Nearly 3 years ago, Breakthrough T1D joined forces with Civica Rx, a non-profit pharmaceutical company, to manufacture and distribute low-cost insulins. Starting on January 1, 2026, the first of those insulins, insulin glargine-yfgn (a long-acting insulin interchangeable with Lantus®), will be available for purchase.
5 pens, no more than $55 – for anyone

While we have seen improvements in the affordability and accessibility of insulin in recent years, Americans who need insulin are still often required to jump through hoops to get the assistance they need to afford their insulin. Civica’s insulin is very simple: it’s no more than $55 for 5 pens, and anyone with a prescription can purchase it at a pharmacy. There are no forms to fill out, co-pay assistance programs to sign up for. It’s that simple.
This insulin is manufactured by BioCon and will be sold as Civica insulin glargine. In California, it will carry the “CalRx” brand.
Civica has shared with us that they expect broad retail availability. In the coming weeks, we look forward to sharing information on where this insulin can be purchased.
A day 3 years in the making
Breakthrough T1D and its amazing advocates have long championed the cause of affordable insulin, tirelessly advocating across multiple fronts—including Congress, employers, health insurance companies, and pharmaceutical manufacturers.
Civica’s announcement marks another significant milestone in the journey toward insulin affordability. This development joins other encouraging progress made in recent years, such as the implementation of a $35 monthly insulin cap for Medicare recipients and the decision by the three major insulin manufacturers to lower their prices.
Despite these advances, Breakthrough T1D continues to advocate for and advance comprehensive legislation that guarantees insulin will remain accessible and affordable for all, regardless of insurance status or income level. Access to insulin is not just a policy issue—it’s a mission priority. We must continue to work to ensure that people with T1D remain healthy and well-supported as the search for cures continues.

“For people with type 1 diabetes, insulin is a lifesaving drug. Too often, the cost of insulin is a barrier that causes people to make difficult decisions between their medication and other necessities,” said Aaron J. Kowalski, Ph.D., Breakthrough T1D CEO. “The availability of Civica’s insulin glargine is a significant next step in ensuring that insulin is accessible and affordable for people with type 1 diabetes and all those who need it, regardless of their insurance status. Breakthrough T1D is proud to partner with Civica as they continue their efforts to develop affordable insulins for the diabetes community.”
More insulins are coming
Civica has not strayed from its initial goal: to sell biosimilar insulin options for the three most-prescribed insulins: glargine (Lantus®), lispro (Humalog®), and aspart (Novolog®). Aspart will be the next insulin sold by Civica and we look forward to future updates about its progress.

ADA Recap Series
This article is the first of our three-part ADA Recap Series. Breakthrough T1D was on site in Chicago, IL from June 20-23 for the American Diabetes Association’s (ADA) 85th Scientific Sessions. We’re here to report on the latest-and-greatest type 1 diabetes (T1D) advancements—including many driven by Breakthrough T1D funding. Look out for tomorrow’s article for updates on Cures.
Improving Lives
Breakthrough T1D’s Improving Lives program focuses on devices, insulins, adjunctive therapies, treatments for complications, and psychosocial interventions to improve the health and quality of life of people living with T1D.
Adjunctive therapies and complications
There was significant focus on GLP-1 receptor agonists (GLP-1RAs) and SGLT inhibitors (SGLTi) in reducing long-term complications and improving glycemic control in people with T1D.

GLP-1 receptor agonists
Glucagon-like peptide 1 receptor agonists mimic the hormone GLP-1, which elevates insulin and regulates appetite. Examples include Ozempic® (semaglutide) and Mounjaro® (tirzepatide), which acts on both GLP-1 and a similar target, GIP.

SGLT inhibitors
Sodium-glucose cotransporter inhibitors target kidney cells to prevent them from reabsorbing glucose into the blood so it gets excreted as waste. Examples include Farxiga® and Zynquista®.
