjoy Dutta, Ph.D., Chief Scientific Officer
- Jonathan Rosen, Ph.D., Director of Research
- Carmen Hurtado del Pozo, Director of European Research
- Hilda Ahnstedt, Program Officer of European Research
- Alessandro Bisio, M.D., Director of Medical Affairs, International
Other attendees:
- Speakers from the European Medicines Agency
- Member of the European Parliament: Elena Nevado del Campo (EPP, Spain)
- People living with T1D
- Researchers
- Industry leaders
Why this matters

People living with T1D have a high risk of developing heart complications, despite advances in T1D care. At this time, there are only a few options available to lower this risk. In the United States and Europe, people with T1D have access to blood pressure medications and lipid-lowering medications (for example, statins) that can reduce the risk of developing CVD.
Other therapies, like the SLGT inhibitors empagliflozin or dapagliflozin, are the standard of care in the U.S. to treat heart failure in people without diabetes or with T2D. This class of drugs has revolutionized treatment and significantly reduced the rates of heart failure in these populations. However, people with T1D were excluded from these clinical trials—despite the fact that heart disease remains a critical concern for the T1D community. While not strictly approved, these drugs are occasionally prescribed off-label to reduce the risk of heart failure in people with T1D. Greater accessibility will require regulatory approval, and these options are not currently available for people living in Europe.
Despite the availability of some treatments that can reduce the risk of heart complications, there are no available cardioprotective therapies in the U.S. or Europe that can prevent CVD in people with T1D. More targeted, innovative therapies and treatment strategies are needed to further reduce the risk of—or ideally prevent altogether—cardiovascular complications in the T1D population.
There is a clear unmet need for the T1D community: the risk of CVD is high, and there are not enough options to treat or prevent it. That’s where events like these come in. They are a call to action for collaborative research efforts, better data integration, new and innovative ideas, and global cooperation towards a shared goal of better heart health for people living with T1D. The growing prioritization of CVD is evidenced by the European Commission’s cardiovascular health plan—which includes the Joint Action on Cardiovascular Diseases and Diabetes—showcasing how stakeholders in Europe are taking charge to accelerate breakthroughs for CVD and T1D.
What we’re saying
“Cardiovascular disease remains the leading cause of death for people with type 1 diabetes, yet targeted therapies and guidelines are still lacking. In collaboration with IDF Europe, ISPAD, and aligned with the European Commission’s cardiovascular plan, this event brings together researchers, clinicians, patient advocates, regulators, and industry partners to drive collaborative solutions and accelerate progress toward better prevention and treatment strategies.”
Addressing the challenge of cardiovascular complications is a cornerstone of our Improving Lives strategy. Breakthrough T1D is actively supporting clinical trials for therapies that can reduce or prevent CVD in people with T1D, and we are working on identifying new projects and researchers to fund to further accelerate these efforts.
We are committed—on a global scale—to making sure people with T1D live the best lives possible, which includes having accessible therapies that reduce the risk of heart complications. Events like these bring together the right people that can make this happen in Europe and beyond.
What’s happening?
Today, Bayer shared data from the phase 3 FINE-ONE clinical trial. These results, which were presented at the American Society of Nephrology Kidney Week in Houston, TX, showed that finerenone (Kerendia™/Firialta™) significantly reduces urine albumin-to-creatinine ratio (UACR), a measure of kidney damage, in people with chronic kidney disease (CKD) associated with type 1 diabetes (T1D). This is an exciting win for the T1D community!

Finerenone
Finerenone is a drug that has already been approved for the treatment of CKD in type 2 diabetes. In CKD, the hormone aldolsterone is overactive, leading to kidney damage. Finerenone blocks this hormone’s activity to protect the kidneys from further damage.
What did the FINE-ONE study show?
The global, phase 3 FINE-ONE clinical trial investigated the use of finerenone in people with CKD associated with T1D. This trial achieved its primary outcome: finerenone significantly reduced UACR—a measure of kidney damage—compared to placebo.
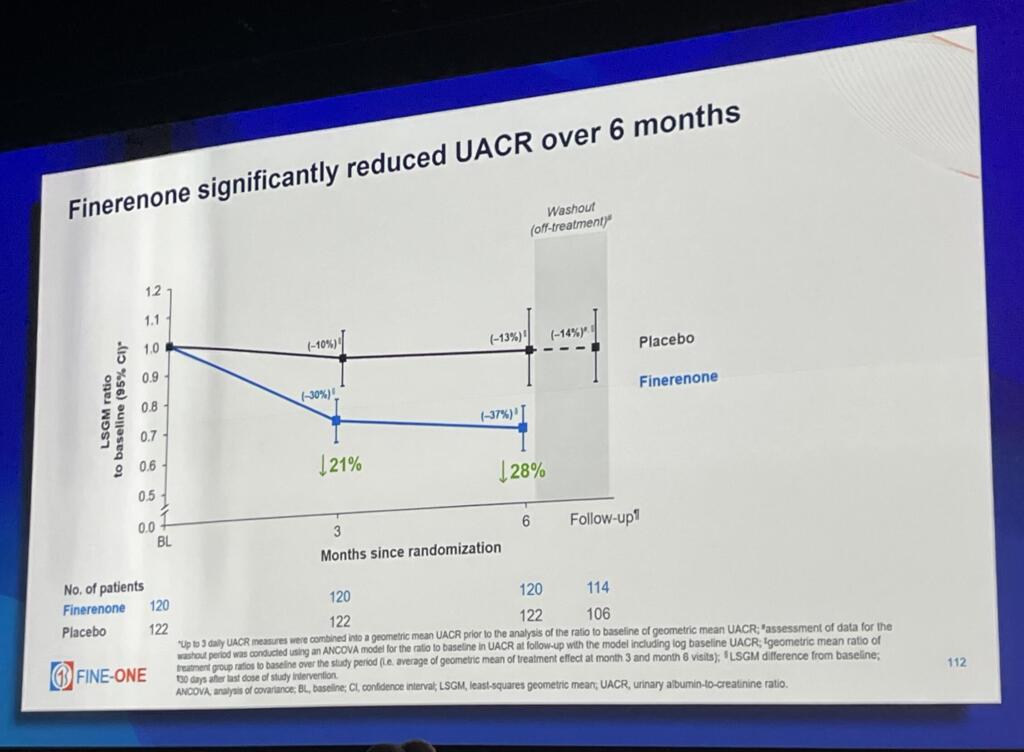
So, what does this mean? Previous clinical trials with people who have type 2 diabetes and CKD have shown that reductions in UACR from finerenone are associated with positive long-term health outcomes. In people with T1D, reductions in UACR caused by finerenone are likely indicative of lower risk of kidney disease progression, kidney failure, and cardiovascular events.
Finerenone was well-tolerated, with no new safety issues reported and few serious adverse events. Based on these results, Bayer intends to submit the data for regulatory review, with the goal of expanding finerenone’s indication to include treatment of CKD in people living with T1D. If finerenone gets approved, this would be an incredible step forward for people living with the burden of both kidney disease and T1D.
Why does this matter for T1D?
CKD is one of the most common complications of T1D. Nearly a third of people living with T1D will develop CKD, increasing the risk of both kidney failure and cardiovascular disease. Right now, the treatment options for people with T1D and CKD are limited—and there is a major need for therapies that can address this.