While SGLTi and GLP1-1RAs have proven effective for heart and kidney disease in type 2 diabetes (T2D) and in people without diabetes, people with T1D have often been excluded from critical trials. Thanks to years of advocacy and support from Breakthrough T1D, T1D trials are ongoing—and real-world evidence suggests that GLP-1RAs and SGLTi could be impactful in the T1D community as well.
Real-world evidence for GLP-1RA use in T1D
- Presenter: Ildiko Lingvay, M.D.; University of Texas Southwestern
- People with T1D have self-reported that they decided to try GLP-1RAs for weight loss and improved glycemic control.
- Real-world evidence suggests that GLP-1RAs have a clinically meaningful impact on weight and reduced insulin dose.
- While GLP-1RAs are generally safe, some people have stopped use because of gastrointestinal side effects. These side effects are also seen in people with T2D and people without diabetes.
A review of SGLTi and GLP-1RAs in reducing chronic kidney disease (CKD) in T1D
- Presenter: David Cherney, Ph.D.; University of Toronto
- In the EMPA-KIDNEY trial that included non-diabetes participants and people with T1D or T2D, empagliflozin (SGLTi) improved kidney health in people with T1D.
- In the ATTEMPT trial, dapagliflozin (SGLTi) improved time in range (TIR), reduced HbA1c levels, and had positive effects on kidneys in youth with T1D.
- The Breakthrough T1D-funded enrolling phase 3 SUGARNSALT trial is testing whether sotagliflozin (SGLTi) can prevent progression of moderate to severe kidney disease in people with T1D, and it includes careful diabetic ketoacidosis (DKA) risk mitigation strategies.
- The SEMA-AP trial found that semaglutide (GLP-1RA) increases TIR in people with T1D when used alongside an AID system.
- The Breakthrough T1D-funded recruiting phase 2 REMODEL-T1D trial is testing if semaglutide (GLP-1RA) can improve kidney health in people with T1D.

Glucokinase
Glucokinase (GK) is an enzyme in liver cells that works in an insulin-dependent manner to regulate blood sugar. In people with T1D who have little insulin reaching the liver, GK can’t work as normal, contributing to higher blood sugar.
Use of a glucokinase activator for glycemic control
- Presenter: Klara Klein, M.D., Ph.D.; University of North Carolina
- In the phase 1/2 SimpliciT1 study, people with T1D who received the GK activator TTP399 showed improvements in blood glucose with fewer hypoglycemic events.
- A different study found that TTP399 does not increase the risk for DKA.
- These studies were done in collaboration with vTv Therapeutics, a company with funding and support from Breakthrough T1D and the T1D Fund: A Breakthrough T1D Venture. The phase 3 CATT1 study for TTP399 is testing whether it can reduce moderate to severe hypoglycemic events in people with T1D.
Adjunctive therapies and complications highlight: Breakthrough T1D-funded research
Halis Kaan Akturk, M.D. (University of Colorado), Janet Snell-Bergeon, Ph.D., MPH (University of Colorado), and Viral Shah, M.D. (Indiana University) presented findings from the Breakthrough T1D-funded ADJUST-T1D clinical trial, which was recently published in the New England Journal of Medicine Evidence. The trial tested whether semaglutide (GLP-1RA) can improve glycemic and weight outcomes in adults with T1D and obesity who are using an AID system. 36% of people treated with semaglutide met the primary endpoints of TIR greater than 70%, time below range less than 4%, and weight loss of 5% or more compared to the placebo, and the drug was well-tolerated and safe. This trial represents critical evidence for use of a GLP-1RA as a potential way to manage both glycemic control and weight in people with T1D.
Ye Je Choi, MPH (University of Washington) reported on the CROCODILE study, which examined metabolic alterations in kidneys of people with T1D. She observed early structural and metabolic changes in kidneys that occurred before the onset of clinical kidney disease and associated structural damage. Her work could contribute to the development of biomarkers that can help predict the onset of kidney disease in people with T1D before it occurs.
Jeremy Pettus, M.D. (University of California at San Diego) conducted a phase 2 clinical trial to address insulin resistance in people with T1D. External insulin therapy can increase levels of insulin in the blood relative to glucose, which reduces sensitivity and may contribute to cardiovascular disease (CVD). Treatment with the glucagon receptor antagonist volagidemab, which prevents the liver from releasing glucose into the blood, reduces insulin requirements by 15%, resulting in improved glycemic control and insulin sensitivity without changes in bodyweight.
Schafer Boeder, M.D. (University of California at San Diego) worked with Dr. Pettus on a phase 1/2 clinical trial that tested whether the addition of SGLTi to the glucagon receptor antagonist volagidemab can further improve glycemic control in people with T1D. The combination of therapies increased TIR up to 86% from 70% and reduced daily insulin use by 27%. Further research is needed to better understand the safety profile of this regimen.
Justin Gregory, M.D. (Vanderbilt University) worked with Dr. Pettus and Dr. Boeder on the above study. He also presented on the use of GLP-1RAs and dual GLP-1/GIP receptor agonists for reducing complications in T1D.
Key takeaways
Clinical trials with GLP-1RAs and SGLTi are providing encouraging evidence about these therapies’ potential to improve long-term health in people with T1D. Breakthrough T1D is working toward a future where these drugs are an option for people with T1D to better manage their blood sugar and reduce complications.
Devices
Real-world insights from Automated Insulin Delivery (AID) systems
- Presenter: David Maahs, M.D., Ph.D.; Stanford University
- Based on published real-world evidence for AID systems in people with T1D, TIR is increased by an average of eight to 15% from baseline in a range of studies across various systems.
- Youth with T1D have better glycemic control and reduced rates of DKA with AID systems. Those with lower TIR at the start of AID system use see the greatest improvements.
Real-world evidence: iLet Bionic Pancreas AID system
- Presenter: Steven Russell, M.D., Ph.D.; Beta Bionics
- The iLet Bionic Pancreas contains a continuously adapting algorithm that automatically determines insulin doses. No carbohydrate counting is required, and meals are only announced as breakfast, lunch, and dinner.
- Data was collected from 16,000 users over two years.
- Users achieved an average HbA1c level of 7.3%, down from 8.9%. This is accompanied by low rates of hypoglycemia and significantly improved self-reported quality of life.
Continuous ketone monitoring: Innovations and clinical applications
- Presenters: Ketan Dhatariya, MBBS, M.D., Ph.D. (Norfolk and Norwich University Hospitals), Lori Laffel, M.D., MPH (Harvard University), Jennifer Sherr, M.D., Ph.D. (Yale University), and Richard Bergenstal, M.D. (HealthPartners Institute).
- DKA rates are increasing in the U.S., but mortality rates from DKA are decreasing.
- The history of continuous glucose monitoring (CGM) offers a roadmap for continuous ketone monitoring (CKM) development, showing how early skepticism gave way to broad clinical impact.
- CKM could allow for earlier detection of rising ketones to prevent DKA. CKM also has the potential to identity infusion set failures, be a valuable addition to AID systems, help monitor early-stage T1D, and more.
- Five new studies funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) will develop CKM for safe and effective use of SGLTi in T1D.
- Tandem, Beta Bionics, Sequel MedTech, and Ypsomed announced plans to integrate Abbott’s dual glucose ketone sensor into their AID systems.
Making the case for time in tight range
- Presenter: Gregory Forlenza, M.D.; University of Colorado
- Dr. Forlenza presented on the benefits and challenges of time in tight range (TITR), also known as time in normal glycemia (TING), defined as blood glucose levels between 70-140 mg/dL.
- TITR will likely be more clinically beneficial than TIR as fluctuations outside of TITR may be better predictors of complications and offer a better therapeutic window for intervention.
- More research is needed to advance therapeutics that will allow people with T1D to achieve TITR before it can be integrated into clinical decisions.
Devices highlight: Breakthrough T1D-funded research
Erin Cobry, M.D. (University of Colorado) presented the results of a Breakthrough T1D-funded clinical trial evaluating an artificial intelligence-powered AID algorithm designed to not require meal announcements. She showed that this algorithm (used without meal announcements) improved overnight TIR, and provided equivalent daytime TIR, compared to participants’ usual care. A major goal for Breakthrough T1D is to advance AID systems that do not require meal announcements to improve both glucose outcomes and quality of life for people with T1D.