Finerenone is the first therapy in three decades to achieve positive outcomes for CKD in people with T1D. This is a huge step forward in the right direction, and this exciting data has the potential to transform the lives of people living with T1D with CKD if finerenone gets regulatory approval.
The bigger picture
Breakthrough T1D strategically collaborated with Bayer to support the FINE-ONE clinical trial, and we are committed to further collaboration with Bayer to advance therapies for people with T1D. Working together with Bayer and other companies will allow us to deliver more treatments to people with T1D to address complications like kidney disease.

“People with type 1 diabetes and chronic kidney disease face an immense burden due to their increased risk for both kidney and cardiovascular events,” said Jonathan Rosen, Ph.D., Research Director at Breakthrough T1D. “Breakthrough T1D remains committed to collaborating with Bayer to improve kidney care for people with type 1 diabetes.”
As a part of our Improving Lives portfolio, we aim to identify and support treatments and therapies that can address complications that arise from T1D, like CKD. The FINE-ONE trial opens the door for more therapies to come that could transform the landscape of available therapies for people with T1D, and industry investment in this space helps drive progress forward faster than ever. The T1D community needs more options, and our strategic industry partnerships will help make this possible.
We look forward to seeing the regulatory decisions that stem from FINE-ONE, and we are incredibly excited for what the future holds for CKD and T1D.

ADA Recap Series
This article is the first of our three-part ADA Recap Series. Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’re here to report on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding. Look out for tomorrow’s article for updates on Cures.
Improving Lives
Breakthrough T1D’s Improving Lives program focuses on devices, insulins, adjunctive therapies, treatments for complications, and psychosocial interventions to improve the health and quality of life of people living with T1D.
Adjunctive therapies and complications
There was significant focus on GLP-1 receptor agonists (GLP-1RAs) and SGLT inhibitors (SGLTi) in reducing long-term complications and improving glycemic control in people with T1D.

GLP-1 receptor agonists
Glucagon-like peptide 1 receptor agonists mimic the hormone GLP-1, which elevates insulin and regulates appetite. Examples include Ozempic® (semaglutide) and Mounjaro® (tirzepatide), which acts on both GLP-1 and a similar target, GIP.

SGLT inhibitors
Sodium-glucose cotransporter inhibitors target kidney cells to prevent them from reabsorbing glucose into the blood so it gets excreted as waste. Examples include Farxiga® and Zynquista®.
While SGLTi and GLP1-1RAs have proven effective for heart and kidney disease in type 2 diabetes (T2D) and in people without diabetes, people with T1D have often been excluded from critical trials. Thanks to years of advocacy and support from Breakthrough T1D, T1D trials are ongoing—and real-world evidence suggests that GLP-1RAs and SGLTi could be impactful in the T1D community as well.
Real-world evidence for GLP-1RA use in T1D
- Presenter: Ildiko Lingvay, M.D.; University of Texas Southwestern
- People with T1D have self-reported that they decided to try GLP-1RAs for weight loss and improved glycemic control.
- Real-world evidence suggests that GLP-1RAs have a clinically meaningful impact on weight and reduced insulin dose.
- While GLP-1RAs are generally safe, some people have stopped use because of gastrointestinal side effects. These side effects are also seen in people with T2D and people without diabetes.
A review of SGLTi and GLP-1RAs in reducing chronic kidney disease (CKD) in T1D
- Presenter: David Cherney, Ph.D.; University of Toronto
- In the EMPA-KIDNEY trial that included non-diabetes participants and people with T1D or T2D, empagliflozin (SGLTi) improved kidney health in people with T1D.
- In the ATTEMPT trial, dapagliflozin (SGLTi) improved time in range (TIR), reduced HbA1c levels, and had positive effects on kidneys in youth with T1D.
- The Breakthrough T1D-funded enrolling phase 3 SUGARNSALT trial is testing whether sotagliflozin (SGLTi) can prevent progression of moderate to severe kidney disease in people with T1D, and it includes careful diabetic ketoacidosis (DKA) risk mitigation strategies.
- The SEMA-AP trial found that semaglutide (GLP-1RA) increases TIR in people with T1D when used alongside an AID system.
- The Breakthrough T1D-funded recruiting phase 2 REMODEL-T1D trial is testing if semaglutide (GLP-1RA) can improve kidney health in people with T1D.

Glucokinase
Glucokinase (GK) is an enzyme in liver cells that works in an insulin-dependent manner to regulate blood sugar. In people with T1D who have little insulin reaching the liver, GK can’t work as normal, contributing to higher blood sugar.
Use of a glucokinase activator for glycemic control
- Presenter: Klara Klein, M.D., Ph.D.; University of North Carolina
- In the phase 1/2 SimpliciT1 study, people with T1D who received the GK activator TTP399 showed improvements in blood glucose with fewer hypoglycemic events.
- A different study found that TTP399 does not increase the risk for DKA.
- These studies were done in collaboration with vTv Therapeutics, a company with funding and support from Breakthrough T1D and the T1D Fund: A Breakthrough T1D Venture. The phase 3 CATT1 study for TTP399 is testing whether it can reduce moderate to severe hypoglycemic events in people with T1D.
Adjunctive therapies and complications highlight: Breakthrough T1D-funded research
Halis Kaan Akturk, M.D. (University of Colorado), Janet Snell-Bergeon, Ph.D., MPH (University of Colorado), and Viral Shah, M.D. (Indiana University) presented findings from the Breakthrough T1D-funded ADJUST-T1D clinical trial, which was recently published in the New England Journal of Medicine Evidence. The trial tested whether semaglutide (GLP-1RA) can improve glycemic and weight outcomes in adults with T1D and obesity who are using an AID system. 36% of people treated with semaglutide met the primary endpoints of TIR greater than 70%, time below range less than 4%, and weight loss of 5% or more compared to the placebo, and the drug was well-tolerated and safe. This trial represents critical evidence for use of a GLP-1RA as a potential way to manage both glycemic control and weight in people with T1D.
Ye Je Choi, MPH (University of Washington) reported on the CROCODILE study, which examined metabolic alterations in kidneys of people with T1D. She observed early structural and metabolic changes in kidneys that occurred before the onset of clinical kidney disease and associated structural damage. Her work could contribute to the development of biomarkers that can help predict the onset of kidney disease in people with T1D before it occurs.
Jeremy Pettus, M.D. (University of California at San Diego) conducted a phase 2 clinical trial to address insulin resistance in people with T1D. External insulin therapy can increase levels of insulin in the blood relative to glucose, which reduces sensitivity and may contribute to cardiovascular disease (CVD). Treatment with the glucagon receptor antagonist volagidemab, which prevents the liver from releasing glucose into the blood, reduces insulin requirements by 15%, resulting in improved glycemic control and insulin sensitivity without changes in bodyweight.
Schafer Boeder, M.D. (University of California at San Diego) worked with Dr. Pettus on a phase 1/2 clinical trial that tested whether the addition of SGLTi to the glucagon receptor antagonist volagidemab can further improve glycemic control in people with T1D. The combination of therapies increased TIR up to 86% from 70% and reduced daily insulin use by 27%. Further research is needed to better understand the safety profile of this regimen.