Key takeaways
Devices have transformed how this disease is managed. Systems are becoming easier to use with less user input—and, critically, people with T1D are doing better. This is the dream Breakthrough T1D had when we launched the Artificial Pancreas project 20 years ago. We will continue to drive toward our goal of developing systems that provide superior health outcomes with minimal user burden.
Insulins
Inhaled insulin treatment for youth with T1D
- Presenter: Michael Haller, M.D.; University of Florida
- Afrezza® is an inhaled, fast-acting insulin that has proven to be effective in adults.
- The phase 3 INHALE-1 study examined Afrezza® in youth with T1D. Users report greater treatment satisfaction and no increase in weight compared to injected rapid-acting insulin analogs.
- Afrezza® is safe for youth with T1D. The most common adverse events were pulmonary-related, such as coughing.

Breakthrough T1D’s Improving Lives team making an impact
Courtney Ackeifi, Ph.D., Senior Scientist, hosted an Improving Lives Happy Hour with Breakthrough T1D-funded researcher Jeremy Pettus, M.D. The discussion included research priorities for adjunctive (non-insulin) therapies for people with T1D and their healthcare providers. They also discussed the importance of industry partnerships and the role of Breakthrough T1D in driving these relationships, which can accelerate new T1D therapies toward the clinic.
Dr. Ackeifi also spoke at the ADJUST-T1D trial update, contextualizing the use of adjunctive therapies like GLP-1RAs for superior glucometabolic control in people with T1D.
Look out for tomorrow’s article for an update on Cures research presented at ADA 2025!
While we look back on 2024, we can reflect upon the incredible progress we’ve made in advancing breakthroughs toward cures and improving everyday life with T1D.
This wouldn’t have been possible without each and every one of you and your continued support of our mission as we drive toward cures for T1D.
Here are the top 11 advances that together, we made happen in 2024:
Breakthrough T1D announced the launch of Project ACT, an initiative aimed at accelerating breakthroughs in T1D cell replacement therapies that do not require broad immunosuppression. Recent advances, such as Vertex’s stem cell-derived islets, have been made possible by Breakthrough T1D’s Cell Therapies program as part of our drive toward cures. The goal of Project ACT is to push research, development, regulatory policies, access, and adoption to increase the rate at which cell therapies without the need for broad immunosuppressants will become available to people with T1D.
Why this matters: Immunosuppressive drugs are a barrier to access to cell replacement therapies because of their toxic side effects, which is why islet transplants are currently only available to people with severe low blood sugar (hypoglycemic) unawareness and episodes. By striving toward a future where we realize the benefits of cell replacement therapies without the downsides of the current regimen of immunosuppressants, we will make islet replacement therapies broadly accessible to the T1D community.
Vertex’s clinical trial of VX-880, a first-generation stem cell-derived islet replacement therapy for people with severe hypoglycemia (requiring the use of immunosuppressants), has transitioned into a phase 1/2/3, or pivotal, trial. This news comes after Vertex shared incredibly promising data in the earlier phases of the trial, including 11 of 12 participants reducing or eliminating the need for external insulin.
The upcoming trial will expand to 50 people who will get a single, target dose of VX-880. The primary endpoint will be insulin therapy independence without severe hypoglycemic events after one year. This is the final clinical testing stage before Vertex can seek FDA approval.
Breakthrough T1D has a decades-long relationship with Vertex and the leading scientists behind stem cell-derived islet therapies, an advancement that would not have been possible without Breakthrough T1D funding and support. The T1D Fund had invested in Semma Therapeutics, which was acquired by Vertex Pharmaceuticals in 2019, eventually leading to the active clinical development of VX-880 in T1D.
Why this matters: This is the first time a scalable cure for T1D is entering phase 3 clinical trials—a significant win and a huge step toward accelerating the delivery of cell therapies to members of the T1D community!