Justin Gregory, M.D. (Vanderbilt University) worked with Dr. Pettus and Dr. Boeder on the above study. He also presented on the use of GLP-1RAs and dual GLP-1/GIP receptor agonists for reducing complications in T1D.
Key takeaways
Clinical trials with GLP-1RAs and SGLTi are providing encouraging evidence about these therapies’ potential to improve long-term health in people with T1D. Breakthrough T1D is working toward a future where these drugs are an option for people with T1D to better manage their blood sugar and reduce complications.
Devices
Real-world insights from Automated Insulin Delivery (AID) systems
- Presenter: David Maahs, M.D., Ph.D.; Stanford University
- Based on published real-world evidence for AID systems in people with T1D, TIR is increased by an average of eight to 15% from baseline in a range of studies across various systems.
- Youth with T1D have better glycemic control and reduced rates of DKA with AID systems. Those with lower TIR at the start of AID system use see the greatest improvements.
Real-world evidence: iLet Bionic Pancreas AID system
- Presenter: Steven Russell, M.D., Ph.D.; Beta Bionics
- The iLet Bionic Pancreas contains a continuously adapting algorithm that automatically determines insulin doses. No carbohydrate counting is required, and meals are only announced as breakfast, lunch, and dinner.
- Data was collected from 16,000 users over two years.
- Users achieved an average HbA1c level of 7.3%, down from 8.9%. This is accompanied by low rates of hypoglycemia and significantly improved self-reported quality of life.
Continuous ketone monitoring: Innovations and clinical applications
- Presenters: Ketan Dhatariya, MBBS, M.D., Ph.D. (Norfolk and Norwich University Hospitals), Lori Laffel, M.D., MPH (Harvard University), Jennifer Sherr, M.D., Ph.D. (Yale University), and Richard Bergenstal, M.D. (HealthPartners Institute).
- DKA rates are increasing in the U.S., but mortality rates from DKA are decreasing.
- The history of continuous glucose monitoring (CGM) offers a roadmap for continuous ketone monitoring (CKM) development, showing how early skepticism gave way to broad clinical impact.
- CKM could allow for earlier detection of rising ketones to prevent DKA. CKM also has the potential to identity infusion set failures, be a valuable addition to AID systems, help monitor early-stage T1D, and more.
- Five new studies funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) will develop CKM for safe and effective use of SGLTi in T1D.
- Tandem, Beta Bionics, Sequel MedTech, and Ypsomed announced plans to integrate Abbott’s dual glucose ketone sensor into their AID systems.
Making the case for time in tight range
- Presenter: Gregory Forlenza, M.D.; University of Colorado
- Dr. Forlenza presented on the benefits and challenges of time in tight range (TITR), also known as time in normal glycemia (TING), defined as blood glucose levels between 70-140 mg/dL.
- TITR will likely be more clinically beneficial than TIR as fluctuations outside of TITR may be better predictors of complications and offer a better therapeutic window for intervention.
- More research is needed to advance therapeutics that will allow people with T1D to achieve TITR before it can be integrated into clinical decisions.
Devices highlight: Breakthrough T1D-funded research
Erin Cobry, M.D. (University of Colorado) presented the results of a Breakthrough T1D-funded clinical trial evaluating an artificial intelligence-powered AID algorithm designed to not require meal announcements. She showed that this algorithm (used without meal announcements) improved overnight TIR, and provided equivalent daytime TIR, compared to participants’ usual care. A major goal for Breakthrough T1D is to advance AID systems that do not require meal announcements to improve both glucose outcomes and quality of life for people with T1D.
Key takeaways
Devices have transformed how this disease is managed. Systems are becoming easier to use with less user input—and, critically, people with T1D are doing better. This is the dream Breakthrough T1D had when we launched the Artificial Pancreas project 20 years ago. We will continue to drive toward our goal of developing systems that provide superior health outcomes with minimal user burden.
Insulins
Inhaled insulin treatment for youth with T1D
- Presenter: Michael Haller, M.D.; University of Florida
- Afrezza® is an inhaled, fast-acting insulin that has proven to be effective in adults.
- The phase 3 INHALE-1 study examined Afrezza® in youth with T1D. Users report greater treatment satisfaction and no increase in weight compared to injected rapid-acting insulin analogs.
- Afrezza® is safe for youth with T1D. The most common adverse events were pulmonary-related, such as coughing.

Breakthrough T1D’s Improving Lives team making an impact
Courtney Ackeifi, Ph.D., Senior Scientist, hosted an Improving Lives Happy Hour with Breakthrough T1D-funded researcher Jeremy Pettus, M.D. The discussion included research priorities for adjunctive (non-insulin) therapies for people with T1D and their healthcare providers. They also discussed the importance of industry partnerships and the role of Breakthrough T1D in driving these relationships, which can accelerate new T1D therapies toward the clinic.
Dr. Ackeifi also spoke at the ADJUST-T1D trial update, contextualizing the use of adjunctive therapies like GLP-1RAs for superior glucometabolic control in people with T1D.
Look out for tomorrow’s article for an update on Cures research presented at ADA 2025!
Having type 1 diabetes (T1D) can increase your risk for additional health problems, particularly if your blood sugar isn’t well-managed. The longer you have T1D, the higher your risk becomes for developing complications. For people living with T1D, eye disease is a troubling reality. Over 40% of people with T1D develop diabetic eye disease, sometimes resulting in blindness.
Fortunately, Breakthrough T1D-funded research advances, including tools like continuous glucose monitors and automated insulin delivery systems, are helping to reduce the risk of complications caused by prolonged high blood sugar.
What is diabetic eye disease?
There are two main types of diabetic eye disease: diabetic retinopathy and diabetic macular edema.
Diabetic retinopathy is one of the most common complications of T1D. It is a progressive condition, often the result of chronic high blood-sugar levels, which can damage the blood vessels in the retina of the eye and eventually cause excess, abnormal blood vessel growth. In advanced stages, contracting scar tissue may cause the retina to detach, which can lead to permanent vision loss.
Diabetic macular edema is a common—but not the only—consequence of diabetic retinopathy. It is caused by a buildup of fluid which causes the macula (the center of your retina that helps you see color and fine details) to swell and thicken, negatively affecting vision.
Learn more about the causes of diabetic eye disease.
How to reduce your risk of eye disease
Careful health monitoring can help reduce your risk of developing diabetic eye disease. This includes:
Keep your blood-sugar in range

Schedule annual eye exams with a retinal specialist

Manage blood pressure and cholesterol

Consult your doctor about early treatment options for preventive measures
Treating diabetic eye disease
Many effective therapies for diabetic eye disease are now available, many of which were made possible through Breakthrough T1D-funded research, including anti-VEGF therapy.
Vascular endothelial growth factor (VEGF) is a protein that promotes the growth of new blood vessels. In people with diabetic retinopathy, VEGF becomes overactive, and the new, fragile blood vessels can rupture and leak fluid. Breakthrough T1D played a critical part in clinical studies that led to the approval of two therapies—Lucentis® (ranibizumab) and Eylea® (aflibercept)—that inhibit VEGF and allow healthy regrowth of damaged blood vessels in the eyes.
Most recently, in February 2025, the FDA approved Susvimo®, now an alternative treatment option with fewer eye injections compared to the standard of care, to treat diabetic macular edema and help maintain vision.
Learn about other treatments for eye disease.
Funded research in eye disease and type 1 diabetes
Since the 1970s, Breakthrough T1D has funded research in the treatment and prevention of diabetic eye disease. We continue to strategically invest in therapies that have short- and long-term impacts on the lives of people with type 1 diabetes, including:
- Randomized Clinical Trial Evaluating Fenofibrate for Prevention of Diabetic Retinopathy Worsening
Fenofibrate is a safe, generic lipid-lowering medication that is being repurposed for T1D. This trial will determine if treatment with fenofibrate is effective at preventing diabetic retinopathy from worsening in individuals with mild to moderately severe early-to mid-stage retinopathy. This study will also give information on whether variation in blood sugar control is a risk factor for future worsening of diabetic eye disease.
- A Phase 2 Study Evaluating Short-Term Efficacy of Tonabersat (Xiflam) for Diabetic Macular Edema
This study will evaluate if treatment with tonabersat (Xiflam) has a short-term effect on central retinal thickness in eyes with good vision and diabetic macular edema or abnormal swelling of the central retina.
We are also supporting additional research to find new ways to prevent the development of diabetic eye disease, stop its progression, and restore lost vision through the Mary Tyler Moore Vision Initiative.
Breakthrough T1D will continue to address the complications of type 1 diabetes, including eye disease, to improve health outcomes for the entire T1D community.
They’re all over the news: Ozempic ®. Trulicity ®. Jardiance ®. Mounjaro ™. And more.
These drugs are all approved for glucose control in type 2 diabetes (T2D). Some of them also have additional indications reflecting their demonstrated benefits for cardiovascular disease, kidney health, and obesity.
None of these drugs are currently approved for people with type 1 diabetes (T1D). However, a recent consensus report addressed the growing interest in GLP-1 receptor agonists as an adjunctive therapy for T1D and their “potential to improve glycemic and metabolic outcomes without increasing the risk of severe hypoglycemia or diabetic ketoacidosis.”
Let’s examine GLP-1 therapies and SGLT inhibitors and consider how drugs like Ozempic may one day help people with type 1 diabetes.
GLP-1 therapies
GLP-1 (glucagon-like peptide-1) receptor agonists work in multiple ways to control blood glucose and obesity. They block the release of glucagon, stimulate insulin production, slow the rate at which your stomach empties, and increase the sensation of feeling full. They are usually injected, but oral versions are available.
In people with T2D, this class of drugs lowers blood sugar levels and, for most people, causes weight loss. GLP-1 therapies have also been shown to reduce the risk of long-term cardiovascular complications often experienced by people with T2D, such as heart attack and stroke.
GLP-1 drugs include Ozempic/Rybelsus/Wegovy (semaglutide), Trulicity (dulaglutide), Victoza (liraglutide), and Mounjaro (tirzepatide), among others.
When GLP-1 treatments hit the market in the early 2000s, Breakthrough T1D and others funded several clinical trials to test whether GLP-1 receptor agonists, in addition to insulin, improved outcomes for people with type 1 diabetes. While some of these studies showed that the addition of GLP-1 therapies to insulin led to improvements in HbA1c, total insulin dose, and weight, the benefits were relatively modest and accompanied by some side effects like hypoglycemia. As a result, these studies did not lead to GLP-1 receptor agonists being highly adopted for use by people with T1D.
However, these trials were done with older GLP-1 drugs. We are investigating whether the newest, most advanced GLP-1 therapies (like Ozempic) will improve the health of people living with type 1 diabetes. (See below!)
SGLT inhibitors
SGLT (sodium-glucose co-transporter) inhibitors are oral medications for people with T2D that lower blood sugar by preventing the kidneys from reabsorbing glucose, leading to the excretion of sugar through the urine.
In addition to improving blood sugar for people with and without T2D, these drugs also provide benefits such as weight loss, blood pressure reduction, and transformative benefits to the heart and kidneys.
SGLT drugs include Jardiance (empagliflozin), Farxiga (dapagliflozin), and Invokana (canagliflozin), among others. Despite demonstrating improved glucose control for people with type 1 diabetes, SGLTs have not been approved for people with T1D in the U.S. Increased risk of diabetic ketoacidosis (DKA) in this population limits the use of these therapies. A key Breakthrough T1D priority is to find ways to mitigate this risk so people with T1D can also take advantage of the SGLT cardiovascular and renal benefits.
Breakthrough T1D-funded research in GLP-1 and SGLT therapies
Breakthrough T1D has a long history with GLP-1 medications like Ozempic. In the 1980s, Breakthrough T1D-funded researcher Pauline Kay Lund, Ph.D., was the first to clone the hormone glucagon and discover two new hormones, one of which was GLP-1.
Today, there is real-world evidence of GLP-1 receptor agonists improving the lives of people with type 1 diabetes.
Real-world evidence
Observational studies using historical patient data from electronic health records (not randomized clinical trials) that can give an idea if a drug might be beneficial or not for a certain indication.
Evidence demonstrates that semaglutide (Ozempic) or tirzepatide (Mounjaro) have the potential to lower A1c, increase time-in-range, and reduce the amount of daily insulin needed in people with T1D. More research is needed in this area. That’s where Breakthrough T1D comes in!
Breakthrough T1D-funded research on GLP-1 and SGLT therapies is investigating the benefits of these drugs for people with T1D, including reducing the risk of common complications of type 1 diabetes like cardiovascular disease and kidney disease.
Snapshot of active clinical trials in GLP-1 and SGLT therapies
Here are a few examples of Breakthrough T1D-funded clinical trials in GLP-1 and SGLT therapies that are currently underway:
| Clinical Trial Name | Study Details |
|---|---|
| REMODEL T1D | Determine whether semaglutide (Ozempic) protects the kidneys in those living with T1D. |
| Triple Therapy in T1DM | Assess whether the addition of dapagliflozin (Farxiga) to semaglutide (Ozempic) and insulin improves glycemic control in those living with T1D. |
| SUGARNSALT | Determine the effectiveness and safety of sotagliflozin (Inpefa) in slowing kidney function decline in those living with T1D and moderate to severe diabetic kidney disease. |
| Dapagliflozin + Pioglitazone in T1D | Examine how adding dapagliflozin (Farxiga) and pioglitazone (Actos) to insulin therapy affects glucose control and ketone concentration in people living with T1D. |
Our commitment to improving lives
Breakthrough T1D strives to improve health outcomes in people living with type 1 diabetes. Insulin therapy alone is often not enough for people with T1D to achieve glucose and metabolic control. The use of adjunctive drugs that complement insulin therapy can help. Since the FDA has already approved these medications for treating other conditions, positive results from these clinical trials could speed up the adaptation of these therapies for people living with T1D.
Learn more about clinical trials and how they are advancing breakthroughs for the T1D community.
While we look back on 2024, we can reflect upon the incredible progress we’ve made in advancing breakthroughs toward cures and improving everyday life with T1D.
This wouldn’t have been possible without each and every one of you and your continued support of our mission as we drive toward cures for T1D.