Tegoprubart: Transplant Survival Without Standard Immunosuppressive Drugs
Tegoprubart, an anti-CD40L immunotherapy that limits the immune response, is being tested in a Breakthrough T1D-funded study in people with T1D and severe hypoglycemia who have received deceased donor islets. Eledon Pharmaceuticals announced promising initial results in which two of three people achieved insulin therapy independence. According to the study, tegoprubart is safer for both people and transplanted cells in comparison to broad immunosuppressants, with milder side effects and greater islet survival. To further support this effort, the T1D Fund: A Breakthrough T1D Venture invested in Eledon.
Cell Pouch: A Home for Transplanted Islets
Breakthrough T1D has been supporting the development of Cell Pouch, an implantable device from Sernova that provides a safe, immune-protected environment for transplanted islet cells. In phase 1/2 clinical trials, all six people who received donor islets within the Cell Pouch achieved sustained insulin therapy independence with immunosuppressants, including long-term islet survival and function over five years without harmful side effects.
Why this matters: Standard of care immunosuppressive drugs that help avoid transplant rejection come with unwelcome side effects, such as increased risk of infection and malignancy and toxicity to kidneys, nerves, and islet cells themselves. Breakthrough T1D is focused on finding alternative ways to keep transplanted islet cells alive and healthy so that cell replacement therapies can become more tolerable and accessible.
In a major effort spearheaded by Breakthrough T1D, the first internationally recognized clinical guidelines for those who test positive for T1D autoantibodies have been published. These include guidance on monitoring frequency, education, and psychosocial support in addition to recommended actions for healthcare professionals (HCPs) when the risk of T1D progression is high. The guidelines were cooperatively developed with over 60 international experts spanning ten countries.
Why this matters: Previously, there had been no consensus on monitoring guidelines for people who test positive for T1D autoantibodies. Standardization of clinical recommendations means that individuals, families, and HCPs have tangible next steps to monitor early T1D progression and catch life-threatening complications sooner.
- Breakthrough T1D is leading a campaign to secure a recommendation for T1D screening from the U.S. Preventative Services Task Force (USPSTF), the main authority for preventative care. Approval would require T1D screening to be covered by insurance—an important step forward in expanding access.
- Driven by Breakthrough T1D’s advocacy efforts, The Centers for Medicare and Medicaid Services (CMS) established a unique ICD-10 code for stage 2 T1D. ICD-10 codes are used by HCPs to classify and document diagnoses, symptoms, and procedures. These codes provide a unified way for doctors and providers to indicate what diseases or conditions a person has in their electronic health record (EHR), empowering HCPs to document accurate diagnoses and provide the best possible care.
Why this matters: T1D early detection is critically important to prevent life-threatening complications at diagnosis and to give people necessary resources to make informed decisions about their health. Integrating T1D screening into the U.S. healthcare system will increase access to care.
The past year has seen some important advances in glucose management therapies and devices:
- Cadisegliatin, an activator of a blood sugar regulator in the liver, is being investigated in a phase 3 clinical trial (TTP399) as an adjunct therapy to insulin for people with T1D, although it is currently placed on clinical hold. vTv Therapeutics, the trial sponsor, is also a T1D portfolio company.
- The Omnipod 5 app is now available for the iPhone, making it easier to control the Omnipod without the need to carry a controller. It can also integrate with the Dexcom G6 continuous glucose monitor (CGM).
- Eversense 365 is the first FDA-approved year-round sensor that can easily integrate with automated insulin delivery (AID) systems. Other sensors require replacement after 10-14 days.
Why this matters: While advancements in glucose management have been pivotal in improving health outcomes for people with T1D, access remains a challenge. AID systems are globally underutilized, and not everyone has the necessary technology to connect devices. Breakthrough T1D is working to not only support advances in glucose management but also increase access.
Related content: While Breakthrough T1D consistently strives to improve the lives of those living with T1D, as an organization we have made incredible progress in the development of AID systems, also called the artificial pancreas systems. Read a historical perspective written by Breakthrough T1D volunteer Doug Lowenstein that covers conception to FDA approval of the first artificial pancreas systems, which changed the lives of people with T1D.