Here are the top 11 advances that together, we made happen in 2024:
Breakthrough T1D announced the launch of Project ACT, an initiative aimed at accelerating breakthroughs in T1D cell replacement therapies that do not require broad immunosuppression. Recent advances, such as Vertex’s stem cell-derived islets, have been made possible by Breakthrough T1D’s Cell Therapies program as part of our drive toward cures. The goal of Project ACT is to push research, development, regulatory policies, access, and adoption to increase the rate at which cell therapies without the need for broad immunosuppressants will become available to people with T1D.
Why this matters: Immunosuppressive drugs are a barrier to access to cell replacement therapies because of their toxic side effects, which is why islet transplants are currently only available to people with severe low blood sugar (hypoglycemic) unawareness and episodes. By striving toward a future where we realize the benefits of cell replacement therapies without the downsides of the current regimen of immunosuppressants, we will make islet replacement therapies broadly accessible to the T1D community.
Vertex’s clinical trial of VX-880, a first-generation stem cell-derived islet replacement therapy for people with severe hypoglycemia (requiring the use of immunosuppressants), has transitioned into a phase 1/2/3, or pivotal, trial. This news comes after Vertex shared incredibly promising data in the earlier phases of the trial, including 11 of 12 participants reducing or eliminating the need for external insulin.
The upcoming trial will expand to 50 people who will get a single, target dose of VX-880. The primary endpoint will be insulin therapy independence without severe hypoglycemic events after one year. This is the final clinical testing stage before Vertex can seek FDA approval.
Breakthrough T1D has a decades-long relationship with Vertex and the leading scientists behind stem cell-derived islet therapies, an advancement that would not have been possible without Breakthrough T1D funding and support. The T1D Fund had invested in Semma Therapeutics, which was acquired by Vertex Pharmaceuticals in 2019, eventually leading to the active clinical development of VX-880 in T1D.
Why this matters: This is the first time a scalable cure for T1D is entering phase 3 clinical trials—a significant win and a huge step toward accelerating the delivery of cell therapies to members of the T1D community!
Tegoprubart: Transplant Survival Without Standard Immunosuppressive Drugs
Tegoprubart, an anti-CD40L immunotherapy that limits the immune response, is being tested in a Breakthrough T1D-funded study in people with T1D and severe hypoglycemia who have received deceased donor islets. Eledon Pharmaceuticals announced promising initial results in which two of three people achieved insulin therapy independence. According to the study, tegoprubart is safer for both people and transplanted cells in comparison to broad immunosuppressants, with milder side effects and greater islet survival. To further support this effort, the T1D Fund: A Breakthrough T1D Venture invested in Eledon.
Cell Pouch: A Home for Transplanted Islets
Breakthrough T1D has been supporting the development of Cell Pouch, an implantable device from Sernova that provides a safe, immune-protected environment for transplanted islet cells. In phase 1/2 clinical trials, all six people who received donor islets within the Cell Pouch achieved sustained insulin therapy independence with immunosuppressants, including long-term islet survival and function over five years without harmful side effects.
Why this matters: Standard of care immunosuppressive drugs that help avoid transplant rejection come with unwelcome side effects, such as increased risk of infection and malignancy and toxicity to kidneys, nerves, and islet cells themselves. Breakthrough T1D is focused on finding alternative ways to keep transplanted islet cells alive and healthy so that cell replacement therapies can become more tolerable and accessible.
In a major effort spearheaded by Breakthrough T1D, the first internationally recognized clinical guidelines for those who test positive for T1D autoantibodies have been published. These include guidance on monitoring frequency, education, and psychosocial support in addition to recommended actions for healthcare professionals (HCPs) when the risk of T1D progression is high. The guidelines were cooperatively developed with over 60 international experts spanning ten countries.
Why this matters: Previously, there had been no consensus on monitoring guidelines for people who test positive for T1D autoantibodies. Standardization of clinical recommendations means that individuals, families, and HCPs have tangible next steps to monitor early T1D progression and catch life-threatening complications sooner.
- Breakthrough T1D is leading a campaign to secure a recommendation for T1D screening from the U.S. Preventative Services Task Force (USPSTF), the main authority for preventative care. Approval would require T1D screening to be covered by insurance—an important step forward in expanding access.
- Driven by Breakthrough T1D’s advocacy efforts, The Centers for Medicare and Medicaid Services (CMS) established a unique ICD-10 code for stage 2 T1D. ICD-10 codes are used by HCPs to classify and document diagnoses, symptoms, and procedures. These codes provide a unified way for doctors and providers to indicate what diseases or conditions a person has in their electronic health record (EHR), empowering HCPs to document accurate diagnoses and provide the best possible care.
Why this matters: T1D early detection is critically important to prevent life-threatening complications at diagnosis and to give people necessary resources to make informed decisions about their health. Integrating T1D screening into the U.S. healthcare system will increase access to care.
The past year has seen some important advances in glucose management therapies and devices:
- Cadisegliatin, an activator of a blood sugar regulator in the liver, is being investigated in a phase 3 clinical trial (TTP399) as an adjunct therapy to insulin for people with T1D, although it is currently placed on clinical hold. vTv Therapeutics, the trial sponsor, is also a T1D portfolio company.
- The Omnipod 5 app is now available for the iPhone, making it easier to control the Omnipod without the need to carry a controller. It can also integrate with the Dexcom G6 continuous glucose monitor (CGM).
- Eversense 365 is the first FDA-approved year-round sensor that can easily integrate with automated insulin delivery (AID) systems. Other sensors require replacement after 10-14 days.
Why this matters: While advancements in glucose management have been pivotal in improving health outcomes for people with T1D, access remains a challenge. AID systems are globally underutilized, and not everyone has the necessary technology to connect devices. Breakthrough T1D is working to not only support advances in glucose management but also increase access.
Related content: While Breakthrough T1D consistently strives to improve the lives of those living with T1D, as an organization we have made incredible progress in the development of AID systems, also called the artificial pancreas systems. Read a historical perspective written by Breakthrough T1D volunteer Doug Lowenstein that covers conception to FDA approval of the first artificial pancreas systems, which changed the lives of people with T1D.
An inquiry spearheaded by the Breakthrough T1D affiliates in the U.K. uncovered risks of developing T1D eating disorders (T1DE), including bulimia, anorexia, or insulin restriction to lose weight. There is a significant gap in education and clinical guidelines for HCPs, a lack of internationally recognized criteria for T1DE diagnosis, and insufficient care integration, leading to preventable complications and healthy years of life lost. Breakthrough T1D recognizes the importance of spreading awareness and support for T1DE, and much work is needed to improve the lives of those living with T1DE.
Why this matters: There is an urgent need to change the way T1DE is approached, including integrated physical care with mental health services to get people with T1DE the access to care that they need.
In a study that included people with T1D, finerenone (Kerendia®) has been shown to improve cardiovascular outcomes in adults with heart failure. The drug is already approved in the U.S. to treat kidney and cardiovascular disease in people with T2D. Based on these results, Breakthrough T1D is supporting a clinical trial (FINE-ONE) in conjunction with Bayer to investigate the use of finerenone for T1D with the hopes of reducing kidney complications.
Why this matters: Kidney and cardiovascular disease remain significant challenges for those with T1D, especially given the FDA’s recent rejection of an SGLT inhibitor to lower blood glucose in people with T1D and chronic kidney disease. Yet, a new clinical trial (SUGARNSALT) will better assess the benefits versus risks.
Breakthrough T1D is advocating for the regulatory approval of C-peptide, a biomarker for insulin production by beta cells, to be used as an endpoint in clinical trials. An endpoint can accurately predict a meaningful benefit in clinical trials for disease-modifying therapies (DMTs; treatments that can slow, halt, or reverse T1D). To support this endeavor, Breakthrough T1D scientists and an expert consensus panel published research with evidence supporting C-peptide as an endpoint. Breakthrough T1D is continuing to engage with regulators, coordinate with industry, and assess more clinical trial data to drive this effort forward.
Why this matters: Current clinical trial endpoints (HbA1c, hypoglycemia, and complications) are not the best way to gauge the clinical benefits of T1D therapies. If C-peptide gets regulatory approval to be used as an endpoint, clinical trials could be smaller and shorter while still accurately assessing the advantages of a DMT. This means that drug development can move more quickly, and people with T1D will be able to access therapies sooner.
Related content: Two years ago, the T1D community received the incredible news that Tzield® had become the first FDA-approved disease-modifying therapy that can significantly delay T1D onset. Breakthrough T1D volunteer Doug Lowenstein recounts the life-changing drug’s journey nearly 100 years after the discovery of insulin.
The T1D Index is a data simulation tool that measures the global health impact of T1D, bridging gaps in our knowledge of public health statistics. T1D Index 2.0 has new and improved functionality, including advanced simulation capabilities, validation of data, and enhanced user experience. Breakthrough T1D contributed to both the development and improvement of the T1D Index.
Why this matters: The T1D index is critical in defining the intercontinental scope of T1D, driving us toward country-specific solutions and improved global health outcomes.
Earlier this year, JDRF rebranded to Breakthrough T1D. While our mission remains the same, our name needs to better reflect who we are and where we’re going. Our new brand aligns with our mission to accelerate life-changing breakthroughs for those of every age living with T1D as we work toward a world without it.
Why this matters: The proof is in the name—each day we strive to increase and accelerate breakthroughs in T1D, and it’s critical for our brand to accurately reflect our mission.
It’s certainly been an exciting year! While we still have more work to do, it’s crucial to celebrate our wins, both big and small, to see how far we’ve come in our push to make T1D a thing of the past.
Together, we’re accelerating breakthroughs for people with T1D, and the support of the T1D community drives our mission forward every single day, leading the way to lifechanging therapies and cures. Let’s see what 2025 has in store!
Update – November 22, 2024
This morning, Lexicon Pharmaceuticals, Inc. (Lexicon) announced they received a “deficiencies preclude discussion” letter from the FDA regarding this drug application. The FDA is not approving sotagliflozin for use in glucose control in people with T1D and CKD and there will be no further discussion in regards to this application.
As a result, Lexicon announced that they will stop pursuing sotagliflozin for use in glucose control in people with T1D and CKD.
Breakthrough T1D is extremely disappointed with this decision. Our funded research exploring the use of sotagliflozin in people with T1D will continue, and we will continue to push on multiple fronts for therapies, like this one, that can help address the burden and unmet needs of the T1D community.
There is a deep unmet need for therapies in addition to insulin for glucose control in people with T1D. This is especially true for those with additional complications like CKD. Today, a U.S. Food and Drug Administration (FDA) advisory committee recommended against the agency approving Lexicon’s sotagliflozin for blood glucose control in individuals with T1D and CKD. Breakthrough T1D believes this drug has benefits for people with T1D and those benefits outweigh the risks.
The case for more therapies
For over 100 years, insulin therapy has been the main treatment for T1D. Better insulin options and new technologies, like continuous glucose monitors and automated insulin delivery systems, have improved health and quality of life for many people with T1D. There is nothing that people with T1D cannot accomplish. But, by nearly every health-related quality of life metric, people with T1D score lower than individuals without T1D. This includes general health, mental health, vitality, physical functioning, social functioning, and others.
If you look specifically at T1D metrics, a similar picture appears. Only 26% of individuals with T1D achieve an HbA1c level under 7%, the American Diabetes Association’s recommended HbA1c target to reduce the risk of long-term diabetes-related complications like cardiovascular and renal disease and diabetic retinopathy.
Despite the significant advances that Breakthrough T1D has been instrumental in realizing, people with T1D are not doing well enough. We need tools that make it easier to do better.
What is sotagliflozin?
Sotagliflozin is an SGLT (sodium-glucose co-transporter) inhibitor. SGLT inhibitors are oral medications originally developed for people with type 2 diabetes (T2D). They lower blood sugar by preventing the kidneys from reabsorbing glucose, leading to the excretion of sugar through the urine.
In addition to improving blood sugar, this class of drugs provides transformative benefits and have been approved for heart and kidney health in people with T2D and people without diabetes. In addition, they promote weight loss and blood pressure reduction.
Benefits of sotagliflozin for people with Chronic Kidney Disease
It has long been recognized that maintaining tight glucose control is crucial for preventing chronic kidney disease (CKD) in individuals with T1D. Recent data indicate that for those with T1D who already have CKD, there is a significant correlation between high HbA1c levels and an accelerated loss of kidney function. Therefore, effective glucose management is vital for individuals with both T1D and CKD to avert serious health complications.
In addition to improving glucose control, sotagliflozin has shown improvement in CKD outcomes in large trials of people with T2D. It is reasonable to expect these benefits would also be seen in those with T1D who also have CKD. While we do not yet have data on long-term kidney outcomes associated with sotagliflozin in T1D, we are encouraged by data demonstrating that sotagliflozin improves key biomarkers of kidney function in people with T1D.
The risk of diabetic ketoacidosis
There is one key consideration for the safe use of sotagliflozin: the risk of diabetic ketoacidosis (DKA).
Using SGLT inhibitors can increase the risk of DKA in people with T1D, so it’s important to monitor and manage this risk. In 2018, experts came together to address these concerns. Their findings led to an international agreement in 2019 on how to reduce the risk of DKA in people with T1D who are using SGLT inhibitors.
However, DKA is a risk for everyone with T1D, not just those taking SGLT inhibitors. It’s important to be aware of DKA as a regular part of managing T1D.
Breakthrough T1D has long supported research to advance safe, effective use of SGLT inhibitors in T1D
Breakthrough T1D’s Improving Lives program supports the development of SGLT inhibitors for T1D; this includes trials to assess benefits and risks of this drug class in T1D, as well as device-based and other strategies for DKA risk mitigation. SGLT inhibitors are FDA approved for T2D and certain non-diabetic populations such as those with CKD or heart failure, and they are used by some people with T1D off-label. Our work supports studies to build a body of evidence that will allow people with T1D to improve health outcomes through safe and effective use of SGLT inhibitors.
What comes next?
Breakthrough T1D believes the benefits outweigh the risks for people with CKD and T1D and sotagliflozin should be approved. That’s why Sanjoy Dutta, Ph.D., Breakthrough T1D Chief Scientific Officer, delivered public comments in support of the drug’s approval today.
As the formal review process moves forward, Breakthrough T1D will continue to invest in research to develop therapies for kidney disease in T1D that can help people live longer and healthier lives. While the advisory committee’s recommendation plays a critical role, it is not the only consideration in the final decision of approval (or not). The overwhelming majority of people with diabetes and world-renowned healthcare providers who commented were in agreement with Breakthrough T1D that the drug should receive approval.