An inquiry spearheaded by the Breakthrough T1D affiliates in the U.K. uncovered risks of developing T1D eating disorders (T1DE), including bulimia, anorexia, or insulin restriction to lose weight. There is a significant gap in education and clinical guidelines for HCPs, a lack of internationally recognized criteria for T1DE diagnosis, and insufficient care integration, leading to preventable complications and healthy years of life lost. Breakthrough T1D recognizes the importance of spreading awareness and support for T1DE, and much work is needed to improve the lives of those living with T1DE.
Why this matters: There is an urgent need to change the way T1DE is approached, including integrated physical care with mental health services to get people with T1DE the access to care that they need.
In a study that included people with T1D, finerenone (Kerendia®) has been shown to improve cardiovascular outcomes in adults with heart failure. The drug is already approved in the U.S. to treat kidney and cardiovascular disease in people with T2D. Based on these results, Breakthrough T1D is supporting a clinical trial (FINE-ONE) in conjunction with Bayer to investigate the use of finerenone for T1D with the hopes of reducing kidney complications.
Why this matters: Kidney and cardiovascular disease remain significant challenges for those with T1D, especially given the FDA’s recent rejection of an SGLT inhibitor to lower blood glucose in people with T1D and chronic kidney disease. Yet, a new clinical trial (SUGARNSALT) will better assess the benefits versus risks.
Breakthrough T1D is advocating for the regulatory approval of C-peptide, a biomarker for insulin production by beta cells, to be used as an endpoint in clinical trials. An endpoint can accurately predict a meaningful benefit in clinical trials for disease-modifying therapies (DMTs; treatments that can slow, halt, or reverse T1D). To support this endeavor, Breakthrough T1D scientists and an expert consensus panel published research with evidence supporting C-peptide as an endpoint. Breakthrough T1D is continuing to engage with regulators, coordinate with industry, and assess more clinical trial data to drive this effort forward.
Why this matters: Current clinical trial endpoints (HbA1c, hypoglycemia, and complications) are not the best way to gauge the clinical benefits of T1D therapies. If C-peptide gets regulatory approval to be used as an endpoint, clinical trials could be smaller and shorter while still accurately assessing the advantages of a DMT. This means that drug development can move more quickly, and people with T1D will be able to access therapies sooner.
Related content: Two years ago, the T1D community received the incredible news that Tzield® had become the first FDA-approved disease-modifying therapy that can significantly delay T1D onset. Breakthrough T1D volunteer Doug Lowenstein recounts the life-changing drug’s journey nearly 100 years after the discovery of insulin.
The T1D Index is a data simulation tool that measures the global health impact of T1D, bridging gaps in our knowledge of public health statistics. T1D Index 2.0 has new and improved functionality, including advanced simulation capabilities, validation of data, and enhanced user experience. Breakthrough T1D contributed to both the development and improvement of the T1D Index.
Why this matters: The T1D index is critical in defining the intercontinental scope of T1D, driving us toward country-specific solutions and improved global health outcomes.
Earlier this year, JDRF rebranded to Breakthrough T1D. While our mission remains the same, our name needs to better reflect who we are and where we’re going. Our new brand aligns with our mission to accelerate life-changing breakthroughs for those of every age living with T1D as we work toward a world without it.
Why this matters: The proof is in the name—each day we strive to increase and accelerate breakthroughs in T1D, and it’s critical for our brand to accurately reflect our mission.
It’s certainly been an exciting year! While we still have more work to do, it’s crucial to celebrate our wins, both big and small, to see how far we’ve come in our push to make T1D a thing of the past.
Together, we’re accelerating breakthroughs for people with T1D, and the support of the T1D community drives our mission forward every single day, leading the way to lifechanging therapies and cures. Let’s see what 2025 has in store!
My name is Phyllis Kaplan, and I’m the Greater New England Chapter Community Engagement Manager at Breakthrough T1D (formerly JDRF). I have lived with type 1 diabetes (T1D) for fifty years and have seen first-hand many advancements over the last five decades that have made managing T1D a little easier.