The FDA are required to make a decision by December 20, 2024.
JDRF’s vision is a world without type 1 diabetes (T1D) and in the past fiscal year, through many top type 1 diabetes advances, we’ve made incredible progress toward that goal.
Your support of our efforts is inseparable from the top type 1 diabetes advances we’ve seen in accelerating cures, improving lives, and advocacy wins for people with T1D and their loved ones.
As we approach the end of fiscal year 2023 (FY23), let’s highlight the many top type 1 diabetes advances we’ve seen.
Top Type 1 Diabetes Advance 1: First T1D Disease-Modifying Therapy
In a historic moment for T1D—and one that Breakthrough T1D had a hand in from the beginning, supporting research from the 1980s on—the U.S. Food and Drug Administration (FDA) approved Tzield™ (teplizumab-mzwv) for use in delaying the onset of clinical disease in at-risk individuals aged 8+.
For the first time in history, Tzield will treat the autoimmune process behind T1D, not the symptoms, altering the course of the disease.
Among our top type 1 diabetes advances, this is the first disease-modifying therapy—treatments that can slow, halt, or reverse the course of the disease—for T1D to be approved, but it won’t be the last.
Additionally, months after Tzield’s FDA approval, Sanofi acquired Provention Bio, the manufacturer of Tzield.
The acquisition brings the first T1D disease-modifying therapy available in the U.S. into the portfolio of a global leading pharmaceutical company, representing an endorsement of the potential of these types of therapies and, we hope, the opportunity to bring this life-changing therapy and others in the pipeline to more people faster.
Tzield and breakthroughs like it put us on the pathway to finding cures and, one day, preventing T1D entirely.
Top Type 1 Diabetes Advance 2: A Blood Pressure Drug Preserves Beta Cell Function
A Breakthrough T1D-funded study found that children and teens newly diagnosed with T1D who took verapamil—a drug already approved to treat high blood pressure—were making more insulin one year after diagnosis than those on placebo. In other words, in the children and teens who took verapamil, more beta cells were healthier one year post T1D diagnosis than those in the children and teens who took the placebo.
This was the second trial that found the drug can preserve beta cells in the newly onset period.
Additional studies may be needed to further validate the results, as well as identify all benefits and potential side effects of the drug. Breakthrough T1D has the strategy to answer these and other questions.
The finding brings us closer to our goal of having numerous disease-modifying therapies widely available for people with type 1 diabetes.
Top Type 1 Diabetes Advance 3: Affordable Insulins for Everyone
Breakthrough T1D and partnering organizations are supporting nonprofit pharmaceutical manufacturer Civica Rx to produce biosimilar insulin that will cost no more than $30 a vial/$55 a box of five pens, regardless of insurance status.
One year after the Civica announcement, Eli Lilly, Novo Nordisk, and Sanofi all announced reductions to the prices of their insulins—including the most used insulins, such as Humalog, NovoLog, and Lantus.
Another big win for insulin affordability was the $35 monthly out-of-pocket co-pay cap for those on Medicare included in the Inflation Reduction Act that Breakthrough T1D fought hard to secure.
In April, the Senate Diabetes Caucus Co-Chairs, Jeanne Shaheen (D-NH) and Susan Collins (R-ME), introduced the INSULIN Act of 2023, another key step toward achieving affordable insulin for all who need it.
The bill seeks to limit out-of-pocket insulin costs by ensuring that people with commercial insurance pay no more than $35 or 25 percent of the net price per month for at least one insulin of each type and dosage form, and includes other important provisions to help make insulin more affordable and accessible.
You can contact your members of Congress and encourage them to support the INSULIN Act of 2023.
Top Type 1 Diabetes Advance 4: Turbo Boosting Cell Therapies
Breakthrough T1D is working to develop and deliver life-changing therapies that place healthy, insulin-producing beta cells back into the bodies of people with T1D. There was a lot of progress in FY23.
Vertex, which previously acquired Semma Therapeutics, also acquired ViaCyte, bringing together the leading companies developing stem cell-based therapies for diabetes.
Vertex is advancing a stem cell-derived islet replacement therapy for T1D. It’s in human clinical trials and showing amazing results, with one participant being off insulin entirely.
Vertex also started a trial with a new product using encapsulated stem cell-derived islets as replacement therapy, and is exploring gene-edited stem cell-based therapies—both with the goal of eliminating the need for immunosuppressive drugs.
Just this past April, Aspect Biosystems—an industry leader in 3D bioprinting technology—and Novo Nordisk announced a partnership to expand the development of a new class of treatments for diabetes and obesity, using Aspect’s bioprinting technology and Novo Nordisk’s expertise in stem cell and cell therapy development.
The Aspect-Novo Nordisk partnership’s initial focus will be on developing bioprinted therapies for transplant that would be designed to maintain normal blood-sugar levels without the need for immunosuppression. This could represent a transformative treatment for people living with T1D.
Additionally, the U.S. Food and Drug Administration (FDA) approved CellTrans’s Lantidra™, the first cell therapy to be authorized in the United States, for use in adults unable to approach average blood glucose levels due to current, repeated episodes of severe low blood sugar. This therapy, which requires the use of immunosuppressive drugs, takes deceased donor islets and places them into people with T1D suffering from repeated severe low blood-sugar, called hypoglycemia, events. This is an exciting first.

Approved! Numerous T1D Management Technologies
Breakthrough T1D funds research to facilitate the development of new therapies and technologies to make day-to-day life with T1D easier, safer, and healthier. In the past year, we saw:
Newly-Approved Artificial Pancreas (AP) Systems and Algorithms
- iLet® Insulin-Only Bionic Pancreas System for ages 6+
- Medtronic MiniMed™ 780G AP system for ages 7+
- Tidepool Loop, an algorithm that will allow for interoperability of continuous glucose monitors (CGMs) and insulin pumps
Newly-Approved Continuous Glucose Monitoring (CGM) Systems
- Dexcom G7® CGM system for ages 2+
A New Tool to Accurately Diagnose Type 1 in Adults
Misdiagnosing adults with T1D as having T2D is an all-too-common problem that can have tragic consequences. Breakthrough T1D and IQVIA teamed up to develop an algorithm using artificial intelligence to examine medical records and identify individuals who were diagnosed with T2D but actually have T1D. This could be used in real time to correct misdiagnoses, offering the potential for future development into a clinical decision support tool.
A First-of-its-Kind Lifesaving Tool: The T1D Index
Breakthrough T1D and other T1D-related organizations launched the T1D Index, a first-of-its-kind data simulation tool that offers the most accurate estimate of T1D ever created. The Index measures and maps how many people live with this condition in every country, the healthy years of life it takes from people living with T1D, the number of people who would still be alive today if they hadn’t died prematurely from T1D complications, and our global strategy to reduce the impact of T1D.
Go Forward
Your partnership has been crucial to these advances and many more. On behalf of our community, thank you for moving us forward and ever closer to a world without T1D.
We are excited for the top type 1 diabetes advances that fiscal year 2024 (FY24) will bring!
Read Past Blogs about Top Type 1 Diabetes Advances:
What We Can Be Proud of in 2022
What We Can Be Proud Of in 2020
Top 10 T1D Breakthroughs of 2019
What is gastroparesis?
Diabetic gastroparesis is a long-term condition that develops when the stomach is unable to empty itself properly. This means that food passes through slowly, leading to problems such as becoming full too early, nausea, and vomiting. Another key problem is that it can make timing of insulin doses more difficult, increasing the risk of hypoglycemia. These symptoms can be extremely troubling and result in poor quality of life.
Up to 50 percent of people with type 1 diabetes (T1D) have it, going from mild stomach-related pain to severe stomach-related events.
How is gastroparesis diagnosed?
There are a number of ways that gastroparesis can be diagnosed:
- Scan—called scintigraphy—that takes images of a radio-labeled meal as it goes through the digestive system at baseline and at 1, 2, and 4 hours
- Breath test, taken with a radio-labeled meal; generally, this takes 3-4 hours
- Wireless capsule that transmits various features (pH, temperature, etc.) at regular intervals to a portable receiver worn by the person, which takes around 6 hours
What is the treatment for gastroparesis ?
The majority of people with T1D have mild-to-moderate gastroparesis that can be managed with good nutrition and hydration from food.
For more severe forms of it, metoclopramide, a drug that is effective against vomiting and nausea, and erythromycin, an antibiotic, have been shown to increase the rate of stomach emptying. Metoclopramide, however, comes with side effects—some of which are severe.
A small percentage of individuals with T1D, though, have severe gastroparesis that is characterized by malnutrition, weight loss, and frequent hospitalizations. People who do not respond to conventional therapies can become dependent upon feeding tubes for nutrition.
For people living with type 1 diabetes (T1D), eye disease is a troubling reality—over 40 percent develop diabetic eye disease, sometimes resulting in blindness. But early detection and timely, appropriate treatment can reduce the risk of blindness and complications by over 95 percent.
Some of our Breakthrough T1D staff members living with T1D have experienced eye disease. Below, they share their experiences with diabetic eye disease to help raise awareness about the importance of being proactive when it comes to vision health.
Arielle Cilaire
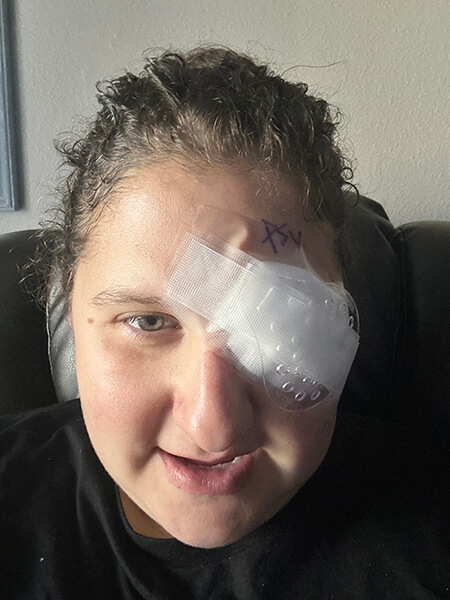
For years I had seen an ophthalmologist to check my eyes. During the pandemic, the office closed, and I had to search for a new one. I ended up making an appointment with a retinal specialist. This was 2021—I had just given birth in 2020. The retinal specialist told me something I never thought I would hear. He said, “You have retinopathy in both eyes, stronger in your left.” I never had signs of retinopathy before, or so I thought. He shared that pregnancy can cause retinopathy to progress. I told him how I happened in his office by chance, and he said that all people with T1D should see a retinal specialist. Most ophthalmologists don’t have the right machines to view the full eye.
A year later, I had laser, eye injections, and a Vitrectomy surgery. After my surgery, my new doctor said, “We changed the course of your retinopathy.” I’m not very old, but my age of diabetes is 30 years and the technology for most of my diabetic life wasn’t great. However, now technology and options are amazing for our eye health and diabetes altogether. This is all thanks to Breakthrough T1D. Please go see a retinal specialist and let’s change the course of diabetic eye health together!
Grace Bennett
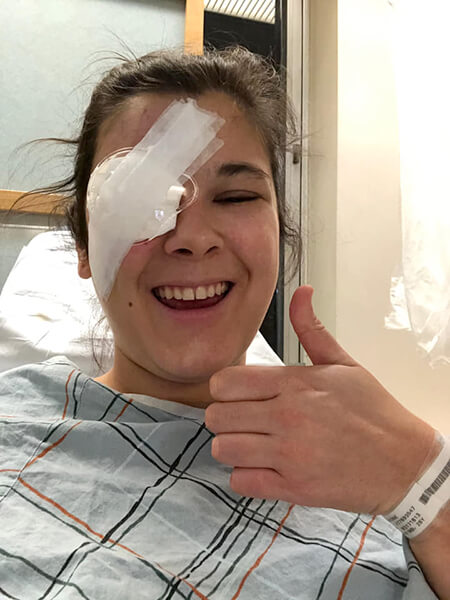
Experiencing eye complications (or any complications) because of T1D is a terrifying thing to go through. Beyond the fear of the unknown and the harsh reality that you’re possibly losing something crucial to your independence and everyday life, the stigma surrounding T1D complications makes going through them much harder.
When I first experienced complications with my vision, the shame and isolation I felt were immeasurable. I thought that it was all my fault and that I couldn’t really complain because it HAD to be because of something I did. My retinal specialist told me that living with T1D over a long period of time (over 20 years in my case) can also cause complications and that they aren’t only caused by neglecting T1D management. Hearing that, I felt much better and was able to focus on addressing the complication itself.
If you meet someone who shares that they have experienced T1D complications don’t use them as some sort of cautionary tale about T1D management. Listen with compassion and understanding—after all, we’re all doing the best we can managing this awful disease.
Liz Walsh
If I could give one piece of T1D advice to my younger self, it would be to not take your eye health for granted. Living with T1D for 30+ years, I learned how quickly things can change (literally) in the blink of an eye. I received a diagnosis of retinopathy that required multiple surgeries, laser treatments, and eye injections to stabilize. It was scary but thankfully, with the treatments and technology available, there was light at the end of the tunnel.
Make (and keep) the appointments to monitor your eye health—the earlier you start this in your T1D journey, the better. Complications can happen even with great control because T1D doesn’t play fair, but taking action to prevent, detect, and manage them early can help keep your future looking bright.
Read Liz’s full story here.
Nate Keeney

In 2017, I woke up with blood in my vision and scared not knowing what would happen next. Three eye surgeries later, I live with just one working eye and one doing the best it can. Going to the eye doctor may be scary. Getting drops, poked, and prodded may seem like a lot, but getting checked (even if you have a good A1c) outweighs going through a more complicated journey.
Even with all that I have been through with my eye, I still live a great life adjusting to what I have left with my vision. Don’t let fear keep you away from having good vision and preventing any major complications!
Learn more about reducing your risk of developing diabetic eye disease.
Through the Mary Tyler Moore Vision Initiative, a special initiative of the Mary Tyler Moore and S. Robert Levine Charitable Foundation and Breakthrough T1D launched to honor Mary’s contributions to diabetes awareness and research, Breakthrough T1D is supporting research to find new ways to prevent the development of diabetic retinal disease, arrest its progression to preserve visual function, and restore lost vision.