A different way of managing T1D
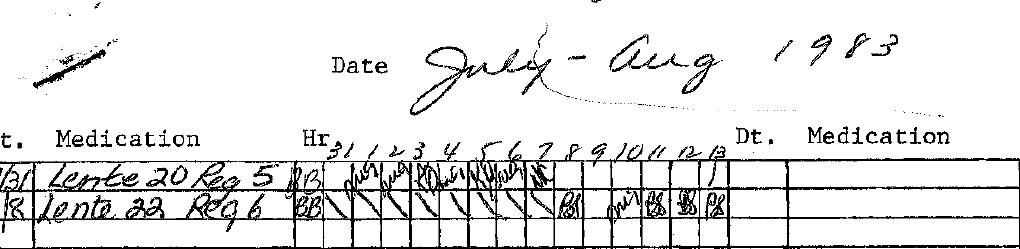
When I was diagnosed in 1974, life with type 1 diabetes looked very different than it does today. Managing T1D was much more regimented and inflexible than today’s more personalized approach. A lot more was dictated by the medical practices of the time, which didn’t leave much room for variation or spontaneity in daily life.
My parents gave me one injection of Ultralente long-acting insulin and regular insulin each day. For a short time, the insulin I took was a beef/pork mix, which soon after was just pork, until the early 1980s when synthetic insulins came on the market.
Home blood glucose testing didn’t exist then. We figured things out using a sliding scale and urine testing with Clinitest tablets, which involved a glass test tube, an eye dropper, and a reagent tablet that changed color based on how much sugar was detected. The mixture quickly turned colors ranging from blue, which meant no sugar, to bright orange, signifying a lot of sugar. To this day, blue is my favorite color, and I can’t help but wonder if this has anything to do with it.
There was also no carbohydrate counting like we do today. Instead, we were given strict meal plans that divided food into categories like breads, fats, proteins, vegetables, and milk, with a set number of calories for each day.
A bright future ahead
The innovations since then have each made a real difference. We have gone from testing urine sugar levels and strict diets to using continuous glucose monitors that provide real-time glucose levels and automated insulin delivery systems that can correct high blood sugar or mitigate low blood sugar. It’s truly amazing! While none of these are perfect solutions, each step brings us closer to a better quality of life.
As good as the technology is now, though, people living with T1D and their caregivers still have to manage a lot of things related to the disease; it’s just a different type of work. Even on a great day, T1D hums along in the background. The constant mental burden—alarms, carb-counting, and decision fatigue—never goes away.
But every time I meet a newly diagnosed child or talk to a parent managing T1D with tools my parents couldn’t have dreamed of 50 years ago, I’m reminded why this work matters. We’ve come so far, and the future is even brighter.
To those who came before me, those walking this path alongside me, and those just beginning their journey, life with T1D can feel lonely, but know you aren’t alone. Keep going!
To the researchers and advocates working toward better treatments and, one day, cures, keep going!
Here’s to progress, resilience, and the next breakthrough!
It’s early October, which means the Nobel Prizes are being awarded in several categories. These prestigious awards recognize outstanding contributions in Peace, Literature, Physics, Chemistry, Physiology or Medicine, and Economic Sciences. They go to the best and brightest people with the best and brightest ideas.
This year, the prizes recognize work that is, in several ways, connected to Breakthrough T1D and the work we are doing to cure type 1 diabetes (T1D) and improve the lives of those living with it.
microRNAs
The 2024 Nobel Prize in Physiology or Medicine was awarded to Victor Ambros and Gary Ruvkun for the discovery of microRNA and its role in post-transcriptional gene regulation.
But what is it?
The DNA in our body has between 20,000 and 25,000 genes. Our body constantly activates those genes, which allows it to function. For example, there is a gene that tells the beta cells to start creating insulin in response to elevated blood glucose levels.
This is called gene expression—it’s when the body produces something, usually a protein, to do something specific in the body. Gene expression happens when the DNA of a gene is copied to form messenger RNA, or mRNA, which is then turned into a protein that has a unique function. This is orchestrated in a highly tissue-specific manner, creating diverse cell types like muscle, nerve, beta cell, and more.
In 1993, we learned that molecules called microRNAs regulate gene expression. They can hamper or promote it. By measuring the presence of microRNAs and using them as a biomarker, we can learn a lot about what is going on inside the body, what genes are being expressed, and in which cell types.
How does this apply to type 1 diabetes?
Breakthrough T1D is actively funding research into the use of microRNAs to advance our mission. In fact, we are currently funding $2.6 million in grants related to microRNAs. Specifically, we’re exploring their use as biomarkers of disease progression and as a therapeutic target to interfere with the autoimmunity behind T1D.
Biomarkers
Biomarkers are, put very simply, things in the body that can be measured. Pulse, body temperature, and blood pressure are all biomarkers. In T1D, there are a handful used in common parlance: blood glucose level, HbA1c, time-in-range, autoantibody status, and more. We want to validate the use of microRNAs as a biomarker of disease progression.
We know through years of work that early detection has many benefits. This includes being educated on what T1D is, preparing for a potential diagnosis, the ability to benefit from a disease-modifying therapy to delay onset, and preventing potentially life-threatening complications at onset. microRNAs have the potential to increase our understanding of disease progression and identify how quickly a person is progressing.
Type 1 diabetes occurs in stages. Today, biomarkers of progression, like autoantibody status, don’t paint the whole picture. If a person tests positive for 2 autoantibodies, that means they are almost 100% likely to develop T1D at some point—but we don’t know how fast. We can measure blood glucose levels, but that is a single data point for dysglycemia. It doesn’t tell us how healthy the beta cells are.
microRNAs have the potential to paint a much more detailed picture. We are funding research to discover whether we can use microRNAs to learn exactly where a person is in their progression to stage 3 T1D and how quickly it’s happening.
In summary—we believe microRNAs can enhance the predictive value on top of the current biomarkers for T1D. They can be a very useful tool in measuring progression and helping people in stages 1 and 2 of T1D.
microRNAs as a therapeutic target
Type 1 diabetes is an autoimmune disease—a disease where the body’s immune system destroys its own cells. In T1D, the T-cells in the immune system destroy the insulin-producing cells in the pancreas.
Our research into stopping disease progression focuses mainly on two areas: modifying the immune system and modifying the beta cells. microRNAs can potentially help us fix the dysregulation in the immune system, slowing down or halting the autoimmune attack.
Breakthrough T1D-funded researchers discovered that blocking microRNA-146a in mice can prevent the T-cells from damaging the beta cells. Preventing damage to the beta cells prevents T1D from progressing. Our hypothesis: if we can block microRNA-146a, we can slow down or halt disease progression. This specific study is in a pre-clinical setting, and if it goes well, it could move into clinical trials in the near future.
David Baker – Nobel Prize in Chemistry
David Baker, a breakthrough T1D-funded researcher, was awarded half the 2024 Nobel Prize in Chemistry for computational protein design.
Baker’s prize acknowledges his work in discovering a way to create new proteins that have never existed before. These proteins can potentially be used to treat diseases, make new vaccines, and more. A few years ago, we saw a potential application to T1D.
There haven’t been any groundbreaking changes in insulin since the discovery of synthetic insulin in the late 1970’s. Breakthrough T1D wants to change that—and is funding researchers, like David Baker, to figure out what the next generation of insulins can look like.
Baker received two grants from us from 2016-2021 to use a computer model to create new, glucose-responsive insulin (GRI). GRI is insulin that can be administered, exist in the blood stream, and only be active when it is needed. Baker’s grant was one of many projects we are pursuing in GRI. This work continues to this day, and there are even GRIs in human clinical trials.
What’s next?
This year’s Nobel Prizes show that we are focusing on the same things the Nobel Committee is focused on: the best and brightest scientists and the best and brightest research. Hopefully, one day, we’ll see a T1D scientist win a Nobel Prize for curing this disease.
Want to learn more?
Visit our Funded Research page, where you can read about every single grant we fund—including David Baker’s work in GRI and the many grants currently being funded to study microRNAs.